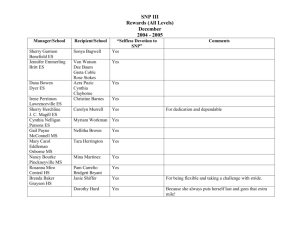
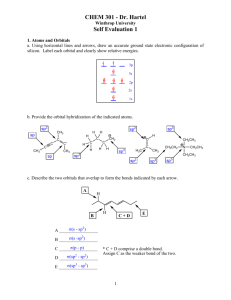
Answers - La Salle University
advertisement

Chemistry 201-01 Hour Examination I 20 September 2002 Name_____Dr. Price________ Answer all of the questions in the spaces provided. Point values are in parentheses. Be sure to read the questions carefully. 1.(15) Propose the best possible Lewis-dot structures for the following species. Use lines for covalent bonds, show any formal charges where appropriate, and indicate the hybridization of each atom other than hydrogen or halogen. a) C2O2Cl2 sp2 O Cl sp2 C C c) HNO2 sp3 b) CHO2 sp2 O Cl H O C sp2 H O O sp2 N sp2 O best with no formal charges sp3 2.(10) For the following reactions identify the acid, base and both conjugate species. Also determine whether or not the reaction proceeds to the right as written. (pKa values in general: alcohols 15-18; alkanes 49-55; carboxylic acids 4-6; phosphoric acid 2.) OLi OH a) (CH3)3CLi + base + (CH3)3CH acid c.b. O COH b) c.a. O COK + KH2PO4 acid base + H3PO4 c.b. c.a. 3.(10) Draw the resonance structure for each of the species below using the arrows indicating the electron flow. O O a) b) 4.(12) Identify and name all of the functional groups in aspartame (Nutrasweet) and Effexor® (antidepressant Vanlafaxine) shown below: amide amine O CH3 CH3 OH N amine alcohol O H2NCHCNHCHCOCH3 ester CH2 HOCCH2 carboxylic acid O arene arene ether Aspartame OCH3 Venlafaxine 5.(12) The molecule shown below is an extremely unusual, unstable and unsaturated hydrocarbon. In the diagram below, identify all orbitals (e.g. px, sp2) and bond types (e.g. π, σ). Additionally, propose a Lewis structure (lines for all bonds) for this hydrocarbon. py sp2 sH Hs σ C π σ sp2 sp pz py σ σ H C sp sp2 C sp2 s pz π σ H H H H C C C H 6.(15) Using Kekulé structures (zig-zag), draw 7 unique structural isomers with the formula of C7H16. There are 9 possible isomers. 7.(16) Give the names for the following compounds using I.U.P.A.C. nomenclature. 8 1 7 2 6 a) (CH3)2CH(CH2)3CH(CH3)2 b) 5 3 4 2,6-dimethylheptane c) CH2CH2CH2Cl CH3CHCH2CH2CCH3 CH3CHCH3 5-ethyl-3,5-dimethyloctane d) CH2CH3 Cl 1,4-dichloro-4,7,8-trimethylnonane CH2CH3 cis 1,4-diethylcytclobutane 8.(10) Compare BF3 and BH3. Which do you suppose is the stronger Lewis acid. Draw accurate depictions of both structures (make sure the bond angles are reasonable) and explain why you feel one is a stronger Lewis acid than the other. very small bond moments H H B F H F B F Both are Lewis acids but BF3 is stronger because the Boron is more electron-deficie thus its empty p orbital is more likely to act as an electron-pair acceptor. Draw the product of the Lewis acid-base reaction between boron trifluoride and ammonia. F BF3 + NH3 F B F N H H H