Bacteriological Examination of Water
advertisement

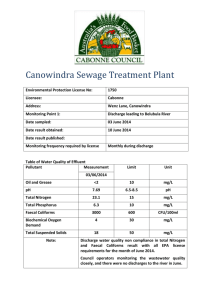
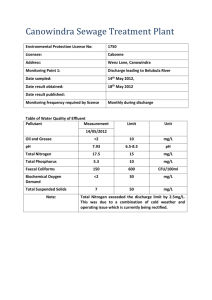
Prins Leopold Instituut voor Tropische Geneeskunde Institut de Médecine Tropicale Prince Léopold Prince Leopold Institute of Tropical Medicine Instituto de Medicina Tropical Principe Leopoldo Nationalestraat, 155 B – 2000 Antwerpen Stichting van Openbaar Nut 0410.057.701 POSTGRADUATE IN TROPICAL MEDICINE AND INTERNATIONAL HEALTH MODULE 2 CLINICAL & BIOMEDICAL SCIENCES OF TROPICAL DISEASES Practical notes __________________________ WATER ANALYSIS PHILIPPE GILLET, BIRGIT DE SMET, JAN JACOBS, JANUARY 2009 Water analysis Practical 1 What to do: In groups of 2 persons: - - Prepare the sampling material needed (1 sampling bottle per participants). Prepare the medium needed for the shorter method based on Lauryl Sulphate Broth, MPN, 3 x 3 tubes. (One group will prepare tubes with single strength broth, 12 tubes per participants + reserve; the other group will prepare tubes with double strength, 6 tubes per participants + reserve). Sterilise the material and the medium. Prepare the incubators for the water analyses. Realise the quality control of the medium. What to remember: - Factors to consider for the sterilisation. Quality controls to consider. Use and interpretation of quality controls. Principe of sampling for water analyses. Practical 1I What to do: In groups of 2 persons: - Interpret the controls. Realise the turbidity determination of 2 water samples. Realise the determination of chlorine residual in 2 water samples. Interpret the results. Individually: - - Realisation of the shorter method based on Lauryl Sulphate Broth, MPN, 3 x 3 tubes on 2 different water samples, Total Coliforms + Thermotolerant Coliforms. Interpret the results. What to remember: - Principe of water analysis (turbidity, chlorine residual and microbiological examination of water). Factors to consider for the microbiological examination of water. Tables of contents Tables of contents ..........................................................................................................3 Water analysis ................................................................................................................4 MICROBIOLOGICAL EXAMINATION OF WATER ...........................................4 Indicator organisms (summary): ............................................................................4 WATER SAMPLING................................................................................................6 A Sampling from a tap or pump outlet .................................................................7 B Sampling from reservoir....................................................................................7 C Sampling from a dug well .................................................................................7 METHODS OF MICROBIOLOGICAL ANALYSIS...............................................7 “SHORTER METHOD” BASED ON LAURYL SULPHATE BROTH: ................9 TURBIDITY MEASURMENT:..............................................................................13 RESIDUAL FREE CHLORINE TEST ...................................................................14 Target values:.......................................................................................................15 ANNEX 1 : LAURYL SULFATE BROTH, MSDS ............................................16 ANNEX 2 : SODIUM THIOSULFATE PENTAHYDRATE, MSDS ...............20 09 01 29_PG_Bacteriological Examination of Water 3 / 24 Water analysis Water, of adequate quantity and quality, is essential for healthy life. The associations between sanitation, water and health are well known. Many diseases are associated with contaminated water and water shortages. The most important factor to take into account is that, in most communities, the principal risk to human health derives from faecal contamination. In some countries there may also be hazards associated with specific chemical contaminants such as fluoride or arsenic, but the levels of these substances are unlikely to change significantly with time. Thus, if a full range of chemical analyses is undertaken on new water sources and repeated thereafter at fairly long intervals, chemical contaminants are unlikely to present an unrecognized hazard. In contrast, the potential for faecal contamination in untreated or inadequately treated community supplies is always present. The minimum level of analysis should therefore include testing for indicators of faecal pollution (thermotolerant (faecal) coliforms), turbidity, and chlorine residual and pH (if the water is disinfected with chlorine). MICROBIOLOGICAL EXAMINATION OF WATER Microbiological examination offers the most sensitive test for the detection of recent and potentially dangerous faecal pollution, thereby providing a hygienic assessment of water quality with high sensitivity and specificity. For this reason it is important to examine a drinking water source frequently by a simple test rather than infrequently by a more complicated test or series of tests. It is ideal to look for individual specific pathogen but it is not practical since they are few in numbers than the non-pathogenic organisms and methods to detect them are costly in time and money. Therefore indicators of human/animal pollution e.g. coliforms are used. Faecal streptococci are regularly present in the faeces in varying numbers but their number is fewer than Escherichia coli and they probably die and disappear at the same rate. The presence of faecal streptococci along with coliforms in absence of Escherichia coli is also confirmatory of faecal pollution. Indicator organisms (summary): Escherichia coli is a member of the family Enterobacteriaceae, and is characterized by possession of the enzymes b-galactosidase and b-glucuronidase. It grows at 44–45°C on complex media, ferments lactose and mannitol with the production of acid and gas, and produces indole from tryptophan. However, some strains can grow at 37 °C but not at 44– 45°C, and some do not produce gas. E. coli does not produce oxidase or hydrolyse urea. Complete identification of the organism is too complicated for routine use, but a number of tests have been developed for rapid and reliable identification. Some of these methods have been standardized at international and national levels and accepted for routine use; others are still being developed or evaluated. Escherichia coli is abundant in human and animal faeces; in fresh faeces it may attain 9 concentrations of 10 per gram. It is found in sewage, treated effluents, and all natural waters and soils subject to recent faecal contamination, whether from humans, wild animals, or agricultural activity. Recently, it has been suggested that E. coli may be present or even multiply in tropical waters not subject to human faecal pollution. However, even in the remotest regions, faecal contamination by wild animals, including birds, can never be excluded. Because animals can transmit pathogens that are infective in humans, the presence of E. coli or thermotolerant coliform bacteria must not be ignored, because the 09 01 29_PG_Bacteriological Examination of Water 4 / 24 presumption remains that the water has been faecally contaminated and that treatment has been ineffective. Thermotolerant coliform bacteria: Thermotolerant coliform bacteria are the coliform organisms that are able to ferment lactose at 44–45°C; the group includes the genus Escherichia and some species of Klebsiella, Enterobacter, and Citrobacter. Thermotolerant coliforms other than E. coli may also originate from organically enriched water such as industrial effluents or from decaying plant materials and soils. For this reason, the term “faecal” coliforms, although frequently employed, is not correct, and its use should be discontinued. Regrowth of thermotolerant coliform organisms in the distribution system is unlikely unless sufficient bacterial nutrients are present, unsuitable materials are in contact with the treated water, the water temperature is above 13 °C, and there is no free residual chlorine. In most circumstances, concentrations of thermotolerant coliforms are directly related to that of E. coli. Their use in assessing water quality is therefore considered acceptable for routine purposes, but the limitations with regard to specificity should always be borne in mind when the data are interpreted. If high counts of thermotolerant coliforms are found in the absence of detectable sanitary hazards, additional confirmatory tests specific for E. coli should be carried out. National reference laboratories developing national standard methods are advised to examine the specificity of the thermotolerant coliform test for E. coli under local conditions. Because thermotolerant coliform organisms are readily detected, they have an important secondary role as indicators of the efficiency of water-treatment processes in removing faecal bacteria. They may therefore be used in assessing the degree of treatment necessary for waters of different quality and for defining performance targets for removal of bacteria. Coliform organisms (total coliforms): Coliform organisms have long been recognized as a suitable microbial indicator of drinking-water quality, largely because they are easy to detect and enumerate in water. The term “coliform organisms” refers to Gram-negative, rod-shaped bacteria capable of growth in the presence of bile salts or other surface-active agents with similar growth-inhibiting properties and able to ferment lactose at 35–37°C with the production of acid, gas, and aldehyde within 24–48 hours. They are also oxidase-negative and nonspore-forming and display b-galactosidase activity. Traditionally, coliform bacteria were regarded as belonging to the genera Escherichia, Citrobacter, Enterobacter, and Klebsiella. However, as defined by modern taxonomical methods, the group is heterogeneous. It includes lactosefermenting bacteria, such as Enterobacter cloacae and Citrobacter freundii, which can be found in both faeces and the environment (nutrient-rich waters, soil, decaying plant material) as well as in drinking-water containing relatively high concentrations of nutrients, as well as species that are rarely, if ever, found in faeces and may multiply in relatively good-quality drinking-water, e.g. Serratia fonticola, Rabnella aquatilis, and Buttiauxella agrestis. The existence both of non-faecal bacteria that fit the definitions of coliform bacteria and of lactose-negative coliform bacteria limits the applicability of this group as an indicator of faecal pollution. Coliform bacteria should not be detectable in treated water supplies and, if found, suggest inadequate treatment, post treatment contamination, or excessive nutrients. The coliform test can therefore be used as an indicator both of treatment efficiency and of the integrity of the distribution system. Although coliform organisms may not always be directly related to the presence of faecal contamination or pathogens in drinkingwater, the coliform test is still useful for monitoring the microbial quality of treated piped water supplies. If there is any doubt, especially when coliform organisms are found in the absence of thermotolerant coliforms and E. coli, identification to the species level or analyses for other indicator organisms may be undertaken to investigate the nature of the contamination. Sanitary inspections will also be needed. Faecal streptococci: Faecal streptococci are those streptococci generally present in the faeces of humans and animals. All possess the Lancefield group D antigen. Taxonomically, they belong to the genera Enterococcus and Streptococcus. The taxonomy of Enterococci has recently undergone important changes, and detailed knowledge of the ecology of many of the new species is lacking; the genus Enterococcus now includes all streptococci that share certain biochemical properties and have a wide tolerance of adverse growth conditions—E. avium, E. casseliflavus, E. cecorum, E. durans, E. faecalis, E. faecium, E. gallinarum, E. 09 01 29_PG_Bacteriological Examination of Water 5 / 24 hirae, E. malodoratus, E. mundtii, and E. solitarius. Most of these species are of faecal origin and can generally be regarded as specific indicators of human faecal pollution for most practical purposes. They may, however, be isolated from the faeces of animals, and certain species and subspecies, such as E. casseliflavus, E. faecalis var. liquefaciens, E. malodoratus, and E. solitarius, occur primarily on plant material. In the genus Streptococcus, only S. bovis and S. equinus possess the group D antigen and therefore belong to the faecal streptococcus group. They derive mainly from animal faeces. Faecal streptococci rarely multiply in polluted water, and they are more persistent than E. coli and coliform bacteria. Their primary value in water-quality examination is therefore as additional indicators of treatment efficiency. Moreover, streptococci are highly resistant to drying and may be valuable for routine control after new mains are laid or distribution systems are repaired, or for detecting pollution of ground waters or surface waters by surface run-off. WATER SAMPLING Several type of bottle may be used for sampling, but glass bottles are best. These should have securely fitting stoppers with non toxic liners. The bottles should hold at least 200 ml of water and should be sterilized. When water that contains or may contain even traces of chlorine is sampled, the chlorine must be inactivated. If it is not, microbes may be killed during transit and an erroneous result will be obtained. The bottles in which the samples are placed should therefore contain sodium thiosulfate to neutralize any chlorine present. The sodium thiosulfate should be added to the bottles before they are sterilized. For 200 ml samples, five drops of aqueous sodium thiosulfate solution [100 g/litter (w/v)] should be added to each clean sample bottle. The stopper is loosely adapted to the bottle and aluminium foil cover is tied to the neck of the bottle to prevent dust from entering. The bottle is the sterilized [in hot-air oven for 1 hour at 160°C or for 40 minutes at 170°C; or in an autoclave at 121°C for 20 minutes. Although recommendations vary, the time between sample collection and analysis should, in general, not exceed 6 hours, and 24 hours is considered the absolute maximum. . It is assumed that the samples are immediately placed in a lightproof insulated box containing melting ice or ice-packs with water to ensure rapid cooling. If ice is not available, the transportation time must not exceed 2 hours. It is imperative that samples are kept in the dark and that cooling is rapid. If these conditions are not met, the samples should be discarded. The cool box used to carry samples should be cleaned and disinfected after each use to avoid contaminating the surfaces of the bottles and the sampler’s hands. Sources of water to be sampled Water sources can be divided into three basic types for the purpose of sampling. a) b) c) Water from a tap or fixed hand pump Water from a reservoir (river, lake, tank, …) Water from a dug well 09 01 29_PG_Bacteriological Examination of Water 6 / 24 A Sampling from a tap or pump outlet 1. 2. 3. 4. Remove any attachments from tap that may cause splashing. Wipe off the dirt from outside the tap. Turn on the tap at maximum flow rate and let the water flow for 1-2 minutes. Sterilize it for a minute with flame using gas burner, lighter or ignited cotton wool soaked in spirit. 5. Open the tap and allow water to flow at medium rate for 1-2 minutes. 6. Open the container for collecting the sample and fill the water by holding the bottle under the water jet. Leave a small airspace to facilitate shaking at the time of inoculation prior to analysis. 7. Stopper the cap and label the container. B Sampling from reservoir 1. Submerge the bottle in the water. 2. Open the bottle inside of the water. 3. Fill it by holding it by the lower part, submerging it to a depth of about 30 centimetres, with the mouth facing slightly upwards. If there is a current, the bottle should face the current. 4. Pull it out when the bottle is filled. 5. Discard a little water to provide airspace 6. Stopper the bottle and label it. C Sampling from a dug well 1. 2. 3. 4. Attach a stone of suitable size to the sampling bottle with a piece of string. Tie a 20 meter length of clean string on the bottle and to a stick. Open the bottle as described above and lower into the well. Immerse the bottle completely in water without touching the sides of the well and lower it down to the bottom of the well. 5. Pull it out when the bottle is filled. 6. Discard a little water to provide airspace. 7. Stopper and label the bottle. METHODS OF MICROBIOLOGICAL ANALYSIS Two methods have been developed for the detection of indicator bacteria in water: membrane filter method and multiple tube method. 1. Membrane filtration method: In the membrane-filtration (MF) method, a minimum volume of 10 ml of the sample (or dilution of the sample) is introduced aseptically into a sterile or properly disinfected filtration assembly containing a sterile membrane filter (nominal pore size 0.2 or 0.45 µm). A vacuum is applied and the sample is drawn through the membrane filter. All indicator organisms are retained on or within the filter, which is then transferred to a suitable selective culture medium in a Petri dish. Following a period of resuscitation, during which the bacteria become acclimatized to the new conditions, the Petri dish is transferred to an incubator at the appropriate selective temperature where it is incubated for a suitable time to allow the replication of the indicator organisms. 09 01 29_PG_Bacteriological Examination of Water 7 / 24 Visually identifiable colonies are formed and counted, and the results are expressed in numbers of “colony forming units” (CFU) per 100 ml of original sample. This technique is inappropriate for waters with a level of turbidity that would cause the filter to become blocked before an adequate volume of water had passed through. When it is necessary to process low sample volumes (less than 10 ml), an adequate volume of sterile diluent must be used to disperse the sample before filtration and ensure that it passes evenly across the entire surface of the membrane filter. Membrane filters may be expensive in some countries. Sample volumes for different water types are: Where the quality of the water is totally unknown, it may be advisable to test two or more volumes in order to ensure that the number of colonies on the membrane is in the optimal range for counting (20–80 colonies per membrane). A full description of this method (also with video) is available on internet: http://www.rcpeh.com/index.php?option=com_content&task=view&id=118&Itemid=152 2. Multiple-tube method: In the multiple-tube method (MT) different amounts of water to be tested are added to tubes containing a suitable culture medium. The bacteria present in the water reproduce and produce acid with or without gas. From the number of tubes inoculated and the number with a positive reaction, the most probable number (MPN) of bacteria present in the original water sample can be determined statistically. The multiple-tube method is applicable to all kinds of water: it can be used with clear, coloured, or turbid water containing sewage or sewage sludge, or mud and soil particles, provided that the bacteria are homogeneously distributed in the prepared test samples. Example of culture media for multiple tube method and MPM 09 01 29_PG_Bacteriological Examination of Water 8 / 24 “SHORTER METHOD” BASED ON LAURYL SULPHATE BROTH: (e.g. FLUKA N°: 17349) Double strength broth: Dissolve 71.2 g of lauryl sulphate broth in 1 litre distilled water. After solubilisation, dispense 10 ml into each test tube containing inverted Durhan-tubes. Sterilise by autoclaving at 121°C for 15 minutes. Cool down slowly to prevent bubbles in Durhan-tubes. Single strength broth: Dissolve 35.6 g of lauryl sulphate broth in 1 litre distilled water. After solubilisation, dispense 10 ml into each test tube containing inverted Durhan-tubes. Sterilise by autoclaving at 121°C for 15 minutes. Cool down slowly to prevent bubbles in Durhan-tubes. Total Coliforms: Arrange three rows of 3 (5) tubes each in a test-tube rack. The tubes in the first row hold 10 ml of double strength lauryl sulphate broth, while the tubes in the second and the third rows contain 10 ml of single strength lauryl sulphate broth. With a sterile pipette (syringe) add 10 ml of the sample to each of the 3 (5) tubes of the first row. With a sterile pipette (syringe) add 1 ml of the sample to each of the 3 (5) tubes of the second row. With a sterile pipette (syringe) add 0.1 ml of the sample to each of the 3 (5) tubes of the third row. Add also in each tube 0.9 ml of sterile distilled water. 1. Incubate the tubes at 37°C for 18-24 hours. 2. Observe change in turbidity and appearance of gas in Durham tubes in bottles. 3. The media receiving one or more of the indicator bacteria show growth (turbidity) and a gas production which is absent in those receiving an inoculum of water without indicator bacteria. Presence of grow and gas indicates positive reaction whereas absence of either or both these features denotes a negative reaction. 4. The presumptive positives are read and remaining negative bottles are reincubated for another 24 hours. Any further positives are added to the previous figures. The probable numbers of coliforms are read from the probability tables of McCrady (Tables in annex). 5. From the number and distribution of positive and negative reactions, count of the most probable number (MPN) of indicator organisms in the sample may be estimated by reference to statistical tables. The test gives presumptive coliforms count as the reaction observed may occasionally be due to the presence of some organisms other than coliforms. For every tube showing fermentation (primary fermentation, presumptive coliforms), you may inoculate one new tube of Lauryl sulphate broth, from the tube showing primary fermentation, and incubated this tube at 44°C respectively. If there is fermentation in the tube incubated at 44°C after 8 to 24 hours, perform indole test by adding Kovac’s reagent. A positive indole test in a broth tube showing gas production at 44°C indicates the presence of E. coli. From the number and distribution of positive and negative reactions at 44°C, count of the most probable number (MPN) of indicator organisms in the sample may be estimated by reference to statistical tables. The test gives E. coli count 09 01 29_PG_Bacteriological Examination of Water 9 / 24 Thermotolerant coliforms + indole (E. coli): Arrange three rows of 3 (5) tubes each in a test-tube rack. The tubes in the first row hold 10 ml of double strength lauryl sulphate broth, while the tubes in the second and the third rows contain 10 ml of single strength lauryl sulphate broth. With a sterile pipette (syringe) add 10 ml of the sample to each of the 3 (5) tubes of the first row. With a sterile pipette (syringe) add 1 ml of the sample to each of the 3 (5) tubes of the second row. With a sterile pipette (syringe) add 0.1 ml of the sample to each of the 3 (5) tubes of the third row. Add also in each tube 0.9 ml of sterile distilled water. 1. Incubate the tubes at 44°C for 18-24 hours. 2. Observe change in turbidity and appearance of gas in Durham tubes in bottles. 3. The media receiving one or more of the indicator bacteria show growth (turbidity) and a gas production which is absent in those receiving an inoculum of water without indicator bacteria. Presence of grow and gas indicates positive reaction whereas absence of either or both these features denotes a negative reaction. 4. The presumptive positives are read, (perform indole test by adding 0.5 ml of Kovac’s reagent: a positive indole test in a broth tube showing gas production at 44°C indicates the presence of E. coli) and remaining negative bottles are reincubated for another 24 hours. Any further positives are tested with Kovacs and added to the previous figures. The probable numbers of E coli are read from the probability tables of McCrady (Tables in annex). 5. From the number and distribution of positive and negative reactions, count of the most probable number (MPN) of indicator organisms in the sample may be estimated by reference to statistical tables. The test gives E. coli count. Turbidity of the medium, accompanied by formation of gas (trapped in the Durhamtubes) within 48 hours is a positive presumptive for coliform bacteria. 09 01 29_PG_Bacteriological Examination of Water Kovac’s test: The test organism is cultured in a medium which contains tryptophane. The breaking down of the tryp-tophane with the releasing of indole is detected by the Kovac’s reagent. This reacts with the indole to produce a red coloured compound. Thermotolerant bacteria with indole production is a positive presumptive for E. coli. 10 / 24 Kovac's reagent: (this reagent is available commercially (e.g. VWR N°1.09293.0100). Amyl or isoamyl alcohol C5H12O 150 ml 4-Dimethylaminobenzaldehyde C9H11NO 10 g Concentrated HCl 50 ml Dissolve the 4-dimethylaminobenzaldehyde, C9H11NO, in 75 ml of isoamyl alcohol, C5H12O, and heat in a water bath at 60 °C for 5 min. Then add 25 ml of concentrated hydrochloric acid. The reagent will be ready for use after about 6 h to 7 h (indicated by a light yellow color). If a brown color results, the reagent should not be used. Kovac's reagent is stable at 4°C for 1 year. 09 01 29_PG_Bacteriological Examination of Water 11 / 24 09 01 29_PG_Bacteriological Examination of Water 12 / 24 TURBIDITY MEASURMENT: High levels of turbidity can protect microorganisms from the effects of disinfection, stimulate the growth of bacteria, and exert a significant chlorine demand. Where disinfection is practiced, the turbidity must always be low, e.g. below 5 NTU (Nephelometric Turbidity units, or JTU for JacksonTurbidity Units), and ideally below 1 NTU for effective disinfection. Measurement of turbidities lower than 5 NTU will generally require electronic meters. However, turbidities of 5 NTU upwards can be measured by simple extinction methods, which are far cheaper and require no consumables. In the monitoring of small community supplies in developing countries, such methods may be preferable. The sequence of steps involved in turbidity determination by an extinction method is shown below. Turbidity may change during sample transit and storage, and should therefore be measured on site at the time of sampling. Add water slowly to the turbidity tube, taking care not to form bubbles. Fill until the mark at the bottom of the tube just disappears. Read the turbidity from the scale marked on the side of the tube. The value is that corresponding to the line nearest to the level of the water in the tube. The scale is not linear, and extrapolation of values between the lines is therefore not recommended. 09 01 29_PG_Bacteriological Examination of Water 13 / 24 RESIDUAL FREE CHLORINE TEST The method recommended for the determination of chlorine residual in drinking water employs N,N-diethyl-p-phenylenediamine, more commonly referred to as DPD. Methods employing orthotolidine and starch–potassium iodide were formerly also recommended. The first of these reagents is a recognized carcinogen and the method is not reliable. The method based on the use of starch–potassiumiodide is not specific for free chlorine, but measures directly the total of free and combined chlorine; it is not recommended except in countries where DPD cannot be obtained or prepared. In this Annex, therefore, only the DPD method is considered. In the laboratory, colorimetry or spectrophotometry may both be used for the determination of chlorine by means of DPD. However, it is common practice and highly recommended for field measurements using simple color match comparators to be done on site. The color is generated following the addition of DPD to the water sample and is matched against standard colored discs or tubes. The method can be used by staff without extensive specialized training. The reagent may be solid (e.g. individually wrapped tablets) or in the form of a solution; the former is more stable. If the solution is used, it should be stored in a brown bottle and discarded as soon as it starts to become discolored. It is important to measure pH at the same time as chlorine residual since the efficacy of disinfection with chlorine is highly pH-dependent: where the pH exceeds 8.0, disinfection is less effective. To check that the pH is in the optimal range for disinfection with chlorine (less than 8.0), simple tests may be conducted in the field using comparators such as that used for chlorine residual. With some chlorine comparators, it is possible to measure pH and chlorine residual simultaneously. Alternatively, portable pH electrodes and meters are available. If these are used in the laboratory, they must be calibrated against fresh pH standards at least daily; for field use, they should be calibrated immediately before each test. Results may be inaccurate if the water has a low buffering capacity. 09 01 29_PG_Bacteriological Examination of Water 14 / 24 Target values: Class Grade Presumptive total coliform count (per 100 ml) E. coli count (per 100 ml) I Excellent 0 0 II Satisfactory 1-3 0 III Suspicious 4-10 0 IV Unsatisfactory >10 0,1 or more International Organization for Standardization (ISO) standards for detection and enumeration of faecal indicator bacteria in water ISO standard Title (water quality) The objective of zero E. coli per 100 ml of water is the goal for all water supplies and should be the target even in emergencies; however, it may be difficult to achieve in the immediate post-disaster period. This highlights the need for appropriate disinfection. An indication of a certain level of faecal indicator bacteria alone is not a reliable guide to microbial water safety. Some faecal pathogens, including many viruses and protozoan cysts and oocysts, may be more resistant to treatment (e.g., by chlorine) than common faecal indicator bacteria. More generally, if a sanitary survey suggests the risk of faecal contamination, then even a very low level of faecal contamination may be considered to present a risk, especially during an outbreak of a potentially waterborne disease, such as cholera. Drinking-water should be disinfected in emergency situations, and an adequate disinfectant residual (e.g., chlorine) should be maintained in the system. Turbid water should be clarified wherever possible to enable disinfection to be effective. Minimum target concentrations for chlorine at point of delivery are 0.2 mg/liter in normal circumstances and 0.5 mg/liter in highrisk circumstances. It is necessary to know the pH of water, because more alkaline water requires a longer contact time or a higher free residual chlorine level at the end of the contact time for adequate disinfection (0.4–0.5 mg/liter at pH 6–8, rising to 0.6 mg/liter at pH 8–9; chlorination may be ineffective above pH 9). Typical sample volumes and numbers of tubes for the multiple-tube method 09 01 29_PG_Bacteriological Examination of Water 15 / 24 How Chlorine Kills Pathogens How does chlorine carry out its well-known role of making water safe? Upon adding chlorine to water, two chemical species, known together as “free chlorine,” are formed. These species, hypochlorous acid (HOCl, electrically neutral) and hypochlorite ion (OCl-, electrically negative), behave very differently. Hypochlorous acid is not only more reactive than the hypochlorite ion, but is also a stronger disinfectant and oxidant. The ratio of hypochlorous acid to hypochlorite ion in water is determined by the pH. At low pH (higher acidity), hypochlorous acid dominates while at high pH hypochlorite ion dominates. Thus, the speed and efficacy of chlorine disinfection against pathogens may be affected by the pH of the water being treated. Fortunately, bacteria and viruses are relatively easy targets of chlorination over a wide range of pH. However, treatment operators of surface water systems treating raw water contaminated by the parasitic protozoan Giardia may take advantage of the pH-hypochlorous acid relationship and adjust the pH to be effective against Giardia, which is much more resistant to chlorination than either viruses or bacteria. Another reason for maintaining a predominance of hypochlorous acid during treatment has to do with the fact that pathogen surfaces carry a natural negative electrical charge. These surfaces are more readily penetrated by the uncharged, electrically neutral hypochlorous acid than the negatively charged hypochlorite ion. Moving through slime coatings, cell walls and resistant shells of waterborne microorganisms, hypochlorous acid effectively destroys these pathogens. Water is made microbiologically safe as pathogens either die or are rendered incapable of reproducing. A typical bacterium has a negatively charged slime coating on its exterior cell wall, which is effectively penetrated by electrically neutral hypochlorous acid, favored by lower pH’s. (Reprinted from The Chlorination/Chloramination Handbook by permission. Copyright © 1996, American Water Works Association.) Source: Connell, 1996. 09 01 29_PG_Bacteriological Examination of Water 16 / 24 ANNEX 1: LAURYL SULFATE BROTH, MSDS 09 01 29_PG_Bacteriological Examination of Water 17 / 24 09 01 29_PG_Bacteriological Examination of Water 18 / 24 09 01 29_PG_Bacteriological Examination of Water 19 / 24 ANNEX 2: SODIUM THIOSULFATE PENTAHYDRATE, MSDS Fisher Products, Material Safety Data Sheet ACC# 21715 Section 1 - Chemical Product and Company Identification MSDS Name: Sodium Thiosulfate Pentahydrate Catalog Numbers: S78930, NC9503498, NC9629890, NC9671016, NC9979554, S445-10, S445-3, S445-50, S445-500, S474-12, S474-3, S474-500, XXS445100KG, XXS47450KG Synonyms: Sodium Hyposulfite; Disodium Salt Pentahydrate; Disodium Thiosulfate Pentahydrate. Company Identification: Fisher Scientific 1 Reagent Lane Fair Lawn, NJ 07410 For information, call: 201-796-7100 Emergency Number: 201-796-7100 For CHEMTREC assistance, call: 800-424-9300 For International CHEMTREC assistance, call: 703-527-3887 Section 2 - Composition, Information on Ingredients CAS#Chemical NamePercentEINECS/ELINCS 10102-17-7Sodium Thiosulfate, Pentahydrate100 unlisted Hazard Symbols: None listed. Risk Phrases: None listed. Section 3 - Hazards Identification EMERGENCY OVERVIEW Appearance: colourless to white solid. Caution! May cause eye and skin irritation. Hygroscopic (absorbs moisture from the air). The toxicological properties of this material have not been fully investigated. May cause respiratory and digestive tract irritation. Target Organs: No data found. Potential Health Effects Eye: May cause eye irritation. Skin: Prolonged and/or repeated contact may cause irritation and/or dermatitis. Ingestion: Ingestion of large amounts may cause gastrointestinal irritation. The toxicological properties of this substance have not been fully investigated. Inhalation: May cause respiratory tract irritation. Low hazard for usual industrial handling. The toxicological properties of this substance have not been fully investigated. Chronic: No information found. Section 4 - First Aid Measures 09 01 29_PG_Bacteriological Examination of Water 20 / 24 Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid. Skin: Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Get medical aid if irritation develops or persists. Wash clothing before reuse. Ingestion: Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water. Inhalation: Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid. Notes to Physician: Treat symptomatically and supportively. Section 5 - Fire Fighting Measures General Information: As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Wear appropriate protective clothing to prevent contact with skin and eyes. Wear a self-contained breathing apparatus (SCBA) to prevent contact with thermal decomposition products. Extinguishing Media: Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam. Flash Point: Not applicable. Autoignition Temperature: Not applicable. Explosion Limits, Lower: Not available. Upper: Not available. NFPA Rating: (estimated) Health: 1; Flammability: 0; Instability: 0 Section 6 - Accidental Release Measures General Information: Use proper personal protective equipment as indicated in Section 7. Spills/Leaks: Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions. Provide ventilation. Section 7 - Handling and Storage Handling: Wash thoroughly after handling. Use with adequate ventilation. Minimize dust generation and accumulation. Avoid prolonged or repeated contact with skin. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation. Storage: Store in a tightly closed container. Keep from contact with oxidizing materials. Store in a cool, dry, well-ventilated area away from incompatible substances. Keep away from strong acids. Store protected from moisture. Section 8 - Exposure Controls, Personal Protection 09 01 29_PG_Bacteriological Examination of Water 21 / 24 Engineering Controls: Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low. Exposure Limits Chemical NameACGIHNIOSHOSHA - Final PELs Sodium Thiosulfate, Pentahydratenone listednone listednone listed OSHA Vacated PELs: Sodium Thiosulfate, Pentahydrate: No OSHA Vacated PELs are listed for this chemical. Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166. Skin: Wear appropriate protective gloves to prevent skin exposure. Clothing: Wear appropriate protective clothing to prevent skin exposure. Respirators: A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant a respirator's use. Section 9 - Physical and Chemical Properties Physical State: Solid Appearance: colourless to white Odour: odourless pH: Not available. Vapour Pressure: Not available. Vapour Density: Not available. Evaporation Rate: Not available. Viscosity: Not available. Boiling Point: Not available. Freezing/Melting Point: 48.5 deg C Decomposition Temperature: > 45 deg C Solubility: 680 G/L WATER (20óC) Specific Gravity/Density:1.7290g/cm3 Molecular Formula: Na2O3S2.5H2O Molecular Weight: 248.18 Section 10 - Stability and Reactivity Chemical Stability: Stable. Conditions to Avoid: Incompatible materials, acids, strong oxidants, exposure to moist air or water. Incompatibilities with Other Materials: Moisture, strong acids, strong bases, strong oxidizing agents. Hazardous Decomposition Products: Irritating and toxic fumes and gases, hydrogen sulphide, sodium oxide. Hazardous Polymerization: Has not been reported. Section 11 - Toxicological Information RTECS#: CAS# 10102-17-7: WE6660000 LD50/LC50: Not available. 09 01 29_PG_Bacteriological Examination of Water 22 / 24 Carcinogenicity: CAS# 10102-17-7: Not listed by ACGIH, IARC, NIOSH, NTP, or OSHA. Epidemiology: No information available. Teratogenicity: No information available. Reproductive Effects: No information available. Neurotoxicity: No information available. Mutagenicity: No information available. Other Studies: See actual entry in RTECS for complete information. Section 12 - Ecological Information No information available. Section 13 - Disposal Considerations Chemical waste generators must determine whether a discarded chemical is classified as a hazardous waste. US EPA guidelines for the classification determination are listed in 40 CFR Parts 261.3. Additionally, waste generators must consult state and local hazardous waste regulations to ensure complete and accurate classification. RCRA P-Series: None listed. RCRA U-Series: None listed. Section 14 - Transport Information US DOTIATARID/ADRIMOCanada TDG Shipping Name: No information available. Hazard Class: UN Number: Packing Group: Section 15 - Regulatory Information US FEDERAL TSCA CAS# 10102-17-7 is not on the TSCA Inventory because it is a hydrate. It is considered to be listed if the CAS number for the anhydrous form is on the inventory (40CFR720.3(u)(2)). Health & Safety Reporting List None of the chemicals are on the Health & Safety Reporting List. Chemical Test Rules None of the chemicals in this product are under a Chemical Test Rule. Section 12b None of the chemicals are listed under TSCA Section 12b. TSCA Significant New Use Rule None of the chemicals in this material have a SNUR under TSCA. SARA CERCLA Hazardous Substances and corresponding RQs None of the chemicals in this material have an RQ. SARA Section 302 Extremely Hazardous Substances None of the chemicals in this product have a TPQ. Section 313 09 01 29_PG_Bacteriological Examination of Water 23 / 24 No chemicals are reportable under Section 313. Clean Air Act: This material does not contain any hazardous air pollutants. This material does not contain any Class 1 Ozone depletors. This material does not contain any Class 2 Ozone depletors. Clean Water Act: None of the chemicals in this product are listed as Hazardous Substances under the CWA. None of the chemicals in this product are listed as Priority Pollutants under the CWA. None of the chemicals in this product are listed as Toxic Pollutants under the CWA. OSHA: None of the chemicals in this product are considered highly hazardous by OSHA. STATE CAS# 10102-17-7 is not present on state lists from CA, PA, MN, MA, FL, or NJ. California No Significant Risk Level: None of the chemicals in this product are listed. European/International Regulations European Labeling in Accordance with EC Directives Hazard Symbols: Not available. Risk Phrases: Safety Phrases: S 37 Wear suitable gloves. S 45 In case of accident or if you feel unwell, seek medical advice immediately (show the label where possible). S 28A After contact with skin, wash immediately with plenty of water. WGK (Water Danger/Protection) CAS# 10102-17-7: 0 Canada - DSL/NDSL None of the chemicals in this product are listed on the DSL or NDSL list. Canada - WHMIS This product has a WHMIS classification of D2B. Canadian Ingredient Disclosure List Exposure Limits Section 16 - Additional Information MSDS Creation Date: 12/12/1997 Revision #5 Date: 3/18/2003 The information above is believed to be accurate and represents the best information currently available to us. However, we make no warranty of merchantability or any other warranty, express or implied, with respect to such information, and we assume no liability resulting from its use. Users should make their own investigations to determine the suitability of the information for their particular purposes. In no event shall Fisher be liable for any claims, losses, or damages of any third party or for lost profits or any special, indirect, incidental, consequential or exemplary damages, howsoever arising, even if Fisher has been advised of the possibility of such damages. 09 01 29_PG_Bacteriological Examination of Water 24 / 24