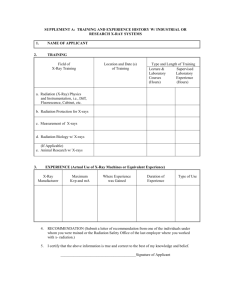
Quantum Physical Phenomena in Life (and Medical) Sciences IV. Röntgen (X-) rays Activated tube Péter Maróti Professor of Biophysics, University of Szeged, Hungary Suggested texts: S. Damjanovich, J. Fidy and J. Szőlősi: Medical Biophysics, Semmelweis, Budapest 2006 P. Maróti, L. Berkes and F. Tölgyesi: Biophysics Problems, A Textbook with Answers, Akadémiai Kiadó, Bp. 1998. Schoolexample for very fast and successful innovation X-ray quantum energy in diagnostics: 30-200 keV in therapy: 5-20 MeV Hand mit Ringen (Hand with Rings): print of Wilhelm Röntgen’s first "medical" X-ray, of his wife's hand, taken on 22 December 1895. Properties of the X-rays Röntgen demonstrated that X-rays: - produced luminescence from the wall of the discharge tube, - traveled in straight lines, - were not deflected by a magnetic field, - were absorbed more by denser metals, - were scattered when passing through a body and - could ionize gases. For complete description of the X-ray, we need - the anode voltage: it determines the energy of the X-ray quanta (photons), - the composition (type) of material of the anode: it determines the wavelength of the characteristic radiation and - the quality and thickness of the filters: they determine the hardness (softness) of the radiation. This talk concentrates on the physical bases of the X-radiation and the very important medical applications in diagnostics (e.g. CT) and therapy are left for other lectures. Generation of X-rays X-ray beams from small spot size Coolidge (high vacuum) tube Double focus x-ray tube The smaller is F the sharper is the imaging spot size, F D object R = F·d/D d R The smaller is R the larger is the contrast Short exposure (long filement) long exposure (short filement) X-ray tubes Coolidge X-ray tube, from around 1917. The heated cathode is on the left, and the anode is right. The X-rays are emitted downwards. Different X-ray tubes bulb diameter 7 cm The larger tube has a length of about 25 centimeter. It has a regulator, a small glass compartment on the tube with a piece of charcoal which could be heated to correct the internal gas vacuum. The small tube measures about 15 centimeter with 5 cm bulb. Rotating anode tube The anode can be rotated by electromagnetic induction from a series of stator windings outside the evacuated tube. A: Anode C: cathode T: Anode target W: X-ray window X-ray generation by accelerators Linac The linear accelerator, or linac, is the electromagnetic catapult that brings electrons from a standing start to relativistic velocity - a velocity near the speed of light. The buncher accelerates the pulsing electrons as they come out of the electron gun and pack them into bunches. To do this the buncher receives powerful microwave radiation which accelerates the electrons in somewhat the same way that ocean waves accelerate surfers on surfboards. When the cyclotron principle is used to accelerated electrons, it has been historically called a betatron. The betatron (Donald Kerst, 1940) is a device for accelerating electrons to higher speeds as they move in a circular orbit. The main question is: how can one keep the electrons in a circular orbit as they move faster and faster? The portable X-ray betatron is a compact circular electron accelerator producing a high energy directional X-ray beam. etarget α The (angular) divergence of the X-ray: Basic Technical Data X-Ray output energy selector: 2 to 6 MeV Dose rate at 3' (air): 3R (3cGY) per minute Focal spot size: .01" x .039" Radiation beam spread angle: 26 degrees Radiographic sensitivity: down to 0.5% AC power input: 110/240V 50/60 Hz α = me·c2 /E me is the mass of the electron, c is the speed of the light and E is the energy of the accelerated electrons when they leave the betatron. X-ray spectra Characteristic radiation: the energy levels of the electrons are quantized, thus the produced radiation is also quantized, demonstrates spectral lines which are characteristic on the target material (element). Bremsstrahlung: Incident electrons are deflected by the negative charge of electrons in the target. Any change in velocity (speed or direction or both) is an acceleration. Accelerating a charge emits radiation. The deflected electrons’ acceleration is NOT QUANTIZED. Thus, the spectrum is continuous. A section from the previous 3D spectrum at constant tube voltage (60 kV) Kα characteristic K lines Kβ Bremsstrahlung λmin Spectrum of the X-rays emitted by an X-ray tube with a rhodium target, operated at 60 kV. The smooth, continuous curve is due to Bremsstrahlung, and the spikes are characteristic K lines for rhodium atoms. The X-rays are extra-nuclear and have two sources: (1). A characteristic (e.g. K- or L-) X-ray photon is created and emitted when an electron drops down orbits to fill a vacancy in an innermost shell. The wavelength of the photon is unique to the atom and affords an unambigious method of identifying the element by use of an X-ray spectrometer . (2). A bremsstrahlung photon is produced when a high speed electron decelerates when passing through the electric field in the close vicinity of an atomic nucleus. The spectrum in this case is continuous up to the maximum energy of the bombarding electrons. Braking radiation („Bremsstrahlung”): Duane–Hunt displacement law It gives the maximum frequency of X-rays that can be emitted by Bremsstrahlung in an X-ray tube by accelerating electrons of electric charge e through an excitation voltage V into a metal target. The maximum frequency νmax is given by νmax = eV/h which corresponds to a minimum wavelength λmin = (hc)/(eV) where h is the Planck’s constant and c is the speed of light. The law is in effect a statement of conservation of energy. The maximum frequency (minimum wavelength) is that at which all of the electrical energy E = eV given to the electron is successfully converted into radiation energy E = hν. This process is also known as the inverse photoelectric effect. Bremsstrahlung: emitted power, efficiency The total emitted energy (or power, if the radiation is stationary) is the area under ¥ the curve: Wtotal = ò 0 DN × dE DE Let’s replace the function by straight line: DN » const × Z × (Emax - E ) DE and calculate the integral (the area of the triangle): Emax Wtotal » const × Z × ò (E 0 After expressing Emax from the anode voltage of the tube: max 2 Emax - E )dE = const × Z × 2 2 Wtotal = const/2 × Z × e 2 Vanode The total emitted power increases proportionally with the number of electrons impacting the anode within unit of time, i.e. with the anode current: 2 Ptotal = cRtg × Z I anode Vanode Here Z is the atomic number of the material of the anode, Vanode is the accelerating voltage, Ianode is the anode current and cRtg ≈ 1.1·10-9 V-1. The total emitted power is proportional to the square of the anode (tube) voltage. The radiation production efficiency in an X-ray tube: 2 cRtg × Z I anode Vanode Ptotal = = cRtg × Z Vanode h= Pinvested I anode Vanode For Tungsten anode at Vanode = 100 kV, η ≈ 0.008 < 1%. The energy is largely transforms to heat. Characteristic radiation: the Moseley's Law the heavier elements have up to three lines The frequency of characteristic K-series X-rays produced from tubes with different anode materials (Ca to Zn) was measured. The ordering of the wavelengths of the X-ray emissions of the elements coincided with the ordering of the elements by atomic number. the lighter elements have two distinctive characteristic X-ray There is linear relationship between the atomic lines number of the element the anode consists of and the square root of the frequency. Moseley's law as an empirical law n = k1 × (Z - k2 ) where ν is the frequency of the main or K x-ray emission line, k1 and k2 are constants that depend on the type of the line. For example, the values for k1 and k2 are the same for all Kα lines, so the formula can be rewritten thus ν = 2.47·1015 ·(Z - 1)2 Hz Moseley’s law: derived from Bohr’s atomic structure and spectroscopic term system The energy of the X-ray lines (as difference of two energy terms) can be derived in a very similar way as done with the optical spectral lines of the H-atom in the Bohr model. The wavenumber of the transition is æ 1 ö 1 = n = R (Z - 1) × ç 2 - 2 ÷ × s çn ÷ l è f ni ø 1 2 where R is the Rydberg constant (1.097·107 m-1), Z is the atomic number, n is the main quantum number in the final (f) and initial (i) states and σ is a constant (≈ 1). initial final As the transitions responsible for the X-rays occur in the inner shell, the atomic number (thus the electric charge) of the nucleus (Z) should have strong influence on the spectroscopic terms. This is why Z appears in the expression of the Röntgen lines (Moseley’s law) but not in those of the optical spectral lines (Balmer’s expression). λmin Problem You are operating an X-ray tube with a chromium (Cr) target by applying an acceleration potential of 60 kV. Draw a schematic of the x-ray spectrum emitted by this tube and label on it the characteristic wavelengths of the Kα line of the characteristic radiation and minimum wavelength of the Bremsstrahlung (λmin). Solution The characteristic X-ray spectrum of Cr will show the characteristic Kβ and Kα lines on top of the continuous spectrum or Bremsstrahlung. We may quantify the wavelengths of Kα and (λmin). 24Cr has Z = 24 atomic number: The wavelength of the K line can be calculated from α the Moseley’s law: λKα = c/ν = (3·108 m/s)/[2.47· 1015 (Z-1)2 ·1/s] = 230 pm. The shortest wavelength limit of the Bremsstrahlung can be calculated from the DuaneHunt displacement law: λmin = hc/(eV) = 20.7 pm. X-ray „fluorescence” (XRF) Georg von Hevesy applied X-rays to excite characteristic X-rays from a sample and to use diffraction to identify its constituent elements. This is termed X-ray fluorescence. Later he introduced a method of activation analysis based on neutron bombardment (neutron activation); this method yields better detection limits. He received the 1943 Nobel Prize in Chemistry for his work on the use of isotopes as tracers in studying chemical processes. Hevesy was the first to apply the radioactive tracer technique to biology, and he later used it in medical research. Typical wavelength dispersive XRF spectrum; note the systematic increase in atomic number toward the left. Diffraction of the X-rays WAVE + CRYSTAL ↔ DIFFRACTION Passage of waves of light through crystalline arrangement of atoms (crystals) should cause diffraction or interference phenomena. It can be reversed: if the radiation shows diffraction pattern in crystal, then the radiation should consist of wave and not of particle. Von Laue took diffraction as positive proof that X-rays are electromagnetic radiation (waves), not particles. Each dot, called a reflection, in this diffraction pattern forms from the constructive interference of scattered X-rays passing through a crystal. The data can be used to determine the crystalline structure. Bragg's law of diffraction and the structure of NaCl The pattern of spots in the Laue diffractogram could be explained by reflection of waves from crystal planes. L. W. Bragg studied the diffraction of Pt-Lα X-rays by NaCl crystal. He concluded that salt consisted of a three dimensional lattice of Na+ and Clions (At that time chemists refused believe that NaCl contains no NaCl molecules, just alternating array of Na+ and Cl- ions!) For a family of parallel crystal planes with spacing d, a monochromatic incident beam of wavelength λ will appear to be reflected by the planes if the incident angle Θ obeys the Bragg’s law: 2 d sin Q = n × l n = 0,1,2,... The extra path length traveled by the ray scattered by the top layer relative to the next layer down is 2d·sinΘ Applicaton of the Bragg’s law to crystals Reflection planes (shaded area) in simple crystals Diatomic crystal: simple cubic latice with two scattering elements at each point Bragg’s reflection at a set of planes from the cubic lattice in a well defined direction X-ray crystallography The phase problem: the intensities of the spots can be measured (and the amplitudes can be calculated) but the phase differences are lost. Methods to get back partly this information: Electron density map 1) Fourier-transfer; Fourier-refinement, 2) Multiple isomorphous replacement (heavy atom replacement at specific loci), 3) Comparison with known structures of similar proteins (biomolecules) Goniometer 3D structure Diffraction patter reaction center protein from photosynthetic bacteria; ~100 kDa acidic, basic, histidine. X-ray crystallography: what the DNA looks like at the atomic level? In the early 1950s, James Watson and Francis Crick (Cambridge University) proposed the double-helix structure of (B-)DNA, which has been called the most important biological work of the past century. The unusual DNA structures (called a Holliday junctions) play a key role in DNA's ability to repair itself – a vital biological function that can be applied in biomedicine and other related fields. It is fundamentally important in explaining the actual biological function of genes - in particular, such issues as genetic "expression," DNA mutation and repair, and why some DNA structures are inherently prone to damage and mutation. Understanding DNA structure is just as necessary as knowing gene sequence. The human genome project (detailed explanation of the genetic sequence of the entire human genome), is one side of the coin. The other side is understanding how the three-dimensional structure of different types of DNA are defined by those sequences, and, ultimately, how that defines biological function. PIXE: Particle-induced X-ray Emissions (particle) A bombardment from an alpha source can be used to stimulate atomic x-ray transitions in a number of materials. The x-ray energies reveal the atomic level spacings with great clarity, and can be used to identify the atoms. The characteristic x-ray lines of the elements are utilized to determine the composition of unknown samples. Attenuation (absorption) of X-radiation: the Beer’s law The absorption of X-ray radiation is described by the general exponential law of radiation attenuation (Beer’s law): I = I 0 × e- m x where I0 is the intensity of the rays that are incident perpendicularly on the absorbent, which is a homogeneous layer of thickness x, I is the intensity of the rays after passing through the layer and μ is the attenuation coefficient, which includes all the significant properties of the absorbent (e.g. tissues in medical applications) and the interaction between the absorbent and the radiation. I0 0 ln I/I0 μ slope: -μ x x Half-value thickness. This is that thickness of the material which reduces the intensity of a narrow homogeneous beam to half its original value: xH = (ln 2)/μ Mass attenuation coefficient: μm = μ/ρ is the attenuation per unit mass of material traversed (ρ is the density). I dI(x)=μ·I(x)dx Mass attenuation coefficient μ/ρ Energy of X-ray quantum E (MeV) (cm2/g) air water fat muscle bone Z = 7,78 ρ = 0,0012 Z = 7,51 ρ = 0,9982 Z = 6,46 ρ = 0,92 Z = 7,64 ρ = 1,04 Z = 12,31 ρ = 1,65 0,01 5,12 5,329 3,268 5,356 28,51 0,1 0,1541 0,1707 0,1688 0,169 0,186 1 0,06358 0,07072 0,0708 0,0701 0,0657 10 0,02045 0,02219 0,0214 0,0219 0,0231 20 0,01705 0,01813 0,017 0,0179 0,0207 Energy-dependence of the attenuation coefficient of X-ray in water I (x) = I(0)·exp(-μ·x) Classic scattering sk Linear attenuation coefficient water Photoeffect t Compton-effect sC Pair production k Energy of the X-ray quantum The X-ray quanta are scattering on the electrons elastically (without energy loss). External photoeffect: the ionizing radiation liberates an electron from the shell. Compton-effect: scattering of photons (X-ray quanta) on free or loosley bound electrons. The high energy (hν >1,02 MeV) photon (X-ray quantum) produces an electron and a positron with opposite direction of speeds in the close vicinity of the electric (Coulomb) field of the nucleus of the absorbing material. Classic scattering Photoelectric absorption (photoeffect) Compton-scattering Pair production Mechanisms of attenuation Comparison of the different attenuation mechanisms of radiation μ: attenuation coefficient, E: energy of the quantum, Z: atomic number of the chemical element of the material Range of energy in soft tissues mechanisms Coherent scattering Photoelectric absorption independent Compton-effect Pair production Ionization processes induced by photons of radiation in matter Matter Survey of different posible interactions when photon enters the matter. Wavy lines = Paths of photons : photoeffect Pair production Comptonscattering Harringbones indicate the paths of the ion pairs produced by the radiation in the matter. Gamma- or X-rays Triplet production The density of the herringbones offers the frequency (density) of the ionization process. : Pair annihilation Straight lines = paths of electrons and positrons Problem. X-rays of the same initial intensity I0 pass through soft tissue x1 = 18 cm thick and through the same soft tissue containing a bone x2 = 4 cm thick. The attenuation coefficients are μ1 = 0.19 cm-1 for the soft tissue and μ2 = 0.42 cm-1 for the bone. What is the intensity ratio of the emerging rays? I1 I2 What is the contrast of the bone? x2 Solution. The ratio of intensities I1 and I2 is x1 soft tissue I0 bone I0 I1 I 0 × exp(- m1 x1 ) = = exp(- ( m1 - m 2 ) x2 ) = 2.5 I 2 I 0 × exp(- m1 ( x1 - x2 ) - m 2 x2 ) In radiography, the relative X-ray intensity difference between the background tissue and the anatomic structure of interest is referred to as the subject contrast (also known as tissue contrast): CS = I bgd - IS I bgd where IS and Ibgd are the intensity of X-rays passing through the anatomic structure of interest and the adjacent background tissue, respectively. If I2 is substituted for IS and I1 for Ibgd, then the contrast of the bone in the present problem is CS = 0.6. Problems for seminar 1. Determine the wavelength of Kα and λmin for molybdenum (Mo). (Z = 42; for Kα line ni = 2 and nf =1; σ =1) 2. Identify the element giving rise to Kα with λKα = 251 pm. (Solution: Z = 23, Vanadium) 3. The anode of an X–ray tube is made of Tungsten (atomic number 74). What are the frequencies of the characteristic Kα (ni = 2) and Kβ (ni = 3) radiations? 4. For radiotherapic purposes, electrons are used which are accelerated by betatron to 25 -45 MeV energy. What will be the angular divergence of the X-ray produced by target metal hit by these electrons at the outlet of the betatron? 5. What is the maximum frequency of the X-ray quantum in a 10 kV X-ray tube if the kinetic energy of the electron hitting the anode would convert completely to radiation? Problems for seminar 6. The anode voltage and current in an X-ray tube are set to 80 kV and 6 mA, respectively. The anode material is tungsten. a) What is the maximum energy of the X-ray quanta? b) What is the short-wavelength limit of the X-ray spectrum? c) What is the radiative power of the X-rays produced in this tube? d) What is the efficiency of X-ray production? e) How much heat is generated per minute? f) What is the velocity of electrons at the anode? g) How many electrons hit the anode per second? 7. The anode voltage of an X-ray tube is supplied by a charged capacitor of high capacitance C = 1 μF. What is the percentage decrease of the initial accelerating voltage of V = 100 kV after an exposure current and time of 5 mA and 2 s, respectively? Why is it important to maintain constant anode voltage during exposure? Problems for seminar 8. X-rays for a chest examination are produced by an X-ray tube at a voltage of V = 80 kV and a current of I = 5 mA with an efficiency of η = 0.65%. a) What is the intensity I0 of X-rays at a distance r = 1 m from the focal point of the tube, if they distribute uniformly in a solid angle 2π (i.e. in a hemisphere)? b) What is the dose absorbed in the chest r = 1 m distant from the tube during transillumination for t = 10 s? The chest is assumed to have a uniform thickness of x = 10 cm. The average attenuation coefficient is μ = 0.18 cm-1, and the average density is ρ = 1.05 g·cm-3. 9. What is the subject contrast given by the blood in a soft tissue environment? The radius of the aorta is x = 1 cm, and the attenuation coefficients of the blood and the surrounding soft tissue are 0.215 cm-1 and 0.211 cm-1, respectively. How much is the contrast increased if iodine is injected into the blood circulation and the attenuation coefficient of the blood is increased to 0.284 cm-1? 10. The lattice of NaCl is cubic with a unit-cell dimension of 5.64 Å. Each unit cell contains 4 Na+ and 4 Cl- ions. The density of NaCl is 2.163 g·cm-3. Calculate the Avogadro’s number using this information. Problems for seminar 11. For λ = 154 pm X-rays, obtain the first order (n = 1) Bragg angles for interplanar spacings of 500 pm, 1 nm, and 100 nm. 12. From the Bragg’s equation of diffraction it can be seen that for a fixed λ, the interplanar spacing d is inversely proportional to sin Θ. In other words, scattering corresponding to the smallest spacing occurs at the maximum value of sin Θ (when Θ = 90o). If λ = 154 pm X-rays are used, what is the theoretical limit of resolution (i.e. what is the minimum spacing observable)?
 0
0
advertisement
Related documents
Download
advertisement
Add this document to collection(s)
You can add this document to your study collection(s)
Sign in Available only to authorized usersAdd this document to saved
You can add this document to your saved list
Sign in Available only to authorized users