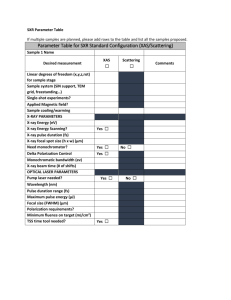
SAXS data reduction and processing
advertisement

Data reduction and processing Al Kikhney Outline • • • • • • • SAXS experiment setup 3D → 2D → 1D Background subtraction High vs. low concentration Rg, MM Volume Distance distribution function p(r) Small Angle X-ray Scattering solution Synchrotron Radiation → 2θ Homogeneous and Monodisperse solution s X-ray detector solvent |s| = 4π sinθ/λ 1-2 mg purified material concentration from 0.5 mg/ml, exposure times: a few seconds/minutes s – scattering vector 2θ – scattering angle λ – wavelength I(s) – intensity Small Angle X-ray Scattering Exposure beamstop Beam lower angle Beam higher angle X-ray detector Small Angle X-ray Scattering Exposure beamstop Beam X-ray detector X-ray detector Small Angle X-ray Scattering Exposure X-ray detector Small Angle X-ray Scattering Radial averaging Log I(s), a.u. Normalization against: • data collection time, • transmitted sample intensity s, nm-1 Small Angle X-ray Scattering Notations and units Log I(q), a.u. |q| = 4π sinθ/λ 2θ – scattering angle λ – wavelength q, nm-1 Small Angle X-ray Scattering Notations and units Log I(s), a.u. |s| = 4π sinθ/λ 2θ – scattering angle λ – wavelength 1 2 3 s, nm-1 Small Angle X-ray Scattering Notations and units Log I(s), a.u. |s| = 4π sinθ/λ 2θ – scattering angle λ – wavelength 0.1 0.2 0.3 s, Å-1 Small Angle X-ray Scattering Notations and units Log I(s), a.u. |s| = 2 sinθ/λ 2θ – scattering angle λ – wavelength s, nm-1 Small Angle X-ray Scattering Notations and units Log I(s), a.u. |s| = 4π sinθ/λ 2θ – scattering angle λ – wavelength s, nm-1 Data quality Radiation damage Log I(s), a.u. sample same sample again RADIATION DAMAGE! s, nm-1 Data quality Log I(s), a.u. sample same sample again average s, nm-1 I(s), a.u. Buffer and sample Solution and Solvent s, nm-1 I(s), a.u. Buffer and sample Solution and Solvent (zoom in) s, nm-1 Looking for protein signals less than 5% above background level… Background subtraction Solution minus Solvent Log I(s), a.u. Log I(s), a.u. sample buffer sample – buffer (subtracted) Normalization against: • concentration s, nm-1 s, nm-1 Data quality “Can I use this data for further analysis?” Log I(s) Log I(s) lysozyme AGGREGATED! s, nm-1 s, nm-1 Dilution series Log I(s) Low and High Concentration LytA protein, 60 kDa , 1 mg/ml 10 mg/ml s, nm-1 Dilution series Log I(s) Low and High Concentration s, nm-1 Merging data Log I(s) Low and High Concentration s, nm-1 Merging data Log I(s) Low and High Concentration s, nm-1 Merging data Log I(s) Low and High Concentration s, nm-1 Data analysis Shape lg I(s), relative 0 Long rod -1 -2 -3 Solid sphere -4 -5 -6 0.0 0.1 0.2 0.3 0.4 s, s nm-1 Hollow sphere Dumbbell 0.5 Flat disc Size Log I(s) s, Å-1 Shape and size Log I(s) a.u. lysozyme apoferritin s, nm-1 Crystal vs. solution Data range 2.00 1.00 0.67 0.50 0.33 Resolution, nm Log I(s) 8 Atomic structure 7 Shape 6 Size 5 Fold 0 5 10 15 s, nm-1 © Dmitri Svergun Radius of gyration (Rg) Definition Measure for the overall size of a macromolecule Average of square center-of-mass distances in the molecule weighted by the scattering length density Log I(s) Radius of gyration (Rg) Guinier plot s Radius of gyration (Rg) Guinier plot Log I(s) Ln I(s) s2 Normal Log plot s Radius of gyration (Rg) Guinier plot Ln I(s) y = ax + b Rg = sqrt(-3a) • Estimate of the overall size of the particles Guinier approximation: I(s) = I(0)exp(-s2Rg2/3) sRg≲1.3 s2 André Guinier 1911-2000 Radius of gyration (Rg) Guinier plot Ln I(s) y = ax + b Rg = sqrt(-3a) • Estimate of the overall size of the particles Guinier approximation: I(s) = I(0)exp(-s2Rg2/3) sRg≲1.3 • Quality of the data – aggregation – polydispersity – improper background substraction • Zero angle intensity I(0) • First point to use s2 Radius of gyration (Rg) Log I(s) Bovine serum albumin (BSA) s, 1/nm Radius of gyration (Rg) Guinier plot Ln I(s) s2 Radius of gyration (Rg) Guinier plot Ln I(s) Rg ± stdev Ln I(0) Forward scattering I(0) Data quality Data range y = ax + b Rg = sqrt(-3a) s2 Log I(s), a.u. Log I(0)apo Molecular mass Guinier approximation Log I(0)lys lysozyme apoferritin s, nm-1 I(0) and Molecular Mass MMsample I(0)sample = I(0) MMBSA BSA MMsample = I(0) sample* MMBSA / I(0)BSA BSA Rg = 3.1 nm I(0) = 11.7 MMBSA = 66 kDa Rg = 1.46 nm I(0) = 3.66 MM = 20.6 kDa Rg = 6.81 nm I(0) = 79.45 MM = 448.2 kDa Porod law I(s) ~ -4 s Intensity decay is proportional to s-4 at higher angles (for globular particles of uniform density) Porod law I(s)*s4 s Porod law Excluded volume of the hydrated particle VP = 2π 2 I (0) ∞ 2 [ I ( s ) − K ] s ds 4 ∫ 0 K4 is a constant determined to ensure the asymptotical intensity decay proportional to s-4 at higher angles following the Porod's law for homogeneous particles Primus Porod plot Excluded volume of the hydrated particle I(s)*s4 I(s)*s4 14 nm3 ~9 kDa 974 nm3 s s ~610 kDa Sasplot Kratky plot Patterns of globular and flexible proteins I(s) s2 Natively unfolded Globular Multidomain with flexible linkers © Stratos Mylonas γ(r) Distance distribution function r, nm γ(r) Distance distribution function p(r) = r2 γ(r) r, nm Distance distribution function p(r) r, nm p(r) function Distance distribution function p(r) function Distance distribution function ( s ) = 4π II(s) Dmax ∫ 0 sin( sr ) dr (r ) pp(r) sr 2 r p r = ( ) p(r) 2π 2 Indirect Fourier Transform ∞ s 2 I(s) I ( s ) sin( sr ) ds ∫0 sr Gnom p(r) plot Distance distribution function Dmax Dmax p(r) p(r) r, nm r, nm Data quality Dmax smin ≤ π/Dmax I(s) p(r) r, nm smin s, 1/nm Yeast bleomycin hydrolase 3GCB Compact dimer 50 kDa Monomer Hexamer Extended dimer p(r) plots 50 kDa Monomer Compact dimer Extended dimer Bad Yeast bleomycin hydrolase 3GCB Hexamer Good Summary • • • • • Exposure 3D → 2D Radial averaging → 1D Log I(s) Normalization • Log plot s Background subtraction Analysis • Guinier plot (Rg, MM) • Porod plot s2 I(s)*s4 • Kratky plot (flexibility) • p(r) plot Ln I(s) p(r) s I(s)*s2 s r Thank you! After questions: group photo! www.saxier.org/forum