Standard Deviation
advertisement

Absolute Error
The difference between the true value and the measured value is referred to as the absolute
error. Assume that analysis of an iron ore by some method gave 11.1% while the true value
was 12.1%, the absolute error is:
11.1% - 12.1% = -1.0%
Relative Error
The relative error is the percentage of the absolute error to the true value. For the argument
above we can calculate the relative error as:
Relative error = (absolute error/true value)x100%
= (-1.0/12.1)x100% = -8.3%
Relative Accuracy
The percentage of the quotient of observed result to the true value is called relative
accuracy.
Relative accuracy = (observed value/true value)x100%
For the abovementioned example:
Relative accuracy = (11.1/12.1)x100% = 91.7%
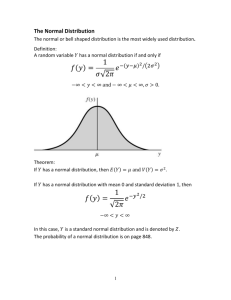
Standard Deviation
Standard deviation for a set of data provides information on the spread of the values.
A
B
What we mean by spread is clear from the graphs A and B. Values used to draw A are not
as close to each others as values in graph B. Therefore, values in A have higher spread from
the mean and have higher standard deviation. On the contrary, values in B are close
together and have a low spread from their mean, and thus have lower standard deviation.
For an infinite or large number of data points (more than 20) or when the true mean is
known, the population standard deviation is defined as:
σ = ( Σ (xi - µ )2 / N )1/2
Where σ is the population standard deviation, µ is the population mean, xi is the individual
data point, and N is the number of data points.
However, in real chemical laboratories where a sample is analyzed, an experiment is
repeated three to five times and thus a very limited data points (3-5) is collected. The
sample standard deviation (s) is defined as:
s = ( Σ (xi - x)2 / N-1 )1/2
x is the average (mean) of the data points. The sample standard deviation is also called
estimated standard deviation since it is only an estimate of σ .
Also, one can define a new term that is the standard deviation of the mean (s(mean))
S(mean) = s / N1/2
Another important expression of deviation is the relative standard deviation (RSD) or
sometimes called coefficient of variation (CV) where
RSD = ( s / x ) X100%
RSD (mean) = ( s(mean) / x ) X100%
Example
The following replicate weights were obtained for a sample: 29.8, 30.2, 28.6, and 29.7 mg.
Calculate s, s(mean), RSD, and RSD(mean)
Solution
First, we find x
X = (29.8+30.2+28.6+29.7)/4 = 29.6
xi
29.8
30.2
28.6
29.7
xi – x
0.2
0.6
1.0
0.1
Σ
s = ( Σ (xi
- x)2 /
N-1 )1/2
s = (1.41/3)1/2
s = 0.69 mg
S(mean) = s / N1/2
(xi – x)2
0.04
0.36
1.00
0.01
1.41
S(mean) = 0.69/(4)1/2
S(mean) = 0.34 mg
RSD = (0.69/29.6)x100% = 2.3%
RSD is same as CV
RSD(mean) = (0.34/29.6)x100% = 1.1%
It should be recognized that as the number of experiments is increased, the precision of the
measurement is increased as well. This means smaller s due to smaller spread. This is because
S α 1/N 1/2 which means that decrease in s as N increases is not linear which implicates that,
after some number of experiments, further increase in the number of experiments will result in
very little decrease in s (try it on your calculator), which does not justify extra time and effort.
Pooled Standard Deviation (sp)
When the same experiments are done using two different methods, the standard deviation can
be pooled in order to determine the reliability of the analysis method (proposed or new).
Sp = { Σ (xi1 – x1 )2 + Σ (xi2 – x2 )2 )/ (N 1 + N2 –2)}1/2
Sp is the pooled standard deviation, x1 , x2 are average values for data set 1 and 2,
respectively, N1 , N2 are the number of data points of data set 1 and 2, respectively.
Example
Mercury in a sample was determined using a standard method and a new suggested
method. Six replicate experiments were conducted using the two procedures giving the
following results in ppm
New Method
10.5
9.9
10.4
11.2
10.5
Standard method
10.1
10.3
10.2
10.3
10.4
Find the pooled standard deviation
Solution
First find x1 and x2
X1 = (10.5+9.9+10.4+11.2+10.5)/5 = 10.5
X2 = (10.1+10.3+10.2+10.3+10.4)/5 = 10.3
Σ(Xi1-X1)2 = { (10.5-10.5)2 + (9.9-10.5)2 +(10.4-10.5)2 + (11.2-10.5)2 + (10.5-10.5)2}
Σ(Xi1-X1)2 = 0.86
Σ(Xi2-X2)2 = { (10.1-10.3)2 + (10.3-10.3)2 + (10.2-10.3)2 + (10.3-10.3)2 +(10.4-10.3)2}
Σ(Xi2-X2)2 = 0.06
Sp 2 = { Σ (xi1 – x1 )2 + (xi2 – x2 )2 )/ (N1 + N2 –2) }
SP = {(0.86+0.06)/(5+5-2)}1/2 = 0.34
Standard Deviation of the Difference
When multiple samples are analyzed by a proposed and standard methods, Sd is the
calculated standard deviation for the difference.
Sd = ( Σ ( Di – D )2 / N-1 )1/2
(7)
Sd is the standard deviation of the difference, Di is the difference between a result obtained
by the standard method from that obtained by the proposed method for the same sample. D
is the average of all differences.
Example
Mercury in multiple samples was determined using a standard method and a new suggested
method. Six different samples were analyzed using the two procedures giving the following
results in ppm
New Method
10.2
12.7
8.6
17.5
11.2
11.5
Standard method
10.5
11.9
8.7
16.9
10.9
11.1
Find the standard deviation of the difference.
Solution
It is wise to construct a table as below
New Method
10.3
12.7
8.6
17.5
Standard method Di
10.5
-0.2
11.9
+0.8
8.7
-0.1
16.9
+0.6
11.2
10.9
+0.3
11.5
11.1
+0.4
_____________________________________
Σ Di = 1.8
D = 1.8/6 = 0.30
Σ ( Di – D )2 = { (-0.2-0.3)2 + (+0.8-0.3)2 + (-0.1-0.3)2 + (+0.6-0.3)2 + (+0.3-0.3)2 +
(+0.4-0.3)2 } = {0.25+0.25+0.16+0.09+0+0.01}
Σ ( Di – D )2 = 0.76
Sd = ( Σ ( Di – D )2 / N-1 )1/2
Sd = (0.76/5)1/2 = 0.39