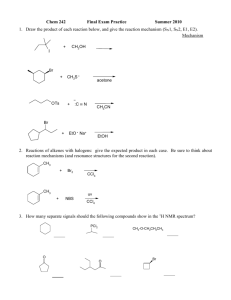
Total Synthesis of Prostagladines
advertisement

SINTESIS • Prostaglandines (PGs) • Taxol • Palytoxin Total Synthesis of Prostaglandines (PGs) Strategy and method Department of Chemistry, Graduate School of Science, Kobe University Masahiko Hayashi Primary Prostaglandins O O O CO2H HO CO2H HO OH PGE1 HO OH PGE2 HO CO2H OH PGF1 OH PGE3 HO HO HO CO2H CO2H HO OH PGF2 CO2H HO OH PGF3 Total Synthesis of Prostaglandins (PGs) Corey’s method vs Noyori’s method Corey: 1990 Nobel Prize: linear synthesis Noyori: 2001 Nobel Prize: convergent synthesis Significance of the synthesis of Prostaglandins (PGs) 1. It is necessary to synthesize PGs, because the quantity in nature (in animals including human being) is too little for actual medical treatment. 2. Artificial modification is possible by synthesis. For example, the analogues of Prostacyclins (PGI2). Natural PGI2 is very unstable as a drug. bioactive = drug Structure of Prostaglandins 9 8 7 5 6 10 14 11 12 13 4 3 16 15 1 2 COOH side-chain 18 17 20 side-chain 19 prostanoic acid side-chain cyclopentane ring O O A B O HO O C COOH O D OH 1 HO COOH O O HO E OH HO F G (OOH at C-15) H 2 COOH COOH OH 3 O COOH O O HO OH PGI2 OH TXA2 Primary Prostaglandins O O O CO2H HO CO2H HO OH PGE1 HO OH PGE2 HO CO2H OH PGF1 OH PGE3 HO HO HO CO2H CO2H HO OH PGF2 CO2H HO OH PGF3 The Major Problems in PG Synthesis 1. Selective creation of the four and five stereogenic centers in PGE and PGF series, respectively. 2. Stereoselective placement of C=C bonds in the side chains . 3. Overcomingunstability of the -hydroxycyclopentanone structure of the PGE series. Corey's solution 1. Use of highly functionalized key interm ediate derived from cyclopentadiene via stereo-defined bicyclic interm ediates. 2. Introduction ofcis and trans double bonds by Wittig-type reaction . Retrosynthetic analysis O O CO2H CO2H RO OR RO OR linear convergent Corey’s method Noyori’s method A B C A 90% 90% 90% C 90% 90% 90% D 90% E 90% F G B 90% D E 90% 90% 90% H G 0.93 x 100 = 73% convergent synthesis C D F 90% G H 収束型 B E 90% F A H 直線型 0.97 x 100 = 48% linear synthesis Corey Method (17 steps + optical resolution) O HO O MeO CO2H Cl CN n-Am AcO HO O Corey lactone OH PGF2 Just-Upjohn Method (16 steps + optical resolution) O O Corey lactone O O CHO Noyori Method (Three Component Coupling) O R+ O CO2Me RO R RO OR PGs PGs Retrosynthetic Analysis of Corey’s Method (C6H5)3P O O CO2H CHO RO OR RO Wittig OR O O O O Wittig CHO RO RO OR CO2-M + Corey's Prostaglandin Synthesis (part 1) MeO NaH, T HF Cl MeOCH2 Cl T HF, -55 °C Cu(BF4 )2 0 °C 12 MeO 10 MCPBA, NaHCO3 11 O CN CN > 90% 1. NaOH, H2 O, 0 °C 6 11 O CH2 Cl2 O HO 6 KI3 , NaHCO3 9 8 10 2. CO2 11 > 95% O HO O 1. Ac2 O, py 90% OMe HO 2. (n-Bu)3 SnH AIBN, PhH 12 AcO 80% PO(OMe)2 O O 1. BBr3 , CH2 Cl2 , 0 °C 8 OMe H2 O, 0 °C OMe 12 O O 10 11 Cl O 8 80% I KOH, H2 O/DMSO 12 MeO 8 9 MeO 2. CrO3 .2py, CH2 Cl2 , 0 °C O H 13 AcO Corey lactone 99% O O O O 14 Zn(BH4 )2 , DME O 14 15 NaH, DME AcO H O 70% 15 AcO H 13 H OH > 97% of a 1:1 mixture of C-15 epimers Corey's Prostaglandin Synthesis (part 2) O O O O 1. K2CO 3, MeOH 14 2. DHP, TsOH, CH2 Cl 2 15 H AcO 13 H OH 8 THPO 6 5 CO2H 13 H OTHP DIBAL-H toluene, -60°C 5 Ph3 P O CO2 6 DMSO H OTHP 1. H2Cr 2O7 , PhH/H2O 2. AcOH, H2O, 37 °C AcOH, H2O, 37 °C HO 15 OH H H H THPO O 9 14 12 DHP = HO 6 THPO O H H H OTHP H CO2H CO2H HO H H OH (+)-PGF 2 HO H H OH (+)-PGE 2 Corey's Prostaglandin Synthesis (part 1) MeO NaH, T HF Cl MeOCH2 Cl T HF, -55 °C Cu(BF4 )2 0 °C 12 MeO 10 MCPBA, NaHCO3 11 O CN CN > 90% 1. NaOH, H2 O, 0 °C 6 11 O CH2 Cl2 O HO 6 KI3 , NaHCO3 9 8 10 2. CO2 11 > 95% O HO O 1. Ac2 O, py 90% OMe HO 2. (n-Bu)3 SnH AIBN, PhH 12 AcO 80% PO(OMe)2 O O 1. BBr3 , CH2 Cl2 , 0 °C 8 OMe H2 O, 0 °C OMe 12 O O 10 11 Cl O 8 80% I KOH, H2 O/DMSO 12 MeO 8 9 MeO 2. CrO3 .2py, CH2 Cl2 , 0 °C O H 13 AcO Corey lactone 99% O O O O 14 Zn(BH4 )2 , DME O 14 15 NaH, DME AcO H O 70% 15 AcO H 13 H OH > 97% of a 1:1 mixture of C-15 epimers Corey's Prostaglandin Synthesis (part 1) MeO NaH, T HF Cl MeOCH2 Cl T HF, -55 °C Cu(BF4 )2 0 °C 12 MeO 10 MCPBA, NaHCO3 11 O CN CN > 90% 1. NaOH, H2 O, 0 °C 6 11 O CH2 Cl2 O HO 6 KI3 , NaHCO3 9 8 10 2. CO2 11 > 95% O HO O 1. Ac2 O, py 90% OMe HO 2. (n-Bu)3 SnH AIBN, PhH 12 AcO 80% PO(OMe)2 O O 1. BBr3 , CH2 Cl2 , 0 °C 8 OMe H2 O, 0 °C OMe 12 O O 10 11 Cl O 8 80% I KOH, H2 O/DMSO 12 MeO 8 9 MeO 2. CrO3 .2py, CH2 Cl2 , 0 °C O H 13 AcO Corey lactone 99% O O O O 14 Zn(BH4 )2 , DME O 14 15 NaH, DME AcO H O 70% 15 AcO H 13 H OH > 97% of a 1:1 mixture of C-15 epimers Corey's Prostaglandin Synthesis (part 1) MeO NaH, T HF Cl MeOCH2 Cl T HF, -55 °C Cu(BF4 )2 0 °C 12 MeO 10 MCPBA, NaHCO3 11 O CN CN > 90% 1. NaOH, H2 O, 0 °C 6 11 O CH2 Cl2 O HO 6 KI3 , NaHCO3 9 8 10 2. CO2 11 > 95% O HO O 1. Ac2 O, py 90% OMe HO 2. (n-Bu)3 SnH AIBN, PhH 12 AcO 80% PO(OMe)2 O O 1. BBr3 , CH2 Cl2 , 0 °C 8 OMe H2 O, 0 °C OMe 12 O O 10 11 Cl O 8 80% I KOH, H2 O/DMSO 12 MeO 8 9 MeO 2. CrO3 .2py, CH2 Cl2 , 0 °C O H 13 AcO Corey lactone 99% O O O O 14 Zn(BH4 )2 , DME O 14 15 NaH, DME AcO H O 70% 15 AcO H 13 H OH > 97% of a 1:1 mixture of C-15 epimers Corey's Prostaglandin Synthesis (part 1) MeO NaH, T HF Cl MeOCH2 Cl T HF, -55 °C Cu(BF4 )2 0 °C 12 MeO 10 MCPBA, NaHCO3 11 O CN CN > 90% 1. NaOH, H2 O, 0 °C 6 11 O CH2 Cl2 O HO 6 KI3 , NaHCO3 9 8 10 2. CO2 11 > 95% O HO O 1. Ac2 O, py 90% OMe HO 2. (n-Bu)3 SnH AIBN, PhH 12 AcO 80% PO(OMe)2 O O 1. BBr3 , CH2 Cl2 , 0 °C 8 OMe H2 O, 0 °C OMe 12 O O 10 11 Cl O 8 80% I KOH, H2 O/DMSO 12 MeO 8 9 MeO 2. CrO3 .2py, CH2 Cl2 , 0 °C O H 13 AcO Corey lactone 99% O O O O 14 Zn(BH4 )2 , DME O 14 15 NaH, DME AcO H O 70% 15 AcO H 13 H OH > 97% of a 1:1 mixture of C-15 epimers Corey's Prostaglandin Synthesis (part 1) MeO NaH, T HF Cl MeOCH2 Cl T HF, -55 °C Cu(BF4 )2 0 °C 12 MeO 10 MCPBA, NaHCO3 11 O CN CN > 90% 1. NaOH, H2 O, 0 °C 6 11 O CH2 Cl2 O HO 6 KI3 , NaHCO3 9 8 10 2. CO2 11 > 95% O HO O 1. Ac2 O, py 90% OMe HO 2. (n-Bu)3 SnH AIBN, PhH 12 AcO 80% PO(OMe)2 O O 1. BBr3 , CH2 Cl2 , 0 °C 8 OMe H2 O, 0 °C OMe 12 O O 10 11 Cl O 8 80% I KOH, H2 O/DMSO 12 MeO 8 9 MeO 2. CrO3 .2py, CH2 Cl2 , 0 °C O H 13 AcO Corey lactone 99% O O O O 14 Zn(BH4 )2 , DME O 14 15 NaH, DME AcO H O 70% 15 AcO H 13 H OH > 97% of a 1:1 mixture of C-15 epimers Corey's Prostaglandin Synthesis (part 1) MeO NaH, T HF Cl MeOCH2 Cl T HF, -55 °C Cu(BF4 )2 0 °C 12 MeO 10 MCPBA, NaHCO3 11 O CN CN > 90% 1. NaOH, H2 O, 0 °C 6 11 O CH2 Cl2 O HO 6 KI3 , NaHCO3 9 8 10 2. CO2 11 > 95% O HO O 1. Ac2 O, py 90% OMe HO 2. (n-Bu)3 SnH AIBN, PhH 12 AcO 80% PO(OMe)2 O O 1. BBr3 , CH2 Cl2 , 0 °C 8 OMe H2 O, 0 °C OMe 12 O O 10 11 Cl O 8 80% I KOH, H2 O/DMSO 12 MeO 8 9 MeO 2. CrO3 .2py, CH2 Cl2 , 0 °C O H 13 AcO Corey lactone 99% O O O O 14 Zn(BH4 )2 , DME O 14 15 NaH, DME AcO H O 70% 15 AcO H 13 H OH > 97% of a 1:1 mixture of C-15 epimers Do not confuse relative configuration and absolute configuration! So far, control of relative configuration (racemic compound) was done, so optical resolution is necessary to obtain chiral compound! Optical Resolution of Hydroxy Acid CO2H CO2H (+)-ephedrine H Ph H + OMe OH 1. PhH, NHMe 2. recrystallization OMe Me HO HO []D23 +37.2° (c 1.0, MeOH) O O KI3 , H2O, 0 °C O CO2H I OMe HO []D26 -45.8° (c 1.0, MeOH) OH OH PGE2 synthetic sample: []D -61° (c 1.0, THF) natural product: []D -61° (c 1.0, THF) Corey et. al, J. Am. Chem Soc., 1970, 92, 397 Corey's Prostaglandin Synthesis (part 1) MeO NaH, T HF Cl MeOCH2 Cl T HF, -55 °C Cu(BF4 )2 0 °C 12 MeO 10 MCPBA, NaHCO3 11 O CN CN > 90% 1. NaOH, H2 O, 0 °C 6 11 O CH2 Cl2 O HO 6 KI3 , NaHCO3 9 8 10 2. CO2 11 > 95% O HO O 1. Ac2 O, py 90% OMe HO 2. (n-Bu)3 SnH AIBN, PhH 12 AcO 80% PO(OMe)2 O O 1. BBr3 , CH2 Cl2 , 0 °C 8 OMe H2 O, 0 °C OMe 12 O O 10 11 Cl O 8 80% I KOH, H2 O/DMSO 12 MeO 8 9 MeO 2. CrO3 .2py, CH2 Cl2 , 0 °C O H 13 AcO Corey lactone 99% O O O O 14 Zn(BH4 )2 , DME O 14 15 NaH, DME AcO H O 70% 15 AcO H 13 H OH > 97% of a 1:1 mixture of C-15 epimers Corey's Prostaglandin Synthesis (part 1) MeO NaH, T HF Cl MeOCH2 Cl T HF, -55 °C Cu(BF4 )2 0 °C 12 MeO 10 MCPBA, NaHCO3 11 O CN CN > 90% 1. NaOH, H2 O, 0 °C 6 11 O CH2 Cl2 O HO 6 KI3 , NaHCO3 9 8 10 2. CO2 11 > 95% O HO O 1. Ac2 O, py 90% OMe HO 2. (n-Bu)3 SnH AIBN, PhH 12 AcO 80% PO(OMe)2 O O 1. BBr3 , CH2 Cl2 , 0 °C 8 OMe H2 O, 0 °C OMe 12 O O 10 11 Cl O 8 80% I KOH, H2 O/DMSO 12 MeO 8 9 MeO 2. CrO3 .2py, CH2 Cl2 , 0 °C O H 13 AcO Corey lactone 99% O O O O 14 Zn(BH4 )2 , DME O 14 15 NaH, DME AcO H O 70% 15 AcO H 13 H OH > 97% of a 1:1 mixture of C-15 epimers OH O ring-chain tautomeric equilibrium 6 9 O HO 6 9 15 15 THPO H H OTHP lactol form THPO H H OTHP hydroxy aldehyde form stable ylide = unreactive unstable ylide = reactive A B C A 90% 90% 90% C 90% 90% 90% D 90% E 90% F G B 90% D E 90% 90% 90% H G 0.93 x 100 = 73% conversent synthesis C D F 90% G H 収束型 B E 90% F A H 直線型 0.97 x 100 = 48% linear synthesis Noyori’ s method Three component coupling Michael- O エノラート捕捉 CO2CH3 RO O CO2CH3 Michael addition + Enolate trap RO OR OR PGE2 1) Li O RO O OR CuI, P(n-C4H9)3 CO2CH3 2) (C6H5)3 SnCl, HMPA 3) I CO2CH3 RO OR Conjugate Addition of Organocopper Reagents RLi + CuI + x P(n-C4H9)3 Li[RCuI(P(n-C4H9)3)y] + (x-y) P(n-C4H9)3 RCu(P(n-C4H9)3)x + (x-z) P(n-C4H9)3 + LiI O O H2O + "RCu" R R = sp 2- or sp3-carbon moiety Vicinal carba-condensation of -unsaturated ketones O R R O R R 1. (C6H5) 3SnCl HMPA 2. RX O R RLi + CuI + (n-C4H9)3P O X CH2=CHX R M O RCHO R OH R R R R RCH(OR') 2 (CH3 )3SiOTf RC(OR') 3 BF3 O O RO' OR' R R R OR' R R R An Organometallic Way to Prostaglandins: The Three Component Couplong Synthesis O O MO RM RO R RX RO R R RO Organocuprate/organotin procedure (1): O O 1. R Li + CuI + ( n-C4H9) 3P CO2CH3 2. (C6H5) 3SnCl, HMPA R3SiO 3. RI RLi = Li R3SiO RI = I OSiR3 SiR 3 = Si(CH 3)2-t-C4H9 OSiR3 CO2CH3 An Organometallic Way to Prostaglandins: The Three Component Couplong Synthesis Organolithium/organozinc procedure: O O 1. R Li + Zn(CH 3)2 CO2CH3 2. RI, HMPA R3SiO R3SiO RLi = Li RI = I OSiR3 SiR 3 = Si(CH 3)2-t-C4H9 OSiR3 CO2CH3 Stork法 LiO O + LiCu 2 HCHO C5H11 C5H11 OR' RO O RO OR' O 1) MsCl pyridine OH C5H11 RO O OR' OR" C5H11 2) i-Pr2NEt RO OR' RO LiCu 2 OR' 3 成分連結を 狙っ たが, α鎖で直接, エ ノ ラ ート が OR" 捕捉でき なかっ た。 そこ で、 ま ず反応性の高求電子 剤である ホルムア ルデヒ ド でいっ たん捕捉し , それ を 延ばし ていく 作戦に妥協。 それを 解決し たのがエ ノ ラ ート の金属交換( 野依法) 。 Synthesis of Prostacyclin (PGI2) O HO CO2CH3 L-selectride R3SiO CO2CH3 R3SiO OSiR3 CO2CH3 PdCl OSiR3 CO2CH3 HCOO -NH 4+ deprotection O O PdCl2 (C6 H5C N)2 PGI2 platelet aggregation inhibitor R3SiO OSiR3 SiR 3 = Si(CH 3)2-t-C4H9 R3SiO OSiR3 Synthesis of Isocarbacyclin Methyl Ester OR' C6H5(CH2) 2Si O CO2CH3 CO2CH3 R3SiO R3SiO OSiR3 CO2CH3 SiR 3 = Si(CH 3)2-t-C4H9 CO2CH3 1. HClO 4 2. CF3CO 2H C6H5(CH2) 2Si R3SiO OSiR3 OSiR3 HO OH isocarbacyclin methyl ester radical cyclization One-pot General Synthesis of Prostaglandins : Three-Component Coupling Synthesis HO O CO2H O HO OH PGD1 O CO2H chain chain RO HO CO2H HO OH PGE1 OH PGF1 O HO CO2Me RO CO2H O OR' OH PGD2 COOH HO O CO2H CO2H O HO HO OH PGI2 OH PGF2 HO OH PGE2 O O O NH O O OH B O A OH H OH O Taxol C D O O Synthetic Strategy for Taxol R. A. Holton (1994) O O OH TESO TESO OBOM taxol (-)-camphor O TBSO OH TBSO H K. C. Nicolaou (1994) O OBn OTBS O OH OBn HO OBn O TBDPSO + NNHSO2 R TBSO O S. J. Danishefsky (1995) H O O O O H O O OTf OTMS H O O OTBS OTBS O H OBn O + I taxol O H O OBn chiral O O O O OH -pinene O O P. A. Wender (1997) O taxol O O O optical resolution O OMe OTBS MeO NC OTMS H O O O AcO O TIPSO TIPSO OTBS H O O OBn O H O OTBS HO O OTroc H OBn OBOM taxol D,H,W N AcO O H,W Bz N OR HO Ph H D BzO OAc O 18 OTES R=H 10 O O 13 A 1 BzHN OH HO O Ph OH 9 B 15 C all HO O 11 B A AcO 2 H; Holton N; Nicolaou D; Danishefsky W; Wender C 4 H BzO H Baccatin III (R = H) 7 3 D,N W 8 Taxol 5 D OAc 20 O Retrosynthesis of Taxol by Mukaiyama (1997) OAc BzHN B O OH OH OBn O R TBSO O Ph O A C D O H HO BzO B OAc PMBO OBn Taxol BnO OBn OBn OTBS OTBS OHC OPMB O O TBS OPMB Ciguatoxin H Hirama (2001, 11) H HO OH OH H O H HO H O H OH H H O H HO OH H OH H O H HO H OH H O O OH OH H H Ring-closing metathesis (RCM) Grubbs (1992-) RCM PCy3 Cl Ph Ru Cl PCy3 Grubb's catalyst Organic Synthesis in the 21 century 1. Truly effficient production of known valuable compounds 2. Creation of new valuable compounds and materials Coupling Reactions of Kishi’s Palytoxin Synthesis C7-C8: NiCl2/CrCl2 (NHK) coupling C22-C23: Wittig reaction & Hydrogenation C37-C38 : Wittig reaction & Hydrogenation C51-C52: Horner-Wadsworth-Emmons reaction C75-C76: Suzuki coupling C84-C85 : NiCl2/CrCl2 (NHK) coupling C98-C99: Wittig reaction Structure of Palytoxin (1981) Uemura, D.; Ueda, K.; Hirata, Y.; Naoki, H.; Iwashita, T. Tetrahedron Lett., 1981, 22, 2781. Moore, R. E.; Bartolini, G. J. Am. Chem. Soc., 1981, 103, 2491. Coupling Reactions of Kishi’s Palytoxin Synthesis C7-C8: NiCl2/CrCl2 (NHK) coupling C22-C23: Wittig reaction & Hydrogenation C37-C38 : Wittig reaction & Hydrogenation C51-C52: Horner-Wadsworth-Emmons reaction C75-C76: Suzuki coupling C84-C85 : NiCl2/CrCl2 (NHK) coupling C98-C99: Wittig reaction Nozaki-Hiyama-Kishi Reaction Swern Oxidation Suzuki Cross-Coupling (Suzuki-Miyaura Cross-Coupling) Total Synthesis of Palytoxin by Kishi group C51をアルデヒドに変換