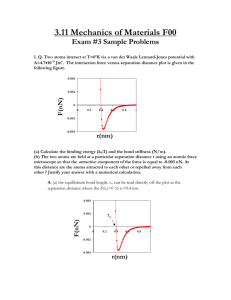
FE Review Materials Science and Structure of Matter
advertisement

FE Review
Materials Properties
Jeffrey W. Fergus
Materials Engineering
Office: 284 Wilmore
Phone: 844-3405
email: jwfergus@eng.auburn.edu
Electrical Properties
• Electrical resistance
– resistance (R) = resistivity (ρ) length (l) / area (A) l
– resistivity is materals property
– conductivity (σ) = 1 / resistivity (ρ)
• Temperature dependence: with increasing
temperature…
A
– Metals: resistance increases (conductivity decreases)
– Semiconductors: conductivity increases (resistivity decreases)
• Extrinsic: like metals in intermediate temperatures
– Insulators: conductivity increases (resistivity decreases)
Mechanical Properties
• Stress-strain relationships
– engineering stress and strain
– stress-strain curve
• Testing methods
– tensile test
– endurance test
– impact test
Stress
Normal
F
A
Shear
F
A
Tension: >0
F
A
Compression: <0
F
A
F
A
Strain
l l o l
Strain
lo
lo
Shear strain
a
l lo
lo l
h
a
tan
h
Tensile Test
thickness
Control length (l)
Strain
l l o l
lo
lo
Measure force (F) with load cell
width
length
Stress
F
F
A w t
Reduced section used to limit
portion of sample undergoing
deformation
Stress-Strain Curve
Ultimate Tensile Strength
Force decreases due to necking
Yield Point
Proportionality
Limit
Stress
Elastic Limit
Slope = E (Young’s Modulus)
Strain
Percent Elongation
(total plastic deformation)
0.2% Offset Yield Strength
Stress
0.2% offset
yield strength
Strain
0.2% strain
True/Engineering Stress/Strain
Stress
Engineering
(initial dimensions)
True
(instantaneous
dimensions)
E
F
Ao
F
T
Ai
Using
and
Ai l i Ao lo
T E 1 E
Strain
E
l l o l
lo
lo
l
dl
ln i
lo
lo l
li
T
T lnE 1
True/Engineering Stress/Strain
True
True stress does not
decrease
Stress
Engineering
Decrease in engineering stress due
to decreased load required in the
reduces cross-sectional area of the
neck.
Strain
Stress
Strain Hardening
Onset of plastic
deformation after
reloading
Strain
Plastic deformation
require larger load
after deformation.
Sample dimensions
are decreased, so
stress is even
higher
Bending Test
Four-point
F/2
F/2
Three-point
F
w
h
L
L/2
By summing moment in cantilever beam
max
3FL
2wh 2
Tension at bottom, compression at top
Hardness
• Resistance to plastic deformation
• Related to yield strength
• Most common indentation test
– make indentation
– measure size or depth of indentation
– macro- and micro- tests
• Scales: Rockwell, Brinell, Vickers, Knoop
Impact
Toughness: combination
of strength and ductility energy for fracture
Charpy V-notch
hi
hf
Fracture energy = mghi -mghf
Ductile-Brittle Failure
• Ductile
• Brittle
– little or no plastic
deformation
– cleaved fracture
surface
Ductile-Brittle
Transition Tempeature
(DBTT)
Fracture Energy
– Plastic deformation
– cup-cone / fibrous
fracture surface
Temperature
Creep / Stress Relaxation
• Load below yield strength - elastic deformation only
• Over long time plastic deformation occurs
• Requires diffusion, so usually a high-temperature
process
• Activation energy, Q (or EA)
Q
EA
creep rate A exp
A
exp
RT
kT
Creep /Stress Relaxation
Creep
F
F
time
F
F
fixed load
Stress Relaxation
time
fixed strain
Permanent deformation
Fatigue
Repeated application of load - number of cycles, rather
than time important.
max
min
ave
Stress
0
Fatigue Limit
(ferrous metals)
max
Number of Cycles to Failure
0
min
Corrosion Resistance
• Thermodynamics vs. Kinetics
– Thermodynamics - stable phases
– Kinetic - rate to form stable phases
• Active vs. Passive
– Active: reaction products ions or gas - non protective
– Passive: reaction products - protective layer
• Corrosion resistance
– Inert (noble): gold, platinum
– Passivation: aluminum oxide (alumina) on aluminum,
chromia on stainless steel
Electrode Potential
• Tendency of metal to give up electron
• Oxidation (anode)
– M = M2+ + 2e- (loss electrons)
• Reduction (cathode)
– M2+ + 2e- = M (gain electrons)
• LEO (loss electrons oxidation) goes GER (gain
electrons reduction)
Corrosion Reactions
• Oxidation - metal (anode)
– M = M2+ + 2e-
• Reduction - in solution (cathode)
– 2H+ + 2e- = H2
– 2H+ + ½O2 + 2e- = H2O
– H2O + ½O2 + 2e- = 2OH-
• Overall Reactions
– M + 2H+ =M2+ + H2
– M + 2H+ + ½O2 = M2+ + H2O
– M + H2O + ½O2 = M2+ + 2OH- = M(OH)2
Electromotive Force
• Gibbs Free Energy (ΔG) =-nFE (Electromotive Force)
– n = number of electrons, F = Faraday’s Constant
– Favorable: Energy decrease (-) = positive voltage
•
•
•
•
Fe2+ + 2e- = Fe: Ered = +0.440 V
Fe = Fe2+ + 2e-: Eox = -0.440 V
H2O = 2H+ + ½O2 +2e-: Ered = +1.229 V
Fe + 2H+ + ½O2 = Fe2+ + H2O: E = 0.789 V
– E does not change with number of moles (ΔG does)
– E must be corrected for non-standard state
• Concentration of H+ (i.e. pH), oxygen pressure…
Galvanic Corrosion / Protection
• At joint between dissimilar metals
– reaction rate of active metal increases
– reaction of less active metal decreases
• Galvanic corrosion
– high corrosion rate at galvanic couple
• presence of Cu increase the local corrosion rate of Fe
• Galvanic protection
– Galvanized steel
Fe
Cu
• presence of Zn decreases the local corrosion rate of Fe
– Galvanic protection
Zn
• Mg or Zn connected to Fe decrease corrosion rate
Fe
Waterline Corrosion
• Oxygen concentration in water leads to variation in
local corrosion rates
Higher corrosion rate near
oxygen access
Rust just below water
surface
Rings of rust left from water
drops
Materials Processing
• Diffusion
• Phase Diagrams
–
–
–
–
Gibb’s phase rule
Lever rule
Eutectic system / microconstituents
Fe-Fe3C diagram (ferrous metals)
• Thermal-mechanical processing
Diffusion
•
•
•
•
Atoms moving within solid state
Required defects (e.g. vacancies)
Diffusion thermally activated
Diffusion constant follows Arrhenius relationship
Activation Energy
Q
EA
D Do exp
Do exp
RT
kT
Gas constant
Temperature
Boltzman’s
constant
Steady-State Diffusion
C
J D
• Fick’s first law (1-D)
x
• J = flux (amount/area/time)
C
• For steady state
J D
x
mass
m 2 m3 mass
J
s m m 2s
C
x
Phase Equilibria
• Gibb’s Phase Rule
• P + F = C + 2 (Police Force = Cops + 2)
–
–
–
–
P = number of phases
F = degrees of freedom
C = number of components (undivided units)
2: Temperature and Pressure
• One-component system
– F=1+2-P=3-P
• Two-component system
– F=2+2-P=4-P
• Two-component system at constant pressure
– F=2+1-P=3-P
“2” becomes “1” at constant pressure
Pressure-Temperature Diagram
Two-phase line: Change T (P)
require specific change in P (T)
(F=1)
Pressure
water
ice
water
vapor
Temperature
One component: H2O
If formation of H2 and O2 were
considered there would be two
components (H and O)
Single-phase area: can change T
and P independently
(F=2)
Three-phase point: One occurs at
specific T and P (triple point)
(F=0)
Phase Diagrams
Two-component @ constant pressure
Three-phase - horizontal line
Peritectic
L +solid (d) solid ()
d
dL
Temperature
d
L
Eutectic
L 2 solids ( + b)
bL
L
b
a
Eutectoid
solid () 2 solids (a + b)
ab
a
A
Composition (%B)
B
b (pure B,
negligible
solubility of A)
Lever Law
• Phase diagram give compositions of phases
– two-phase boundaries in 2-phase mixture
• Mass balance generate lever law
Temperature
Solid
Comp.
(XS)
Alloy
Comp.
(Xalloy)
Liquid Opposite arm over total length
Comp.
(XL)
Right arm for solid
%solid
L
%liquid
Composition (%B)
X L XS
Left arm for liquid
S
A
X L X alloy
B
X alloy X S
X L XS
70 wt% Pb -30 wt% Sn
A ssessed P b - Sn p h ase d i ag r am .
At 183.1°C
First solid
%liq.61.8%Sn
256°C
30%Sn( alloy ) 18.3%Sn( Pb )
30%
61.8%Sn( liq.) 18.3%Sn( Pb )
L
% prim.Pb18.3%Sn
12.8 wt% Sn
61.8%Sn( eut .) 30%Sn( alloy )
70%
61.8%Sn( eut .) 18.3%Sn( Pb )
(Pb)
70 wt% Pb -30 wt% Sn
A ssessed P b - Sn p h ase d i ag r am .
At 182.9°C
First solid
%b97.8%Sn
256°C
30%Sn( alloy ) 18.3%Sn( Pb )
15%
97.8%Sn( liq.) 18.3%Sn( Pb )
(Pb)
Eutectic
(Pb)+β
%Pb phase18.3%Sn
12.8 wt% Sn
97.8%Sn( liq.) 30%Sn( alloy )
85%
97.8%Sn( liq.) 18.3%Sn( Pb )
Microconstituents
Primary Pb
%Prim.Pb18.3%Sn
61.8%Sn( eut .) 30%Sn( alloy )
70%
61.8%Sn( eut .) 18.3%Sn( Pb )
Eutectic Microsconstituent ((Pb)+bSn)
%L61.8%Sn
30%Sn( alloy ) 18.3%Sn( Pb )
30%
61.8%Sn( liq.) 18.3%Sn( Pb )
Phases in Eutectic Microsconstituent
%bin eut . 97.8%Sn
61.8%Sn( eut .) 18.3%Sn( Pb )
55%
97.8%Sn( liq.) 18.3%Sn( Pb )
%Pbin eut . 18.3%Sn
97.8%Sn( liq.) 61.8%Sn( eut .)
45%
97.8%Sn( liq.) 18.3%Sn( Pb )
A ssessed F e- C p h ase d i ag r am .
Fe-Fe3C Phase Diagram
Austenite
Cementite
Ferrite
Cast Irons
Hypoeutectoid
Hypereutectoid
Steels
Pearlite (ferrite + cementite)
%C = 0.77%
Time-Temperature-Transformation
(TTT) Diagram
Decomposition of Austenite at fixed
temperature
800°C
fs
727°C
ps
Temperature
200°C
100°C
Pearlite: High Temp
slow nucleation
pf
bs
bf
ms
mf
Log Time
Key
Main symbol
f = ferrite
p = pearlite
b = bainite
c = cementite
(Fe3C)
Subscripts
s = start
f = finish
Coarse pearlite
Fine pearlite
Bainite:
Diffusion slow
for pearlite
Martensite
athermal (diffusionless)
Quench / Hardenability / Tempering
• Quench - rapidly cool
– in steel: cool fast enough to Ms to prevent pearlite / bainite
formation
• Hardenability
– ease of forming martensite in steels
– alloying elements inhibit pearlite / bainite formation, promote
martensite formation
• Tempering of steels
– reheating martensite to form transition carbides
– improve toughness
Cold Working
• Plastic deformation creates dislocations, which
increases strength / decreases ductility
• Reduction in Area used to quantify degree of cold
working
Ai Af
%CW %RA
100%
Ai
w i l i w f lf
100%
wi li
for w f w i
l l
%RA i f 100%
li
%RA
d2
i
%RA
4
d
4
d2
f
4
2
i
100%
d2 d2
i
f
d
2
i
100%
Cold Worked Properties
600
16
14
500
12
Stress (MPa)
Yield Strength
Tensile Strength
Percent Elongation
10
300
8
6
200
4
100
2
0
0
0
10
20
30
40
50
Percent Cold Work
60
70
80
Percent Elongation
400
Balancing Strength / Ductility
600
35
30
500
Stress (MPa)
Yield Strength
Tensile Strength
20
Percent Elongation
300
15
200
Sy > 310 MPa
requires
%CW > 22%
10
100
5
0
0
0
10
20
30
40
50
Percent Cold Work
Both Properties
requires
22% < %CW < 31%
60
70
80
Percent Elongation
25
400
Elongation > 10%
requires
%CW < 31%
Balancing Strength / Toughness
600
50
Yield Strength
Tensile Strength
550
45
Fracture Toughness
500
40
Stress (MPa)
450
35
y = 250 MPa
13% CW
400
30
350
25
KIc = 16 MPa m0.5
39% CW
300
20
250
Example
for 31% CW
Sy = 364 MPa
Kic = 22 Mpa m½
15
31% CW
Sy = 364 MPa
KIc = 22 MPa m0.5
200
150
10
5
100
0
0
10
20
30
40
Percent Cold Work
50
60
70
Fracture Toughness (K Ic) (MPa m 0.5)
Sy > 250 MPa
and
Kic > 16 Mpa m½
requires
13% < %CW < 39%
Cold Work / Anneal / Hot Work
• Annealing can eliminate effect of cold work
– recovery - stress relief, little change in properties
– recrystallization - elimination of dislocations, decrease in
strength, increase in ductility
– grain growth - increase in grain size, decreases both
strength and ductility
• Hot working
– deforming at high enough temperature for immediate
recrystallization
– list cold-working and annealing at the same time
– no increase in strength
– used for large deformation
– poor surface finish - oxidation
– After hot working, cold working used to increase strength
and improve surface finish
Organization from 1996-7 Review Manual
(same topics in 2004 review manual)
• Crystallography
• Materials Testing
• Metallurgy
Crystallography
• Crystal structure
– atoms/unit cell
– packing factor
– coordination number
• Atomic bonding
• Radioactive decay
Bravais Lattice
Crystal System
Centering
(x,y,z): Fractional coordinates proportion of axis length, not
absolute distanct
P: Primitive: (x,y,z)
I: Body-centered: (x,y,z); (x+½,y+½,z+½)
c
C: Base-centered: (x,y,z); (x+½,y+½,z)
b
a
a
b
F: Face-centered: (x,y,z); (x+½,y+½,z)
(x+½,y,z+½); (x,y+½,z+½)
Centering must apply to all atoms
in unit cell.
Bravais Lattices (14)
Crystal
System
Cubic
Tetragonal
Orthorhombic
Rhombohedral
Hexagonal
Monoclinic
Triclinic
Parameters
abc
ab
abc
ab
abc
ab
abc
ab
abc
ab
abc
a b
abc
ab
Primitive
Body(Simple) Centered
X
X
X
X
X
X
FaceCentered
BaseCentered
X
X
X
X
X
X
X
X
Atoms Per Unit Cell
• Corners - shared by eight unit
cells (x 1/8)
– (0,0,0)=(1,0,0)=(0,1,0)=(0,0,1)=(1,1,0)
=(1,0,1)=(0,1,1)=(1,1,1)
• Edges - shared by four unit cells
(x 1/4)
– (0,0,½)= (1,0,½)= (0,1,½)= (1,1,½)
• Faces - shared by two unit cells (x
1/2)
– (½,½,0)= (½,½,1)
Common Metal Structures
• Face-Centered Cubic (FCC)
– 8 corners x 1/8 + 6 faces x 1/2
– 1 + 3 = 4 atoms/u.c.
• Body-Centered Cubic (BCC)
– 8 corners x 1/8 + 1 center
– 1 + 1 = 2 atoms/u.c.
• Hexagonal Close-Packed (HCP)
– 8 corners x 1/8 + 1 middle
– 1 + 1 = 2 atoms/u.c.
– 12 hex. Corner x 1/6 +2 face x 1/2 + 3
middle = 6 atoms/u.c.
Packing Factor
P.F .
• Fraction of space occupied by atoms
• For FCC
r
a
• For BCC
P .F .
4
34 r 3 4 34 r 3
a
3
4
2
3
3 2
P .F .
34 r 3 2 34 r 3
a
3
4
3
3
3
8
4
r
2
0.74
body diagonal a2 a2 a2 4 r a
2
ri 3
a b c
face diagonal a 2 a 2 4 r a
a
4
3
4
r
3
0.68
Density
atom mass
mass
u.c. mole
Density
atom volume volume
mole u.c.
For nickel:
- Atomic weight = 58.71 g/mole
- Lattice parameter = 3.5239 Å=3.5239 x 10-8 cm
- Avogadro’s No. = 6.02 x 1023 = 0.602 x 1024 = atoms/mole
g
4 atom
58
.
71
g
u
.
c
.
mole
Density
8.915
3
3
24 atom
8
cm
0.602 x 10
3.5239 x 10 cm
mole
Close Packed (CN=12)
Highest packing density for same sized spheres
FCC and HCP structures
Cube Center (CN=8)
Same atoms: BCC
Different atoms: CsCl
Octahedral Site (CN=6)
In FCC:
- Center (½,½,½)
- Edges (0,0,½),(0,½,0),(½,0,0)
- 4 per unit cell
- All filled - NaCl structure
8-sided shape
Tetrahedral Site (CN=4)
In FCC:
- Divide cell into 8 boxes - center of small box
- (¼,¼,¼),(¾,¼,¼),(¼,¾,¼),(¾,¾,¼)
(¼,¼, ¾)(¾,¼, ¾),(¼,¾, ¾)(¾,¾, ¾)
-8 per unit cell
-All filled - CaF2 structure; half-filled - ZnS
4-sided shape
Radius Ratio Rules
Critical radius is size of atom which just fits in site
Define minimum for bonding (i.e. atoms must touch to bond)
CN 8
Critical Radius for CN 8 = 0.732
CN 6
Critical Radius for CN 6 = 0.414
CN 4
Critical Radius for CN 4 = 0.225
CN 3
planar
Close Packed Plane
A
A
B
HCP: ABABABABABABABAB
FCC: ABCABCABCABCABC
Same packing density (0.74)
Same coordination (CN=12)
A
B
C
Miller Indices
Planes
Directions
(hkl)
specific
{hkl}
family
[hkl]
specific
<hkl>
family
- No commas
- No fractions
- Negative
indicated by bar
over number
A family of planes includes all planes which are
equivalent by symmetry - depends on crystal system.
- For cubic: (110),(011) and (101) are all {110}
- For tetragonal: (011) and (101) are {101}
but (110) is not (ca)
Miller Indices - Directions
c
a
-1
b
x
y
z
1/2 -1 -1/3 (x 6)
3 6 2
-1/3
1/2
1
1/4
1/2
x
1
y
1/4
z
1/2 (x 4)
4 1 2
Miller Indices - Planes
c
c
1
a
b
1
4
b
a
1
4
1
2
x
y
intercept 1/4
reciprocal 4 0
z
-1/2
-2
4 0 2
2
Miller Indices - Planes
c
c
1
a
b
3
a
1
b
1
3
1
4
1
4
1
2
x
y
z
intercept 1/4 -1/3 -1/2
reciprocal 4 -3
-2
4 3 2
2
Atomic Bonding
• Covalent
– sharing electrons
– strong
– directional
• Ionic
– trading of electrons
– electrostatic attraction or
ions
– strong
– non-directional
• Metallic
– metal ions in sea or
electrons
– moderately strong
– non-directional
• Secondary
– Van der Waals
– H-bonding
– electrostatic attraction of
electric dipole (local
charge distribution
– weak
Radioactive Decay
• Loss of electrons/protons/neutrons
– alpha - 2 protons / two neutrons (i.e He nucleus)
– beta - electrons
– gamma - energy
time
t
• Exponential decay
N No exp
time constant
t1
t1
1 No
ln2 2
N 21 No No exp 2 ln 2
No
ln2
amount
t ln2
0.693 t
N No exp
No exp
t1
t1
2
2
original amount
half life