Jason Li
advertisement

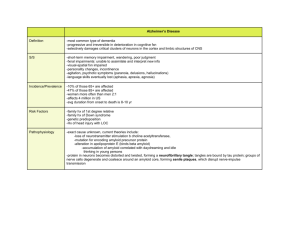
Jason Li 2015 COSMOS Cluster 1: Biotechnology Alzheimer’s Disease (AD) Neurodegenerative disease It is not a normal phenomenon Most common form of dementia Leads to impairment of memory and cognition Causes loss of connections among and death of neuron cells. Causes of Alzheimer’s-Tau Protein Hyper-phosphorylation of tau protein inside neuron cells Normal tau proteins stabilizes microtubules When hyper-phosphorylated, Tau proteins start clumping together and forming tangles instead of doing their normal job. Blocks transportation of essential supplies inside the cell Causes of Alzheimer’s-Amyloid Plaques They are also known as beta-amyloids (Aβ) Located in synapse of neuron cells Misfolded proteins that tend to clump together. Insoluble Occupy synapses and lead to death of neuron cells. Tau Protein or Plaques? Which one causes Alzheimer’s more? There is no certain answer Both contribute and lead to eventually death of neuron cells. Nattokinase Extracellular enzyme secreted by bacterium Bacillus subtilis natto found in the food natto Natto: fermented soybeans that are more commonly eaten in Asia, especially Japan. Despite its name, it is a protease. A previous research has shown that Nattokinase can cleave samples of beta-amyloid but not from the brain Another research has shown that Nattokinase can be taken orally by rats and enter their brain tissues. Researchable Question What effects would nattokinase have on the level of beta amyloids and the overall gene expression in a mouse’s brain tissues with Alzheimer’s? Significance of Experiment Further progress in alleviating the disease Prove the potential of food treatment Natto would be a convenient food to eat Because there is no clear understanding of the relationship between amyloid plaques and tau protein, if amyloid can be reduced with nattokinase, then effects of reducing the plaques on the disease in general can be observed Breakdown of Experiment 1. 2. feeding of nattokinase 3. extract brain tissue 4.thioflavin t binding assay to detect beta amyloid 5. mRNA sequencing to test gene expression Thioflavin T Binding AssayBackground Info Dye-binding assay to diagnose amyloid fibrils ex vivo or in vitro Thioflavin T: a benzothiazole dye The dye exhibits enhanced fluorescence when binding to amyloid fibrils RNA-Seq Background Info Detects gene expression of mRNA in the whole brain Converting mRNA into cDNA libraries for further analysis through sequencing. Advantages over other tests like microarrays and hybridization: faster and cheaper, more data output. Sequencing only the exons in RNA transcripts, the RNA responsible for producing protein products. Groups of Mice Group 1: Negative control group: healthy mice Group 2: Positive control group 2: AD mice fed with water only Group 3: AD mice treated with low dose of nattokinase Group 4: AD mice treated with high dose of nattokinase Hypothesis In the brain tissue, addition of nattokinase reduces amyloid plaques and influences some gene expressions in the brain. Predicted level of amyloid plaques among the groups: Group 2 (positive control with untreated AD) > Group 3 (treated with low dose of nattokinase) > Group 4 (treated with high dose of nattokinase) > Group 1 (negative control without AD). Step 1: Oral Administration of Nattokinase Obtain mouse models Purchase nattokinase from a dietary supplement market in USA. Feed the experimental group with nattokinase suspended in water orally for 45 days daily Step 2: Purification of βA Animals fasted overnight Brain extraction Purification of protein: Homogenizing brain tissue Ultracentrifuging or chromatography 3. Thioflavin T Binding Assay Get isolated sample of Aβ Incubate sample with dye added Screen for fluorescence using screening machine Step 4. RNA- Seq Necessary materials can be purchased from Illumina, a prominent biotechnology company Isolating all RNA: centrifuging and diluting lipids and proteins Purifying mRNA (there is a lot more rRNA than mRNA) Synthesize cDNA using reverse transcriptase Fragmentation of cDNA Addition of adapters to fragments for primers to bind in sequencing process Sequencing of cDNA fragments Analysis of sequence 5. Analysis of mRNA sequence Assemble expressed gene sequences using overlapping sequences of fragments Identifying all expressed genes using Blast Comparison of treated and untreated Challenges of the Experiment Keep in mind: the brain is complex Thioflavin T assay: might be biased by the presence of exogenous factors. RNA-Seq: difficulty of interpreting the data Further Areas of Research Switching from experiments for mice to humans The exact mechanism by which amyloid plaques and tau protein cause neuronal damage Gene expression of abnormal proteins in AD Gene therapy might be possible to be developed Bibliography "Alzheimer's Disease & Dementia | Alzheimer's Association." Alzheimer's Disease & Dementia | Alzheimer's Association. Alzheimer's Association, n.d. Web. 28 July 2015. "Tau Protein." Wikipedia. Wikimedia Foundation, n.d. Web. 29 July 2015. Murphy, M. Paul, and Harry LeVine. "Alzheimer’s Disease and the β-Amyloid Peptide." Journal of Alzheimer's Disease : JAD. U.S. National Library of Medicine, n.d. Web. 29 July 2015. "Amyloid-Degrading Ability of Nattokinase from Bacillus Subtilis Natto."Journal of Agricultural and Food Chemistry. National Taiwan University, n.d. Web. 29 July 2015. "Serrapeptase and Nattokinase Intervention for Relieving Alzheimer’s Disease Pathophysiology in Rat Model." Human and Experimental Toxicology. N.p., n.d. Web. July 2013. Jameson, LaramieP., Nicholas W. Smith, and Sergei V. Dzyuba. "Dye-Binding Assays for Evaluation of the Effects of Small Molecule Inhibitors on Amyloid (Aβ) Self-Assembly." ACS Chemical Neuroscience. American Chemical Society, n.d. Web. 29 July 2015. Jameson, LaramieP., Nicholas W. Smith, and Sergei V. Dzyuba. "Dye-Binding Assays for Evaluation of the Effects of Small Molecule Inhibitors on Amyloid (Aβ) Self-Assembly." ACS Chemical Neuroscience. American Chemical Society, n.d. Web. 30 July 2015. Walsh, Dominic M., Eva Thulin, Aedín M. Minogue, Niklas Gustavsson, Eric Pang, David B. Teplow, and Sara Linse. "A Facile Method for Expression and Purification of the Alzheimer’s Disease-associated Amyloid β-peptide." The Febs Journal. Blackwell Publishing Ltd, n.d. Web. 30 July 2015. Image Credits http://www.webmd.com/alzheimers/ss/slideshow-alzheimers overview http://flipper.diff.org/app/items/info/5017 http://www.uccs.edu/biology-educationalresources/alzheimers-disease.html http://www.oksfood.com/sushi/nattomaki_hosomaki.html https://en.wikipedia.org/wiki/Wood_mouse#/media/File:Apode mus_sylvaticus_bosmuis.jpg http://www.homeremedycentral.com/en/supplements/aminoacid/nattokinase.html http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3503347/ http://www.assay-protocol.com/biochemistry/proteinfibrils/amyloid-beta