Minerals - Perry Local Schools
advertisement

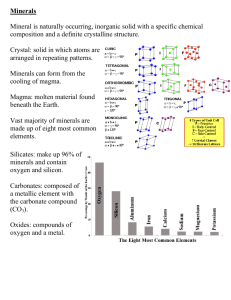
Name __________________________________________________________ Period _____ Minerals Article Ever wonder what a mineral is? Let’s find out! Minerals are the 'building blocks' of the Earth. We use minerals a lot. We even eat some! All minerals are made of small particles called atoms. A mineral is a naturally occurring, inorganic, solid which possesses a characteristic internal atomic structure and a definite chemical composition. If we take the definition of a mineral term by term, it becomes easier to understand. Here’s the break-down: Minerals must occur naturally; they cannot be made or manufactured. This means man-made substances such as steel would not be considered minerals. They result from geological processes. Minerals must be inorganic substances. This means that minerals are not living and are not formed by living things. Minerals must be crystalline solids. This means that minerals are not liquids, gases or plasmas but are solids. Being crystalline means the atoms in these solids have an orderly, repeated pattern called a crystal. While growing, minerals develop a distinct crystal form with smooth, flat planes called crystal faces. The geometric pattern of the crystal faces reflects the arrangement of the atoms of the crystal structure. Minerals "grow," or crystallize, from many types of solutions. They may precipitate from evaporating sea water, or crystallize from magmas when lava cools. The crystalline arrangement of the atoms in a mineral is one of the most important things about a mineral. Minerals have definite chemical compositions. This means that minerals are made up of atoms of specific elements. Substances that have all four of these features are minerals. Minerals have distinctive physical properties such as color, crystal form, cleavage, luster, streak, and hardness. In order to identify a mineral, you usually have to look at several physical properties to distinguish it from other minerals. What is Hardness? The hardness of a mineral is its ability to resist scratching. A mineral’s hardness is known as its “scratchability”. Hardness is dependent on the crystal structure. In 1812, Friedrich Mohs, a German mineralogist, developed a hardness scale that is still used today. Here is how the Mohs Hardness Scale works. The scale ranks the order of the hardness of minerals and some common objects from 1 to 10. The hardest mineral known, a diamond, was assigned the hardness number 10, and the softest mineral known, talc, was given the hardness number 1. Any mineral or object can scratch the surface of another mineral or object that has a lower hardness than itself. For example, your fingernail can scratch the minerals with a hardness of 2 or lower, like talc and gypsum. A copper penny can scratch calcite, gypsum, and talc. HARDNESS INDEX COMMON SCALE MINERAL OBJECTS 1 Talc 2 Gypsum 3 Calcite 4 Fluorite 5 Apatite 6 Orthoclase 7 Quartz 8 Topaz 9 Corundum 10 Diamond Fingernail Copper Penny Glass What is Luster? Luster refers to how light is reflected from the surface of a mineral. The two main types of luster are metallic and nonmetallic. Minerals with metallic luster look like metal, such as a silvery appearance or that of a flat piece of steel. Minerals that have nonmetallic luster do not look like metal but can range from being dull to glassy. There are many types of nonmetallic luster. Nonmetallic luster includes vitreous, resinous, pearly, silky, adamantine, greasy and dull/earthy. Vitreous is the most common type of non-metallic luster. Vitreous: Glassy luster. (quartz) Resinous: The luster of sap from a tree. (sulfur) Pearly: The luster of pearls. (talc) Silky: The luster of silk. (gypsum) Metallic Adamantine: Brilliant luster with a lot of shine. (diamond) luster Greasy: Looks like it is covered in a thin layer of oil. (turquoise) Nonmetallic luster Dull: Earthy appearance, not shiny at all. (bauxite) What is color? Minerals are colored because certain wave lengths of light are absorbed, and the color results from a combination of those wave lengths that reach the eye. The color of a mineral is the first thing most people notice, but it can also be the least useful in identifying a mineral. Most minerals occur in more than one color. Fluorite can be clear, white, yellow, blue, purple, or green. Defects and impurities can cause differences in color. Minerals that show a variation in color are called allochromatic. For some minerals, color is directly related to one of the major elements and can serve as a means of identification. Malichite is always green, azurite is always blue, and rhondonite is always red or pink. Most metallic minerals have a color that is constant, such as the brass-yellow of chalcopyrite and the copper-red of niccolite. These minerals may also tarnish which is espeically true of the mineral bornite. It is called the "peacock ore" because of the blue-velvet tarnish that develops on the surface. What is streak? A mineral’s streak is determined by rubbing it on a streak plate, which is a piece of unglazed porcelain. The streak of a mineral is the color of the powder left on a streak plate. The streak plate has a hardness of glass, so minerals with a Mohs Hardness greater than7 will scratch the streak plate and will not leave a powder on the streak plate. Streak can be useful for identifying metallic and earthy minerals. Minerals with a metallic luster have a dark streak. Nonmetallic minerals usually give a white streak because they are very light-colored. Other minerals may have very distinctive streaks. Hematite, for example, always gives a reddish brown streak no matter what type of luster it displays. What is cleavage? Cleavage is the ability of a mineral to break along preferred planes. Cleavages are clean, flat breaks. Certain planes of weakness exist in some minerals because of their particular atomic structure, or the crystals in their structure. Atomic bonds may be weaker in some directions than in others, so the mineral will tend to break, or cleave, in that direction. Minerals may have cleavage in only one direction, in only two directions, or in three or more directions. The cleavage angles at which these planes intersect may be distinctive. Minerals that have "perfect" cleavage almost always break in a preferred direction. Minerals that have "good" cleavage sometimes will break in a particular direction, and other times they may not. Types of Cleavage What is fracture? Not all minerals cleave easily. Some minerals fracture instead. Fractures can be smoothly curved, irregular, jagged or splintery. The most common types of fracture are conchoidal, splintery, hackly, uneven and crumbly. Conchoidal: Fracture that resembles a semicircular shell with a smooth, curved surface. Splintery – Fracture the makes long splinters or spikes form. Hackly – Fracture that leaves rough, jagged, points. Uneven - Fracture that leaves a rough or irregular surface. Crumbly - Fracture of minerals that crumble when broken. What is transparency? Transparency describes how well light passes through a mineral sample. There are three degrees of transparency: transparent, translucent, and opaque. You can see objects through a transparent mineral. You can see light, but no objects through a translucent mineral. You cannot see anything through an opaque mineral. What are minerals? Minerals are natural solid materials in the soil that were never alive. All of the land on our planet--and even the ocean floor--rests on a layer of rock made up of minerals. Minerals have also been found on other planets, on our moon, and in meteorites that landed on Earth. Some minerals, such as gold and silver, are made up entirely of one element. But most are formed from two or more elements joined together. The most common mineral is quartz, which is made of silicon and oxygen and is found all over the world. Sand is made up mostly of quartz. Graphite, which is used in pencils, is another common mineral. Other minerals, like diamonds, are very rare and valuable. Amazingly, diamonds and graphite are different forms of the same element--carbon. Minerals are the building blocks of rocks, but they are also found all around us. The chart below shows you where you can find various minerals in your own home or in buildings you visit often, like school! MINERALS HOUSEHOLD ITEM Copper, Zinc, Nickel, Chrome, Clay, Iron Plumbing Fixtures Stone, Brick, Iron Fireplace, Stove, Furnace Limestone, Clay, Shale, Gypsum, Aggregate Foundation, Driveway Trona, Silica, Feldspar Windows (Glass) Copper, Aluminum Wiring Coal Power Plant Electricity Copper, Zinc, Iron Door Fixtures Clay, Iron Sewer Pipes Clay, Stone, Aluminum Exterior Walls Gypsum Interior Walls Titanium, Mineral Fillers Paint Silicates, Slate Roof Iron, Zinc, Bauxite Drain, Gutters Iron, Zinc Nails, Screws Silica, Feldspar, Vermiculite Insulation Phosphate Fertilizer For The Lawn Lead Solder copper pipes and electical wiring Tin Solder copper pipes and electical wiring WHO SAYS WE DO NOT NEED MINERALS? In order to maintain how we live in the United States, here are some minerals used every day: 18,000,000 tons of raw material must be mined, cut or harvested to meet the demands of U.S. citizens (about 150 pounds for every man, woman and child); 640 acres (one square mile) of carpeting is woven using barite and calcium carbonate (limestone); 9,700,000 square feet of plate and window glass (about 223 acres) are used, enough to cover 200 football fields, using silica sand and trona; 2,750 acres of pavement are laid - four times as much surface area as is mined - enough concrete and asphalt to make a bicycle path 7 feet wide from coast to coast using sand, gravel, stone aggregate, and limestone; 94,000,000 eraser tipped pencils are purchased (enough erasers to correct all mistakes from 1,500 miles of notebook paper - about 129 acres of "goofs') using graphite, kaolin, pumice, copper and zinc; 426 bushels of paper clips (35,000,000) are purchased. Seven million are actually used, 8-9 million are lost and almost 5 million are twisted up by nervous fingers during telephone conversations, all using iron, clay, limestone, trona and zinc; 164 square miles of newsprint is used to print 62.5 million newspapers (enough to line a bird cage 12 miles wide and 13 miles long) using trona and kaolin; 400 acres of asphalt roofing are nailed down, utilizing silica, borate, limestone, trona, feldspar, talc, and silica sand; 187,000 tons of cement are mixed (enough to construct a four foot wide sidewalk from coast to coast) using limestone, sand, gravel, and crushed stone aggregate; 36,000,000 light bulbs are purchased, all made from tungsten, trona, silica sand, copper and aluminum; 10 tons of colored gravel is purchased for aquariums; 80 pounds of gold are used to fill 500,000 dental cavities; 50,000 pounds of toothpaste (2.5 million tubes) are used (enough to fill a small jet liner) requiring calcium carbonate, zeolites, trona, clays, silica and fluorite. 1,000,000 photographs are snapped (more than 29 acres of wallet sized photos) using silver and iodine.