G6-B Act.1 - Chemistry Laboratory
advertisement

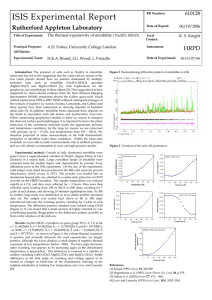
Page 1 of 3 CSA CHEMISTRY LABORATORY REPORT SY: 2012 – 2013 Title:Names and Formulas Of Ionic Compounds Yr&Sec:3B Group# 6 Members Luigi Custodio Margarette Castro Dhiren Dhanani CSA SCIENCE PROMOTES THE CULTURE OF EXCELLENCE Experiment Date [ June 20, 2012] M, T, W, Th, F Date Submitted [ June 25,2012] Performance Rubrics Lab Report Rating 50 11 24 25 Lab Activity Score ASSESSMENT Performance Criteria PARTS ABSTRACT 1. We plan to mix the chemicals, Observe the Formation Of Compounds, and to record their names and formulas. This will guide us in either discovering new compounds or re-creating old ones. It will also help us in differentiating the different chemical reactions that will occur when 2 chemicals are mixed. We plan to use a Pencil, Paper, Ruler, Reaction Surface and a Dropper to mix the chemicals, record the observations and to differentiate the chemicals that we’ll use . We’ll also use the chemicals: FeCl3, MgSO4, CuSO4 and group them as chemical A. And we’ll use the chemicals: Na2CO3, Na3PO4, NaOH, Na2So4 and group them as Chemicals B. Once we are done grouping, we will then proceed with the experimentation. In order to Fully Accomplish the task at hand, We have devised a procedure and we used the table form the activity sheet as a basis. 1st , we grouped the chemicals, the chemicals without calcium are marked as group A. Then, the ones with calcium are marked as group B. 2nd , We then mix the chemicals from Group A to Group B. 3rd, We then distinguish whether the precipitate is Grainy, Milky, Cloudy or Gelatinous. 4th , We record the Data and we Construct a report using the recorded data as a result from the experiment. By accomplishing the experiment, We plan to find the outcome of the combined chemicals and differentiate it from one another. We’ll also use the newly formed compounds as a basis whether a chemical reaction was formed or not. We therefore conclude that chemical reaction takes place when 2 certain chemicals are mixed together. This is significant in our lives because everything in this world is made up of chemicals. And when we further study the Science of Chemistry, we can therefore improve everything around us. We recommend the students that they practice their patience and work together as one because Chemistry is hard stuff. 2. 3. This serves as the overview of the report. Objectives, materials used, procedure, findings, conclusion, application and/or recommendation are BRIEFLY mentioned. This part should be CONCISE. P S Points Score 5 2 Remarks (For teacher’s use only.) Page 2 of 3 PRESENTATION OF DATA A. Sample Calculation/s 5 4. Not Applicable 5. 6. B. Tables and/or Graphs Na2CO3 (CO32-) Na3PO4 (PO43-) NaOH (OH-) Na2SO4 (SO42-) FeCI3 MgSO4 CuSO4 (Fe3+) (Mg2+) (Cu2+) No Reaction No Reaction No Reaction No Reaction No Reaction 7. Gelatinous No Reaction Gelatinous No Reaction Gelatinous No Reaction Gelatinous 8. IF APPLICABLE, demonstrate how each formula is being used in computing for the values. The computations are presented logically, never setting aside the rules learned in math classes. The group chooses the PROPER form of presenting the data, whether in tabular form, graphical form or both. The design of the tables and graphs is APPROPRIATE for the activity (including but not limited to the interval of values, determination of constants, proper placement of dependent, independent or manipulated variables, as the need arises). All parts of the table or graph are labeled PROPERLY (including but not limited to title, units, x-axis, y-axis, header, etc.) ANALYSIS 1. 5 Describe each Precipitate? Cloudy Milky Grainy Gelatinous 2. 2 Write Formulas and Names of the Chemical compounds produced in the mixings in ionic. Na2CO3+FeCI3= Fe2(CO3)3 Na2CO3 +MgSo4= Na2SO4(aq) + MgCO3(s) Na2CO3+CuSo4= CuCO3(aq) + Na2SO4(ag) Na3PO4+FeCI3= Na3PO4+MgSo4= Na3PO4+CuSo4= FePO4 + NaCl Na2SO4 + Mg3(PO4)2 Cu3(PO4)2 (s) + 3 Na2SO4 (aq) NaOH +FeCI3= NaOH +MgSo4= NaOH + CuSo4= NaCI Fe(OH)3 MgOH(aq)+Na2SO4 Cu(OH)2 + Na2SO4 Na2SO4 + FeCI3= NaCl+FeSO4 Na2SO4 +CuSo4= Na2SO4+CuSO4 Na2SO4 +MgSo4= Na2SO4+MgSo4 9. When the original procedure paper CONTAINS Follow-up Questions given by the teacher, the lab report answers all the questions. 10. If the original procedure paper does NOT contain Follow-up Questions, the lab report presents a discussion on the implications of the data gathered. 11. Matters that are indirectly discovered are discussed including the effects of the limitations of the activity on the results. 12. All discussions of data are in lieu with the objectives of the activity and are supported by the data. 13. Information from published resources are properly cited. 1 Your table confuses me…I do not know what you want to show. Page 3 of 3 CONCLUSION We conclude that mixing chemical compounds does create different chemical reactions. These chemical reactions changes the physical appearances of the combined chemicals therefore ushering into a new type of chemical compound. These chemical reactions create new types of elements/chemicals. And with new elements/chemicals come more chemical reactions. These reactions change the appearances of the chemicals therefore creating a compound. The world is full of elements, and we have barely breached into the surface of chemistry. 5 14. Questions directly or indirectly posted by the objectives are addressed clearly and are consistent with the gathered data. RECOMMENDATION We recommend Patience, Keen Observation, determination and Full Cooperation from every member as this subject is hard to accomplish. And after the students have surpassed the particular subject and have moved on to the next level, they will have the satisfaction of Fulfilling a shared goal as a group. The students will need every hands available in their group in order to fully unleash their potential as one. Unity is the key to Victory. References http://www.webqc.org/balance.php?reaction=NaOH%28s%29+%2B+ MgSO4%28aq%29+%3D+MgOH%28aq%29+%2B+Na2SO4 http://answers.yahoo.com/question/index?qid=20071001144345AA3 51cO http://www.scienceforums.net/topic/39990-naoh-cuso4/ Almost correct just be specific. 2 1 15. Weaknesses of the design of the experiment are discussed. 16. Future students and teachers are being advised on how to IMPROVE the activity. APPLICATION Chemistry is the building foundation of every man made things in this world. we can apply this in our lives by using chemistry to further improve the use of anything in our lives, or to invent new things that will make life easier for us. And, after we have dug deeper into the Foundation of Chemistry, we can make a world of Limitless Potential. 3 Better if you are going to include the technical aspect of the experiment not only about the attitude and behavior of each student in the group. 2 1 17. The topic of the activity is being related to the situation in the community. The more recent, the better. 18. Following the APA format of citation Be specific. 1 1