1 CHE 106 General Chemistry Lecture, Fall 2008
advertisement

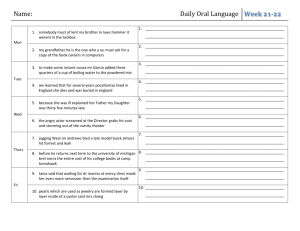
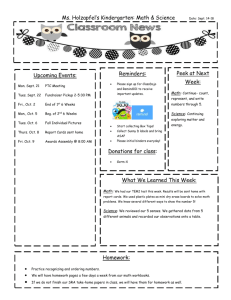
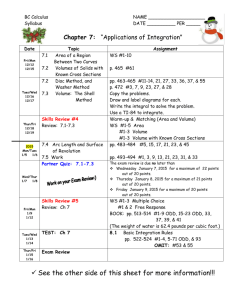
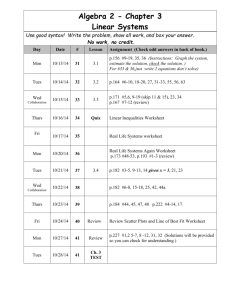
CHE 106 General Chemistry Lecture, Fall 2008 GENERAL COURSE INFORMATION Instructor Information Dr. Mary Boyden Office: 3-008 Center for Science and Technology Office Hours: Mondays 1:45-2:45 Phone: 443-2127 Email: mnboyden@syr.edu Website: blackboard.syr.edu (CHE.106.M013) Class Time and Location Monday, Wednesday & Friday 12:45-1:40, Stolkin Auditorium (Physics) REQUIRED Textbook and Supporting Material (all available in the SU bookstore) • General Chemistry, 9th ed. by D.D. Ebbing and S.D. Gammon (Houghton Mifflin, 2009) • Student Solutions Manual (General Chemistry, 9th edition); D. Bookin, D.D. Ebbing, and S.D. Gammon (Houghton Mifflin) • Publisher Web Site: http://college.hmco.com/pic/ebbing9e Course Description This course concentrates on the fundamental principles and laws underlying chemical action, states of matter, atomic and molecular structure, chemical bonding, stoichiometry, properties of solutions, chemical equilibrium, and introductory thermodynamics. Your basic responsibilities include: ! Attend all lectures, recitations and exams and bring a scientific calculator. ! Read the assigned material prior to class. ! Study your lecture notes and assigned text reading. ! Do assigned homework problems on time and review them before exams. ! Do not fall behind! ! Take all examinations! If you have any questions about the homework, the quizzes, the final, or anything else, it is the responsibility of the student to find the appropriate TA or Professor Boyden to get help in a timely manner. Laboratory You should take CHE 107, a one-credit lab course which fulfills the general chemistry lab requirement. You will find CHE 107 helpful. It gives you a additional experience with chemistry concepts and introduces you to useful laboratory skills. CHE 107 is taught and graded totally separately from CHE 106. Please do not ask your CHE 106 TAs questions about the labs. Academic integrity Your work on all exams, quizzes, and any assignments labeled "individual work" must be your own. Cheating will be penalized and reported to the College of Arts and Sciences. Your signature on exams affirms that you have understood this academic-integrity requirement. In the case of homework assignments, collaborative work with other students in the course is encouraged. 1 Absences Absences due to a religion’s holy day requirements will be excused. You must notify Professor Boyden and your TA in writing at least one week before the absence; an opportunity to make up lecture or recitation class work will be provided. Medical absences will be excused based on written advice from the Health Center or a health-care provider (based upon clinical findings and prescribed treatment recommendations). The medical document must specifically indicate that you were unable to attend class/recitation. All such absences will be verified by Chemistry Department staff. No verbal excuses will be accepted. There will be absolutely NO makeup examinations except in the case of advance-notice approved absences. Disability If you have a learning or physical disability, please see Professor Boyden as soon as possible during the first 2 weeks of the course to arrange for appropriate accommodations. No provisions/accommodations will be made if the instructor is notified after examinations. Exams Exams will cover both material covered in lecture and the assigned text readings. Some questions will come from lecture (not covered in text) and others from the text (not covered in lecture). Many questions will be problems similar to assigned homework exercises. Lots of practice with problems is the key to success in this course. Each hourly exam will focus on specific chapters and is given during the regular class period. The final exam will cover the entire semester. First Hour Examination Second Hour Examination Third Hour Examination Fourth Hour Examination FINAL EXAMINATION Wednesday, September 17th Wednesday, October 8th Wednesday, October 29th Wednesday, November 19th Monday, December 8th, 8:00-10:00 Final Grade Determination Course grades are based on total points out of a possible 500 points. Curving will generally not be applied, but I reserve the right to do so. In such a case, scores will only be curved up and never down. The final grade will be computed as follows: Four Hour Exams (75 points each) 300 (60%) Final Exam (150 points) 150 (30%) Recitation (50 points) 50 (10%) Course Total : 500 points (100%) Grade ranges based upon raw score percentages: A = ! 90% A- = 88-89% B+ = 85-87% B = 80-84% B- = 75-79% C+ = 70-74% C = 60-69% C- = 55-59% D = 40-54% F = < 40% 2 APPROXIMATE COURSE SCHEDULE The following schedule of classes lists the topics that will be covered on a particular date along with the relevant reading in the textbook. It is in your best interest to have completed the reading before the scheduled lecture time since it will make the lecture much easier to follow. Copies of the lecture notes will be put on the course web site. DATE TOPIC Mon. Aug. 25 Syllabus/Course Overview Wed. Aug 27 Introduction to Chemistry TEXT READING (Chapter.Section) Preface Study the syllabus 1.1 - 1.3 Appendix A.1 Fri. Aug 29 Matter, Measurement Mon. Sept. 1 Labor Day (no class) Wed. Sept. 3 Measurement 1.6-1.8 Atomic Theory & Structure 2.1 - 2.3 Atomic Mass, Periodic Table 2.4 - 2.6 Chemical Substances 2.6-2.8 Reactions 2.9-2.10 Mon. Sept. 15 Mass/Moles 3.1-3.2 Wed. Sept. 17 First Exam Chapters 1, 2 Determining Chemical Formulas 3.3-3.5 Mon. Sept. 22 Stoichiometry 3.6-3.8 Wed. Sept. 24 Ions, Chemical Reactions 4.1-4.3 Chemical Reaction 4.4-4.6 Solutions 4.7-4.8 Quantitative Analysis 4.9-4.10 Fri. Oct. 3 Thermochemistry 6.1-6.3 Mon. Oct. 6 Thermochemistry 6.4-6.6 Wed. Oct. 8 Second Exam Chapters 3, 4, 6.1-6.3 Fri. Oct. 10 Thermochemistry 6.7-6.9 Mon. Oct. 13 Light Waves, Photons 7.1-7.2 Wed. Oct. 15 Bohr Theory, Quantum Mechanics 7.3-7.4 Fri. Oct. 17 Quantum Numbers, Atomic Orbitals 7.5 Fri. Sept. 5 Mon. Sept. 8 Wed. Sept. 10 Fri. Sept. 12 Fri. Sept. 19 Fri. Sept. 26 Mon. Sept. 29 Wed. Oct. 1 3 1.4 - 1.5 Mon. Oct. 20 Electronic Structure 8.1-8.2 Wed. Oct. 22 Electronic Structure 8.3-8.4 Fri. Oct. 24 Periodicity 8.5-8.6 Mon. Oct. 27 Periodicity 8.7 Wed. Oct. 29 Third Exam Chapters 6.4-6.9, 7, 8.1-8.6 Fri. Oct. 31 Ionic Bonds 9.1-9.3 Mon. Nov. 3 Covalent Bonds 9.4-9.7 Wed. Nov. 5 Covalent Bonds 9.8-9.11 Molecular Geometry 10.1-10.2 Mon. Nov. 10 Bonding, Molecular Orbital Theory 10.3-10.4 Wed. Nov. 12 Molecular Orbital Theory 10.5-10.6 Fri. Nov. 14 Molecular Orbital Theory 10.6-10.7 Mon. Nov. 17 Special Topic: Proteins 24.3 Wed. Nov. 19 Fourth Exam Chapters 8.7, 9, 10 Fri. Nov. 21 Gas Laws 5.1-5.2 Mon. Nov. 24 Gas Laws 5.3-5.5 Wed. Nov. 26 Thanksgiving Break (no class) Fri. Nov. 28 Thanksgiving Break (no class) Mon. Dec. 1 Gas Laws 5.6-5.7 Wed. Dec. 3 Review 1-5 Fri. Dec. 5 Review 6-10 CUMULATIVE FINAL EXAMINATION (8-10 AM) All material Fri. Nov. 7 Mon. Dec. 8 4 RECITATION AND HOMEWORK SCHEDULE Each week in recitation, the homework exercises specified below will be discussed. Students will have the opportunity to ask questions about these exercises and also the relevant text and lecture material. At the end of each recitation, students will be given a short quiz taken from that week’s homework set. Recitation grades are based on quizzes, attendance, and participation. Most assigned exercises use odd-numbered problems. The text book contains the answers to these problems. The Solutions Manual contains detailed solutions to these problems. If you are having difficulty, refer to the solutions manual to support your learning. You may wish to extend your practice problems to even-numbered problems. Week of: August 25 September 1 September 8 September 15 September 22 September 29 October 6 October 13 October 20 October 27 November 3 November 10 November 17 November 24 December 1 Problems Due: No recitations are held this week. No recitation is held on Monday. No quizzes are given this week. Chapter 1 37,41,45,47,51,52,53,55 Appendix Exercises 1,2,3,4 Chapter 1 27,57,61,63,65,67,77,79,81,82,83,89 Chapter 2 27,31,37,43,45, Chapter 2 48,49,51,55,56,57,59,61,67,69,77,79,97,98ab,99 Review for the exam. Chapter 3 21,27,35,37d,39,41cd,45acd,46,51,55,57ac, 65,67,69,71,75 Chapter 3 77,79,82,83,86,89,91,93,97 Chapter 4 29,31abc,33,34c,37ab,38c,41,43ab,51,57,58a,65 Chapter 4 71,73,83,84 Chapter 6 41,45,47,50 Review for the exam. Chapter 6 51 [change to iron(iii)],53,55,56,59,61,62,71,72,73, 75,77,79,80,83 Chapter 7 35,37,38,43,44,45,49,50,53,57,58,61,63,69,70 Chapter 71,5,9,13,16,18,25,31 (do these after 35-70) Chapter 8 41,42,43,45,47(use Cl),48,49,51,57,58,59 Chapter 8 2,3,5,7,11 (do these after 41-65) Review for the exam. Chapter 8 61,62,63,65 Chapter 9 35acd,37,38a,39,43 Chapter 9 51,55,57,58,59,61,62b,63a,65ac,67,68a,75 Chapter 10 33,34ab,35,36bd Chapter 10 41,43,8,9 Review for the exam. No recitations are held on Wednesday or Friday. No quizzes are given this week. Chapter 5 37,39,45 Chapter 5 57,61,81,29,30,32abc,33 5