AP Chemistry Syllabus 2011
advertisement

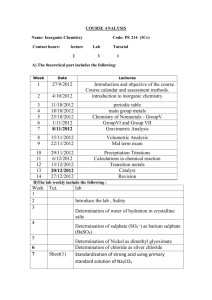
Malden High School Academic Year 2011-2012 Course Syllabus Name of Course: AP Chemistry Level: Advanced Placement Instructor: Martin Berryman Extra help and make-up are available: Course Number: 03461 mberryman@malden.mec.edu 7:00 – 7:30 A.M. (every day)) 2:15 – 3:15 P.M. (Tuesday) 2:15 – 2:45 P.M. (Wednesday, Thursday) Class Schedule: Malden High School’s schedule features seven 55 minute class periods with only six periods meeting each day. Students will be registered for a double period each day in their schedules. The second period will alternate between laboratory and AP Study Group with problem sets assigned for completion when lab does not meet. This will enable the students to complete the required experimentation as well as practice AP multiple choice and open response questions. Textbook: Zumdahl, Steven. Chemistry, 5th ed. Hall, James. Experimental Chemistry, 4th ed. Flinn Scientific AP Laboratory kits AP Laboratory protocols from NMSI Laboratory Sources: Demonstration Resources: Summerlin, Lee R. and James L. Ealy, Jr. Chemical Demonstrations: A Sourcebook for Teachers Shakhashiri, Bassam. Chemical Demonstrations: A Handbook for Teachers of Chemistry, Volumes I and II Online Homework Site: Class Notes: https://quest.cns.utexas.edu http://apchemistrynmsi.wikispaces.com/AP+Chemistry+Class+Lecture+Notes General Course Description This is a rigorous and challenging college-level course that thoroughly covers the following topics: Chemical calculations, matter and change, formulas and equations, states of matter, atomic structure and bonding, gases, chemical reactions, descriptive chemistry, nuclear chemistry, kinetics, equilibrium, acid-base, oxidation-reduction, electrochemistry, and thermodynamics. The course emphasizes laboratory work and analysis of experimental data. Students are expected… To keep a notebook/binder for a) class notes, b) text reading notes, c) problem samples, and d) daily questions To be equipped for class with a graphing calculator, carbonless laboratory notebook (100 page), the Princeton Review “Cracking the AP Chemistry Exam – 2012 Edition”, writing utensils and necessary reference tables. To be prepared for the beginning of each class which entails short questions and answer sessions based on previous class, reading assignments and problems. Using laboratory data students will produce laboratory reports which include procedure, observations, data, calculations, and analysis. To complete chapter and online problem sets to be collected and reviewed prior to exams. To complete at least four to six hours per week outside of school to accomplish course objectives. To work cooperatively and independently during problem solving and laboratory experiments. Assessment: Student grades will be based on online and written homework, laboratory reports, chapter quizzes and exams. Course Outline The course is organized on a quarterly basis rather than weekly. This affords increased flexibility in accommodating student abilities. QUARTER First UNITS Foundations Intro and Lab Safety Nomenclature Stoichiometry Solution Stoichiometry Electrochemistry EXPERIMENTS Determination of Mole Relationships in a Chemical reaction Determination of % Water in a Compound and Empirical Formula Determination of Concentration by OxidationReduction Titration of H2O2 Determination of an Electrochemical Series and Measurements Using Electrochemical Cells and Electroplating - QUARTER Second QUARTER Third QUARTER Fourth Additional Topics Covered UNITS General Equilibrium – LeChatelier Solubility Equilibrium Acid-Base theory and pH Buffers Kinetics: Instantaneous and Relative Rates (Introduction to Differential rate Law) UNITS Kinetics continued… Integrated Rate Law and Reaction Mechanisms Arrhenius Equation and Activation Energy Thermochemistry Thermodynamics (Entropy and Free Energy) Types of Reactions Atomic Structure and Bonding UNITS Gases (Kinetic theory of Matter and Gas Laws) Gas Stoichiometry States of Matter (Intermolecular Forces, Types of Solids, Phase Changes and Diagrams) Colligative Properties - - - EXPERIMENTS Colorimetric or Spectrophotometric Analysis and determination of Keq Standardization of NaOH and Acid-Base Titration Determination od appropriate Indicators and Buffers EXPERIMENTS Determination of the Rate of Reaction, its Order, and its Activation Energy Determination of Enthalpy change Associated with a Reaction and Hess’s Law EXPERIMENTS Determining the Molar Volume of a Gas Determination of Molar Mass by Vapor Density Determination of Molar Mass by Freezing Point Depression Nuclear Chemistry, though not taught as a separate unit, was included in the study of: atomic structure, rates and half-life, energy (nuclear fission and fusion) Organic Chemistry topics were infused throughout the instruction of acids and bases, nomenclature, structure, solubility, and reactions.