The Gold Penny Lab - Miss Hwang's Chemistry Class
advertisement

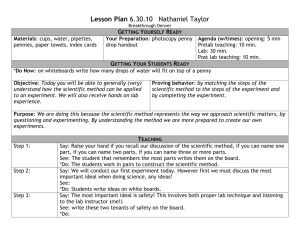
The Gold Penny Lab In this lab, you will be converting a regular penny into a “gold” penny. In doing so, you are following a tradition that goes back to the earliest days of chemistry. The modern practice of chemistry started with the study of alchemy in medieval Europe and the Middle East. Alchemists believed that by doing certain chemical reactions, you could turn cheap metals into gold. Some of them did, anyway- the others would convince people that they could do this and sell them the secret. Safety: In this lab, it is very important that you wear your goggles, appropriate footwear, and aprons at all times! The chemicals that we are working with are extremely dangerous and can cause serious injury. Pre-Lab Assignment: *Please type and answer in complete sentences. (3 pt each) **Have it ready before starting the lab. 1. Watch the video on youtube. Search for Alchemy Lab by chemteacher08. The link is on my website in case you can’t find it. http://www.youtube.com/watch?v=TCjeC1FNPdo 2. Read the procedures for making a silver penny and a gold penny. Make a list of the equipment you will need to do this experiment. 3. Sodium Hydroxide is very dangerous. Use the website http://www.atsdr.cdc.gov/tfacts178.html to find out how it could hurt you if you use it improperly. List 2 ways that sodium hydroxide can affect your health: 4. What is alchemy? 5. Come to class with a penny prepared by following the direction in part one. Part One- Preparing the Penny You need a shiny penny to do this lab! 1. If your penny is not shiny, place it into a dish, and pour distilled white vinegar over it. 2. Allow the penny to sit for 5 minutes. 3. Take the penny out, and clean it with water and a paper towel. 4. If the penny is still not shiny, use a piece of steel wool to sand off the dirt. Part Two: Making a Silver Penny 1. Carefully put one full spoonful of granular zinc into an beaker. 2. Use a graduated cylinder to measure 20 mL of NaOH solution. Pour this into the beaker with the zinc. 3. Heat the beaker and its contents so that the solution is gently simmering. 4. Carefully add the penny to the solution and allow to simmer for 1 minute. Do not let the water boil! The set up should look like this: * We are using a beaker instead of the dish* 5. After 1 minute, use tongs to turn the penny over and allow it to simmer for another minute or until you observe a uniform color. 6. With tongs, remove the penny form the beaker and drop the penny into a beaker of water. 7. With tongs, remove the penny from the water. 8. Dry the penny with a paper towel. Silver Penny Observation (3 pt each) 1. Describe what your penny looks like at this point in time: 2. Do you think that this is real silver? Why or why not? Part Three: Making a Gold Penny 9. Using the tongs, hold the penny heads up in the flame of the Bunsen burner for 3 seconds. 10. Remove from heat, flip the penny and re-heat tails side up for another 3 seconds. 11. Repeat carefully until you see a color change. WARNING: Pennies will melt if left in the fire too long! The zinc core of a penny melts easily! 12. Set the penny on the bench top to cool. Rinse the penny a second time. Gold Penny Observation (3pt each) 3. Describe what your penny looks like at this point in time: 4. Do you think that this is real gold? Why or why not? Cleaning Up Disposal of Sodium Hydroxide: After it has cooled, carefully pour the sodium hydroxide into the sink. Rinse down the drain with water. Disposal of Zinc: Once sodium hydroxide is out of the dish, pour a little water into the dish, swirl, and drain. Rinse with water again, drain. Use paper towels to wipe the zinc into the trash can. Equipment/Glassware: Wash everything and return it to where you got it. Alchemy Post-Lab Questions (3pt each) 1. An alloy is a mixture of two or more types of metals. There are three very common alloys: Brass, Bronze, and Steel. Look up the metals that compose each type of alloy. 2. What metals are used to make pennies? Conclusion (10 pt) 1. How does this experiment tie back to what you learned about ions? Discuss using these terms: ionic bonding, cation, anion, valence electron, zinc, copper… a. Include these terms, and its definitions. Please highlight these terms in your lab report. b. Research on line for the best explanation of this experiment. c. Explain thoroughly. Discuss and connect back to the core concept of ionic bonding. d. Paragraph 1 – discussion of ionic bonding e. Paragraph 2 – discussion of the explanation of the lab Alchemy Final Lab Report Title - 2pt Purpose - 2pt Pre-lab Assignment – 12pt Observation Questions - 1. Silver penny 2. Gold penny - 6pt Post-lab questions – 6pt Conclusion – 10pt Instructions for the lab report *Must be in complete sentences, typed in 12pt font, Times New Roman* *re-include the pre-lab assignment into your final lab report* *attach the handwritten observation questions to the back of the final report* *compile them in the same order as above* * PLEASE TURN IN QUALITY WORK*