USEFUL DEFINITIONS
Element
A substance which cannot be split into anything simpler by chemical means.
Atom
The smallest part of an element that can take part in a chemical reaction.
Molecule
The smallest particle of a compound (a combination of two or more elements).
It is also the name given to the smallest part of those elements which do not exist
as atoms in the free state i.e.
hydrogen
nitrogen
chlorine
iodine
H2
N2
Cl2
I2
oxygen
fluorine
bromine
O2
F2
Br2
N.B. ionic compounds (e.g. sodium chloride) do not exist as molecules.
USEFUL DEFINITIONS
Ion
The name given to any electrically charged atom or molecule.
• positively charged ions are known as cations
• negatively charged ions are known as anions
Like charges repel but unlike (opposite) charges attract. If the
sum of all the positive charges is equal and opposite to all the
negative charges then the species will be neutral (no overall
charge).
Symbol
A symbol represents one atom, or one mole, of an element.
Valency
Is a numerical measure of the combining power of an
atom / ion.
Historically, it was the number of hydrogen atoms
which will combine with one atom, or group of atoms.
It is also the number of positive (+) or negative (-)
charges on an ion. e.g. Mg+2, K+, S2-
Valency may also be defined as the number of electrons
that an atom donates or accepts to form the duplet state
(i.e., 2 electrons in outermost shell) or octet state (i.e., 8
electrons in outermost shell).
The valency of an element is always a whole
number.
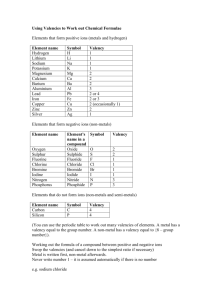
TABLE OF VALENCY
valency Element/ group losing elements
1
2
All group 1 elements, hydrogen (H), Silver
(Ag), ammonium (HN4)
All group 2 elements, Zinc (Zn),
3
All group 3 elements
4
Carbon C), Silicon ( Si),
Valencies of some elements & Radicals
Valency
Element/ group gaining electrons
1
All group 7 elements, hydroxide (OH),
Nitrate (NO3), Hydrogen carbonate (HCO3)
2
Oxygen (O), Sulphur (S), Sulphate (SO4),
Carbonate (CO3)
Elements With Variable Valencies
Element
Symbol Valency Nomenclature
Copper
Cu
1, 2
Copper [I] or Cuprous
Copper [II] or Cupric
Silver
Ag
1, 2
Silver [I] or Argentous
Silver [II] or Argentic
Mercury
Hg
1, 2
Mercury [I] or Mercurous
Mercury [II] or Mercuric
Iron
Fe
2, 3
Iron [II] or Ferrous
Iron [III] or Ferric
Tin
Sn
2, 4
Tin [II] or Stannous
Tin [IV] or Stannic
Lead
Pb
2, 4
Lead [II] or Plumbous
Lead [IV] or Plumbic
Phosphorus
P
3, 5
Sulphur
S
2, 4, 6
USEFUL DEFINITION
Formula
A formula represents one molecule of a compound, or the
simplest ratio of the ions present. As with symbols, a formula
represents a single particle or one mole of particles.
The number of atoms or groups of atoms in a formula is
given by putting a small number just below and behind the
symbol(s). As the appearance of a symbol indicates one
atom is present, a 1 isn’t written (you put NaBr not Na1Br1).
In some formulae brackets are used to avoid ambiguity.
Aluminium sulphate has the formula Al2(SO4)3 to show that
there are two Al’s to every three SO4‘s. Without the brackets
it would appear as though there were forty three O’s i.e.
Al2SO43.
Using valency to write chemical formulae
The following steps are to be considered
when writing chemical formulae
• Write each symbol
• Write the valency at the top right-hand side of
each symbol
• Cross over the valencies
• Write the crossed-over valency at the bottom
right of the other symbol.
• Where necessary simplify the ratio of
valencies
Example
what is the formula of aluminium
oxide?
Answer
1.
Al O
2.
Al3O2
3.
Al2O3
4.
Al2O3
Hence, the chemical formula of
aluminium oxide is Al2O3
.
Chemical formula of Calcium hydroxide
1.
Ca OH
2. Ca2 OH1
3. Ca1 OH2
4. Ca(OH)2
Hence, the chemical formula of calcium hydroxide
is Ca(OH)2
,
chemical formula of Magnesium oxide
1.
Mg O
2. Mg2 O2
3. Mg2 O2
4. Mg2 O2
5.
MgO
Hence, the formula of Magnesium oxide is MgO
Iron (III) Sulphate
1. Fe SO4
2. Fe3SO42
3. Fe2SO43
4. Fe2(SO4) 3
The chemical formula of Iron (III) Sulphate is
Fe (SO )
CONSTRUCTION OF FORMULAE
It is useful to learn some formulae...
Acids
Gases
hydrochloric acid
HCl
nitric acid
HNO3
sulphuric acid
H2SO4
ethanoic acid
CH3COOH
ammonia
NH3
carbon dioxide
CO2
methane
CH4
carbon monoxide CO
sulphur dioxide
SO2
Write the formulae for the following compounds
sodium sulphate
zinc carbonate
copper I nitrate
Potassium chloride
sulphuric acid
Iron II nitrate
Lead II carbonate
Calcium hydroxide
Magnesium sulphate
Practical
word equation
1. calcium carbonate + hydrochloric acid → calcium chloride
+ carbon dioxide + water
2. copper + silver nitrate = copper nitrate + silver
3. methane + oxygen = carbon dioxide + water
4. sulphuric acid + magnesium = magnesium sulphate +
hydrogen
5. copper (II) carbonate = copper (II) oxide + carbon dioxide
6. iron (II) sulphate + sodium hydroxide = iron (II) hydroxide +
sodium sulphate
 0
0