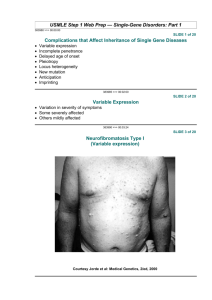
Marfan Syndrome (MFS) Description
advertisement

What is Marfan’s Syndrome? Marfan’s Syndrome is a genetic disorder that affects 1 in 10,000 – 20,000 individuals. This Marfan Syndrome (MFS) disorder affects the body’s connective tissue. It is a malformation of Chromosome 15, the part that is used to produce this tissue. Javad Tavakkoly Bazzaz MD, PhD Department of Medical Genetics, Faculty of Medicine, It usually occurs equally in males and in females. Tehran University of Medical Sciences (TUMS) It Description is life threatening Symptoms Heritable disorder of the connective tissue One of the symptoms of this syndrome is that people with it usually tend to develop an enlargement of the aorta, the main blood vessel that takes blood from the heart to the rest of the body. Connective tissue affects: Growth and development Cushioning of joints Vital organs Not tied to any particular sex, race, or ethnic group Another symptom is that many people are very tall and thin. 1 in 5,000 people in US have disorder Organ Systems Affected Skeletal CNS Oral Skin Cavity Eyes Cardiovascular Pulmonary Skeletal Tall, slender stature Joint hypermobility Thumb sign Wrist sign Reduced elbow extension Affects long bones Arm span > Height <170 degrees arms/legs/fingers/toes may be disproportionately long ratio >1.05 Long, narrow face Sternal deformity Pectus escavatum (funnel chest) Pectus carinatum (pigeon’s chest) Scoliosis Pes plantus (flat feet) Positive Walker Wrist Sign Tall and slender Skeleton Disproportionately long appendages the distal phalange of the first and fifth fingers of the hand overlap when wrapped around the opposite wrist Indented or protruding sternum Arched palate, overcrowded teeth, receding mandible Curvature of spine Positive Steinburg Thumb Sign the thumb projects beyond the ulnar border while completely opposed within the clenched hand Oral Cavity Arched palate crowded dentition Eyes Off-center or dislocated lenses Nearsightedness Development of cataracts at a younger age 30s to 50s Mandible malocclusion Retinal detachment Holes or tears in inner lining Heart and Blood vessels Abnormally large mitral valve leaflets Heart and Blood vessels Stretched aortic valve leaflets Causes prolapse Present in 75 % of cases Aneurysm may form Mitral regurgitation Can lead to breathless, extreme exhaustion, irregular pulse Aortic regurgitation Left ventricle must compensate Heart murmurs Long-term damage to heart Weakened middle layer of aortic wall Chest pain, heart failure Tears in inner and middle aortic layers Middle layer separates New channels for blood flow Nervous System Lungs Diminished alveoli elasticity Susceptibility to asthma, bronchitis, pneumonia In rare cases, develop emphysema Stretching and enlargement of dura membrane Pushes on and wears down vertebrae Can protrude through vertebral column and into abdomen 5% experience spontaneous lung collapse Sleep disordered breathing Snoring most common Caused by partial obstruction of airway by connective tissue Dural cysts Increased susceptibility to learning disabilities Who is affected by Marfan’s? Skin Stretch Many marks Shoulders, hips, lower back Increased risk for abdominal hernias people are affected by this syndrome and many of those affected by this kind of syndrome are athletes. Some of them use this syndrome to their advantage, especially basketball and volleyball players. Due to Marfan’s Syndrome many athletes sometimes die without warning. The average age of death for people with Marfan’s Syndrome is 32. What Is The Cause of Death? cause of death in people with Marfan’s Syndrome are complications with the heart. Cures?? The beta Since Aortic the aorta is enlarging and thinning, the aorta might tear and the aortic valve might also start a leak, thus leading to heart failure. blockers to cure or try to treat this syndrome. valve replacement. Epidemiology Work-up Incidence at least 1 in 5,000 in the US Inheritance H&P including family history AD, Variable Penetrance Gender men, women Eye Exam slit lamp lens dislocation Risk Child of parent with MFS: 50% chance Prenatal testing available but given unpredictable phenotype not widely used Echo looking for Aortic root dilation Two unaffected parents: 1 in 10,000 ~ 25% from a spontaneous mutation Differential Diagnosis Homocystinuria marfanoid habitus, ectopia lentis, mental retardation, osteoporosis MASS phenotype Diagnostic Criteria Major criterion: highly specific & rarely occur in the general population MVP, mild aortic dilatation, striae atrophica, skeletal involvement Congenital contractural arachnodactyly arachnodactyly, joint contractures, scoliosis, crumpled ear malformation Ehlers-Danlos syndromes types II & III marked joint hypermobility, paparaceous scars, mitral prolapse marfanoid habitus, small testes and genitalia, learning difficulty aortic dilation may involve primarily the aortic root or the mid-ascending aorta Aortic coarctation with associated ascending aortic enlargement Loeys-Dietz syndrome Diagnostic Criteria Chest wall deformities Long, thin arms/legs Scoliosis greater than 20 degrees Klinefelter syndrome Congenital bicuspid aortic valve disease with associated aortopathy aortic root enlargement (aortic aneurysm) aortic dissection lens dislocation dural ectasia at least 4 major skeletal features Minor criterion: features with Marfan’s but may also occur in the general population Diagnosis requires: + FamHx: only 1 Major criteria – FamHx: at least 2 Major criteria What is Marfan’s Syndrome Fibrillin Æ building block of microfibrils, formation of extracellular matrix Main protein of CT Microfibils including elastic fibers Abundant in the aorta, ligaments, cilliary body of the eye MFS: mutation in the fibrillin-1 gene (FBN1) chromosome 15 Basic Genetic Information Autosomal Dominant Dominant Negative Mutation – the altered gene product antagonizes the product of the normal gene Haploinsufficiency – when a diploid organism has only one functional copy of a gene, the other copy being mutated FBN-1 Gene Located on chromosome 15 Codes instructions for the creation of protein Fibrillin 1 Marfan’s is caused by over 500 different mutations on FBN1 60% mutations are change in one protein building block. 40% mutations produce small protein that can’t function Affects FBN-1 Gene Fibrillin 1 protein Connect with other Fibrillin 1 proteins to make microfibrils, which become connective tissue. The fibrillin‐1 gene FBN1 spans about 235 kb of genomic DNA on chromosome 15q21.1, and has a transcript size of 9749 nucleotides; the coding sequence of FBN1 is spread over 65 exons, and three alternatively spliced non‐coding 5′ exons have been described. With several exceptions, single exons code for the domains in fibrillin‐1 as shown here. J Med Genet. 2006 October; 43(10): 769–787. Defective Fibrillin 1 Protein Amount of fibrillin 1 protein produced by cells is reduced Structure and stability of protein is affected Transport of fibrillin 1 protein out of the cell is impaired Less microfibril leads to more active TGF-beta, which leads to Marfan’s symptoms fibrillin 1deficiency contributes to MFS progression through altered cell-matrix interactions and dysregulated TGF- signaling. Microfibrils mainly trap transforming growth factor-beta (TGF-beta) and keeps them inactive. A new paradigm has emerged from the study of mouse models of MFS. Specifically, the new paradigm postulates that matrix sequestration is critical to regulated activation of latent TGF-β and that perturbations of this function contributes to the pathogenesis of MFS disease. TGF Beta Pathway: TGF Beta Receptor 2 is a Ser/Thr Receptor Kinase Function of TGF Beta Pathway in Normal Cells Pattern formation during embryonic development Cell growth and proliferation Cellular differentiation Extracellular matrix production Angiogenesis Tissue repair Immune regulation Apoptosis http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=mboc4.figgrp.2875 TGF-β TGFβ – Cytokine involved in scarring controls proliferation, cellular differentiation acts as an antiproliferative factor in normal epithelial cells TGF-β is a secreted protein part of a superfamily which includes inhibins, activin, antimüllerian hormone, bone morphogenetic protein, decapentaplegic and Vg-1 Fibrillin-1 indirectly binds a latent form of TGFβ Keeps TGFβ sequestered and unable to exert its biological activity MFS: low levels of fibrillin-1 Æ TGFβ levels rise 2/2, inadequate sequestration Æ inflammatory reaction releasing proteases Æ degrade elastin fibers & other components of the ECM TGF-β and ARB ARB (losartan) inhibits TGF-β signaling Attenuation pathologic changes in the aortic wall progressive dilation of the aortic root TGF-β activation The 3 TGF-β isoforms are synthesized and homodimeric pro-TGF-β = TGF covalently linked to latency associated protein (LAP) Intracellularly cleaved by furin-type enzymes--> mature TGF-β , remains non-covalently associated with LAP= SLC Few cells produce and secrete diffusible SLC. Majority secrete TGF as part of the LLC, formed within secretory vesicles: covalent disulfide bond between the SLC and a member of the LTBP family LTBP-1,-3,-4 bind TGF-βs. LTBPs belong to the superfamily of fibrillin-like ECM proteins Bind a series of ECM proteins TGF-β is synthesized in excess, and bioavailability depends on activation Testing and Diagnosis Genetic Types Complete bi-directional DNA sequencing Familial known mutation test Prenatal If parent or close relative has FBN1 or TGFBR mutation Costs FBN1 gene sequencing $1,599 TGFBR gene sequencing $599 Familial mutation test $250 Testing and Diagnosis Physical Activity No definitive genetic tests available Avoid contact and strenuous sports because of the risk of damaging the aorta and injuring the eyes Individual restrictions based on severity and discussed with physician Accuracy 99.9% accuracy in detecting mutations 70-90% mutation detection rate in individuals with clinical diagnosis Contact/collision high potential: strenuous basketball, boxing, field hockey, football, ice hockey, lacrosse martial arts, rodeo, skiing (water) soccer, wrestling Limited contact: strenuous baseball, bicycling (intense) gymnastics, horseback riding, skating (ice & roller) skiing (downhill & cross-country) softball, squash, volleyball Noncontact: strenuous aerobic dancing (high impact) crew, running (fast), weightlifting Noncontact: moderately strenuous aerobic dancing (low impact), badminton bicycling (leisurely), jogging, swimming (leisurely) table tennis, tennis Noncontact: nonstrenuous golf, bowling riflery, walking Flo Hyman July 31, 1954-Jan. 24, 1986 Olympic Volleyball Player 6 foot 5 inches Died on court from Marfan Marfan syndrome type 1 Marfan syndrome type 2 The classical Marfan syndrome (MFS1, OMIM 154700) is a systemic disease of connective tissue. MFS1 is caused by mutations in the fibrillin-1 gene (FBN1) on chromosome 15q21.1. All organ systems may be affected, but particularly the skeleton, the eye and the cardiovascular system. The classical Marfan syndrome (MFS1, OMIM 154700) is a systemic disease of connective tissue. MFS1 is caused by mutations in the fibrillin-1 gene (FBN1) on chromosome 15q21.1. All organ systems may be affected, but particularly the skeleton, the eye and the cardiovascular system. Although there are rare cases of very severely affected neonates, the symptoms normally develop during the first two decades of life. It is often difficult to decide on the basis of the clinical symptoms alone whether a child or adolescent from an affected family has inherited the disorder; genetic diagnosis may then be indicated. Although there are rare cases of very severely affected neonates, the symptoms normally develop during the first two decades of life. It is often difficult to decide on the basis of the clinical symptoms alone whether a child or adolescent from an affected family has inherited the disorder; genetic diagnosis may then be indicated. Because of the very variable phenotypic development, it is often not possible to achieve a reliable diagnosis of classical Marfan syndrome in adults either. It often happens that only some of the symptoms are displayed throughout life. Because of the very variable phenotypic development, it is often not possible to achieve a reliable diagnosis of classical Marfan syndrome in adults either. It often happens that only some of the symptoms are displayed throughout life. Marfan syndrome type 2 Marfan syndrome type 2 (MFS2, OMIM 154705) is very similar to classical Marfan syndrome (MFS1). Ectopia lentis has not yet been found in any patient with MFS2. However, this symptom may also be missing in MFS1, so that it is not suitable for differential diagnosis. MFS2 has been mapped onto chromosome section 3p22. The syndrome is caused by mutations in the gene for transforming growth factor beta receptor 2 (TGFBR2) or, more rarely, of the TGFBR1 gene. Patients have been identified in France, Japan, Germany, and Italy. It is not possible to distinguish MFS2 from MFS1 with clinical genetic testing, but only by performing a molecular genetic analysis. This differentiation is nevertheless of great importance. It is thought that TGFBR mutations may be associated with an aggressive clinical course, due to vascular complications. This is accompanied by earlier aortic dissection. MASS syndrome The designation MASS syndrome (OMIM 60438) is an acronym for mitral valve prolapse, aortic dilatation, with skeletal and skin involvement. The patients do not fulfill the clinical criteria for Marfan syndrome, but exhibit at least mitral valve prolapse. In some patients this syndrome is caused, as in MFS1, by mutations in the FBN1 gene, but the cause of the disease is not known for most of these patients. Mitral valve prolapse may be a component of the familial mitral valve prolapse syndrome (OMIM 157700), but can also occur in the Ehlers-Danlos syndrome and in osteogenesis imperfecta. Weil-Marchesani syndrome The phenotypic presentation of the WeilMarchesani syndrome (WMS, OMIM 608328) may be regarded as the opposite of MFS1 and MFS2. The patients are rather small. They have short fingers and suffer from stiff joints. However, they share ectopia lentis with MFS1. Like MFS1, WMS is caused by mutations in the FBN1 gene. Aside from the autosomal dominant form, there is also an autosomal recessive form of WMS (OMIM 277600), caused by mutations in the ADAMTS10 gene. Loeys-Dietz syndrome type 1 This syndrome was first described in 2005 (LDS1, OMIM 609192), but is probably identical to the Furlong syndrome (OMIM 610168), which has been known since 1987. Almost all symptoms of the classical Marfan syndrome can also be found in LDS1. In addition, the patients suffer from craniofacial malformations (cleft palate or cleft uvula, hypertelorism) and particularly from vascular changes, extending to dilatation, aneurysm formation, and dissection throughout the arterial system. Like MFS2, LDS1 is caused by mutations in genes TGFBR1 (on ch. 9q33–q34) and TGFBR2 (on ch. 3p22). The gene products play a decisive role in intracellular transduction of TGF-beta signals. It is known from animal models for mitral valve prolapse and pulmonary symptoms that these signals are important for the development of the vascular system and of the visceral cranium. The developmental disturbances in the vascular walls are evident in even very young patients. This may explain why complications often develop so early. The mean age for surgical vascular replacement is 16.9 years. Congenital contractural arachnodactyly Congenital contractural arachnodactyly (CCA, OMIM 121050) resembles MFS clinically, but is accompanied by characteristic joint contractions and often a deformity of the auricles ("crumpled ears"). CCA is caused by mutations in the fibrillin-2 gene (FBN2). Loeys-Dietz syndrome type 2 The symptoms of Loeys-Dietz syndrome type 1 (LDS1) overlap with the vascular type of the EhlersDanlos syndrome (EDS type 4, EDS4, OMIM 130050). EDS4 is often caused by mutations in the collagen type 3 gene. Loeys et al. investigated 40 patients with the clinical presentation of EDS4, but without collagen type 3 disturbances or craniofacial malformation. In 12 patients, they identified mutations in one of the TGFBR genes. These patients are now assigned to the Loeys-Dietz syndrome type 2 (LDS2, OMIM number not yet allocated). As with LDS1, LDS2 patients suffer from aggressive arterial aneurysms. The mean age for vascular replacement is 26.9 years. Shprintzen-Goldberg syndrome The Shprintzen-Goldberg syndrome (SGS, OMIM 182212) exhibits common features with both MFS1 and MFS2, as well as with LDS1 (or the Furlong syndrome) and LDS2. The most striking feature of SGS is cranial synostosis, which does not occur in MFS1 or MFS2, but does occur in LDS1. No mutations have been found in any of the TGFBR genes in SGS, although there are mutations in FBN1. These clinical findings indicate that there is a connection between fibrillin deficiency and disturbances in the TGFB signal cascade. This could open specific therapeutic options. Questions? Familial thoracic aortic aneurysm Just as with mitral valve prolapse, aortic aneurysm may either by a component of a syndrome or may be inherited separately (OMIM 607086). Both the syndromal and familial forms are histologically linked to Erdheim-Gsell cystic medial necrosis, with loss of elastic fibers, deposition of material resembling mucopolysaccharides and cystic changes in the medial aortic wall. Five responsible chromosome regions have been identified (AAT1 to AAT5). AAT3 can be caused by mutations in the TGFBR2 gene, AAT4 by mutations in the gene for myosin heavy chain 11 (MYH11) and AAT5 by changes in the TGFBR1 gene. The underlying genes are not yet known for AAT1 and AAT2. Familial thoracic aortic aneurysm with dissection (TAAD) can also be caused by mutations in the ACTA2 gene. It has long been known that a bicuspid aortic valve (BAV) is often associated with cystic medial necrosis and the risk of an acute aortic syndrome. Familial forms of the BAV may be caused by mutations in the NOTCH1 gene (OMIM 190198). It has become increasingly important for the prognosis and treatment of these syndromes to identify the genetic causes of aneurysm formation.