AIM - Mathematic.in
advertisement

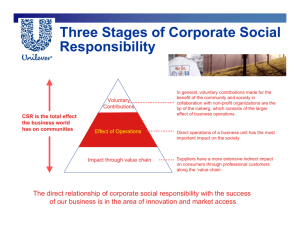
AIM TO CALCULATE THE ACIDITY OF DIFFERENT SAMPLES OF TEA LEAVES AND TO EXTIMATE THE AMOUNT OF GLYCOXIDE ALATED COMPOUNDS : e.g. CAFFIENE AND THROPHYLLINE PRESENTS IN EACH SAMPLES AND TO FIND THE RELATIONSHIP BETWEEN THEM. WHY I CHOOSE THIS PROJECT The fact that the morning cup of tea gives the feeling of well being performance being followed by decreased efficiency later during the day, puzzeles us. Naturally the question arises, “what is the reason behind it ?” There are many samples to tea leaves such as Assam Tea, Red lable Tea, Green lable, etc. To taste of tea samples differ from one another, but why ? Now answer to the two question written above is provided by two factors : i) Amount of Caffeine. ii) Acidity of different samples of tea. INTRODUCTION Tea is a beverage made by pouring boiling water over dried tea. Leaves . It has yellowish brown colour and slightly bitter taste. SCIENTIFIC CLASSIFICATION The tea plant is a member of the tea family Theaceae . Its scientifen came is Thea Sinensis. HISTORY According to legend, the use of thea was discovered by Emperor Sgen Nung of China above 2737 B.C. The custom of tea drinking gradually spread to Japan and other countries of the Orient. Tea was introduced to Europeans about 1600 by merchants who improved it from Far East. The beverage quickly won with popularity. FROM LEAF TO CUP The tea plant grows in tropical and subtropical climates. The plant, and evergereen quickly at low altitudes at low altitudes where the air is warm. The plant grows slowly in cool air, adding to this flavour. Tea plants have small, white , sweet smelling flowers. Each flower produces three seeds that look like hazel nuts. PROCESSING TEA There are three main kinds of tea : 1. Black, 2. Green and 3. Oolong . They differ in method used to process the tea leaves. BLACK TEA To make black tea, workers first spread the leaves on shelves called withering racks. Air is blown over the leaves to remove excess moisture leaving them soft and flexible. Next, the leves are crushed between the rollers of a machine to release the flavourful juices. Then, in a fermenting room the tea leaves change chemically under controlled humidity and temperature until they turn coppery in colour. Finally, the leaves are dried in oven and become brownish black. GREEN TEA Green Tea is made by steaming the leaves in large vats. The steaming prevents the leaves from changing the colour. The leaves are then crushed in a machine are dried in ovens. OOLONG TEA Oolong Tea is made by partially fermenting the leaves. This gives tea leaves a greenish brown colour. GRADES OF TEA Grades of Tea very accordingly to the size of leaves. The size of a tea leave has nothing to do with the quality of tea. To sort the process tea leaves by rfade, they are passed across screens with different size holes. The largest leaves, selected for packaging as loose tea, are classified in order of size as - Orange pekoe, pekoe and pekoe Souchong. The smaller or broken leaves are generally used in tea bags are classified as - Broken orange pekoe, Broken pekoe fannings and fannings. BREWING TEA Tea is brewed by pouring boiling water over one teaspoon of loose tea, or one tea bag per cup. To obtain the best flavour the tea should steep for 3-5 minutes before being served. CAFFIENE Chemical formula of caffeine ( C8 H10 N4 O2 H2O) Caffiene is an odourless, slightly bitter solid. It is a stimulant and is fould in small amount in coffee, tea and colas. Caffeine dissolves in water and alcohol and has crystals that look like needless. When taken in small amounts, caffeine increases the circulation and is considered harmless for most people. When taken in large amount, however, it caouse nervousness and lost of sleep. The use of caffeine also may cause headaches and digestive disturbances. Caffiene has been linked to the development of birth defects in laboratory animals and many physicians advise pregnant women to avoid excessive caffeine consumption. Caffiene was produced from plants in the pure form in 1820. It can now be madei nteh laboratory. Caffiene is used as stilulant of the heart and nervous system in certain is used as stilulant of the heart and nervous system in certain disorders. It is also a remedy fir poisoning by alcohol, opium, and other drugs that depress the nervous system. REQUIREMENTS APPARATUS 1. Beakers 250 ml. 2. Pippete 5 ml. 3. Burette stand and Burette. 4. Conical flasks 5. Brunner. 6. Measuring flask. 7. Separating funnel. 8. Filter paper. 9. Weight box. 10. Analytical balance. MATERIALS REQUIRED Tea leaves of different kinds. CHEMICALS 1. Lead Acetate. 2. Chloroform. 3. N/50 NaOH solution. PROCEDURE A) ESTIMATION OF ACID CONTENT PRESENT IN TEA LEAVES 10, gm of each of tea leaves are mixed in the beakers. Each containing of 200 ml of water. The contents of the beaker are then heated constantly for about 30 minutes and the extract is filtered out. 5 ml of tea extract is taken in a conical flask and added to 20 ml of distilled water. It is now shaken to prepare a homogeneous mixture and titrated against N/50 NaOH solution. The same procedure is carried for other types of tea leaves. B) MODE OF TAKING OBSERVATIONS A white plate is put below the conical flask, while titrating tea extract against n/50 NaOH solution,. The moment colour of the solution in conical flask becomes dark, titration is stopped and thus volume of NaOH is calculated. C) TO EXTRACT THE AMOUNT OF GLYCOXIDE COMPOUND PRESENT IN TEA LEAVES The well boiled and measured tea extract is treated with lead acetate which precipitates the glycoxide compound present in the tea leaves in the form of lead complex and is fiecered. The filtrage is treated with small amount of chloroform. Chloroform layer containing glycoxide compound is seprated by separating funnel. From the chloroform extract, chloroform . is distilled out leaving residue in the flask. The residue is dissolved in hot water and the glycoxide compound is obtained. It is assumed that the amount of glycoxide compound present is proportional to the amount of Caffeine present. OBSERVATION TABLE NO. 1 S.NO. TEA LEAVES INITIAL VOLUME FINAL VOLUME VOLUME OF NaOH USED 1 Brooke Bond 0 ml 4.1 ml 4.1 ml 2. Taj Mahal 10 ml 13.6 ml 3.6 ml 3. Lipton 20 ml 23.3 ml 3.3 ml 4. Open Tea 0 ml 3.1 ml 3.1 ml CALCULATIONS Gram molecular weight of solute NaOH is 40 Molarity is 0.02 M Now, Gram of solute per litre Molarity = 40 0.02 x 40 = Grams of solute (NaOH) per litre. .8 gm = Grams of solute (NaOH) per litre. .2 gm = Grams of NaOH per 250 ml. TABLE NO. 2 S.NO. TEA LEAVES NORMALITY OF TEA EXTRACT STRENGTH OF ACID 1. Brooke Bond 4.1/250 0.076 Wgm/lt. 2. Taj Mahal 3.6/250 0.015 Wgm/lt. 3. Lipton 3.3/250 0.013 Wgm/lt. 4. Open Tea 3.1/250 0.012 CALCULATIONS N/50 NaOH solution is same as that of M/50 NaOH solution NORMALLY EQUIATION : N1V1 = N2V2 Where N1 = N/50 , N2 = V1 = Volume of NaOH taken V2 = Volume of tea extract used, 5 ml. ? Wgm/lt. TABLE NO. 3 Compound present – now is noted in this table S.NO. TEA LEAVES TEA EXTRACT Mgm / GLYCOXIDE COMPOUND 1. Brooke Bond 100 ml 50 Mgm 2. Taj Mahal 100 ml 56.4 Mgm 3. Lipton 100 ml 59.4 Mgm 4. Open Tea 100 ml 62.6 Mgm RESULT 1. When a Graph between acidity strength in tea and weight of glycosoidal compound is plotted, we obtained a straight line which shows the amount of caffeine present in tea leaves is inversely proportional to the strength of the acids in the tea extract. 2. Different tastes of different teas is due to the variation of amount of Caffiene present. 3. The best type of tea is lipton green lable. BIBLILOGRAPHY 1. PL Soni : Inorganic Chemistry. 2. World Book. 3. Encyclopedia of Science and Nature.