THE VAPOUR PRESSURE OF PURE ORGANIC COMPOUNDS BY
advertisement

63
Revista Mexicana de Física 30 no. 1 (1983)63-82
THE VAPOUR PRESSURE
OF PURE ORGANIC COMPOUNDS
BY DYNAMIC EBULLlOMETRY
Armando Olivares Fuentes,
Arturo Treja
Joe' Suárez Cansino and
Rodrfguez
Instituto Mexicano del Petrdleo
Subdirección de LB.P. - TEFETRA
Eje Central lázaro Cárdenas 152
Deleg. Gustavo A. Madero
07730 México, D.F.
(recibido noviembre 30, 1982; aceptado junio 30, 1983)
ABSTRACT
A dynarnic ebu1liometer has been used to measure vapour pressures
of 2-propanol,2,2,4-trimethylpentane,
n-pentane, methanol, ethanol, acetone
and benzene from approximately26 to 78 kPa. The ebul1iorneter is similar
to one reported in the literature although with modifications which make
it original together with the auxiliary equipment and measuring technique.
The values obtained in this work to test the ebulliorneter are compared
with data previously reported by other workers and the overall agreernent
is good. The experimental results were fitted to the Antoine equation
and from it derived quantities were obtained for each of the seven
substances studied.
64
RESlNEN
Un ebulliómetro dinámico ha sido utilizado para medir presiones
de vapor de iso-propanol, 2,2,4-trimetilpentano,
n-pantano, metano!, etanol, acetona y benceno desde 26 a 78 kPa. El ehulliómetro está basado en
uno reportado en la literatura aunque incluye modificaciones que lo hacen
original junto con el equipo auxiliar y la técnica de medición.
Los vapo
res obtenidos en este trabajo se comparan con datos previamente reporta ~
dos por otros grupos de investigación y la comparación es satisfactoria.
Los resultados experimentales fueron ajustados a la ecuación de Antoine y
a partir de ella se derivaron otras propiedades para cada una de las siete sustancias estudiadas.
1.
The knowledge
ces and mixtures
operation
HITRODUCTlON
of the thennodynamic
is oí great importance
of many industrial
processes.
point of view those properties
to give information
propcrties
of pure substan-
for the simulation,
design and
Further more, from a theoretical
are used to test models of solutions
on the molecular
and
interactions.
TABLE 1
nn
(293.2
This
work
2 - propanol
Substance
nn
(298.20
Literature(l)
This
work
Literature(l)
1. 3778
1.3772
1.3754
1.3753
0
K)
K)
2,2,4-trimethylpentane
1.3916
1.:\915
1. 3894
1.3890
n - pentane
1. 3580
1. 3575
1. 3560
1.3547
methanol
1. 3292
1.3286
1. 3270
1. 3266
ethanol
acetone
1. 3619
1. 3602
1. 3595
1.3614
1.3588 (2)
1.3571
1. :>594
1. 3570 (2)
benzene
l.5140
1. 5011
1. 4987
1. 4979
Table I.
Refractive
index at 293.2 and 2.98.2° K.
65
One oí the thennodynamic
oí pure ar single - compo-
properties
nent liquids that has becn widely studied is the vapour pressure
function
of temperature
ebulliometer
(T).
Thus, we repart a modified
which has becn tested measuring
2-propanol
methanol
=
in kPa (1 atmosphere
26 to 78, 2,Z,4-trimethylpentane
all~glass
the vapour pressure
pure substances as a function oí temperature.
p for eaeh substance
(p) as a
dynamic
oí seven
The approximate rangcs oí
101.325 kPa
=
760 mmHg) are:
19 to 78, n-pentane
26 to 124,
26 to 78, ethanol 11 to 78, acetone 26 to 78 and benzene 16 to
78. Values obtained elswhere oí the refractive
substanccs are presentcd in rabIe l.
indexes oí these
APPARAIDS
2.
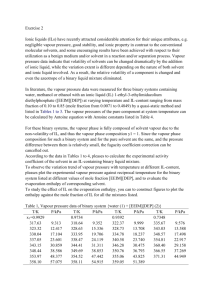
A circulating ar dynamic ebulliometer similar to that describcd
by Rogalski and co-workers(3,4)
was used. Fig. 1 shows a schematic
diagram
of the ebulliometer.
ebulliometer
Thc main modification
is in the measuremcnt
present
of the equilibrium
the vapour and liquid phases having the thermometer
ebulliometer
between
with a threadcd glass joint.
the thermomcter
in our
tempcrature
between
sealed in the
This allows direct contact
and the t"u phases thus the response
is fas ter
than using thennowells.
The working
principIe
oí the ebulliorneter is now described.
liquid and vapour is continuosly
aid of an e1ectrica1
heater and pumped by the Cottre11 pump A (see Fig. 1)
e
towards thc equilibrium
chamber
the joint D.
tape wound externaIly
A heating
partiaI condensation
thermodynamic
where the thermometer
equiIibrium
technique
with the
is sealed with
around the arm B prevents
of the vapour before reaching
As part of the measuring
generated
A
stream oí overheated
the equilibrium
the steady state corresponding
has to be established
ehamber.
to thc
to carry out measurements
oí p-T and this was done in the present work by setting a value of drops
berwcen
70 to 90 as eOlmted
in the drop eounter
F.
Finally thc equilibrium
temperature is determined in e as explained aboye írem the contact between
the thermomcter and the liquid and vapour phascs.
Mixing oí thc liquid phase from
phase írem the condenser
E is achieved
e
and the condensed vapour
in the devices
1 and J before thc
66
8
G
F
1
J
Fig •.1.
The dynamic ebulliometer
different parts).
A
(see the text for description
liquid sample is again heated in A and the cycle started.
oi the
The apparatus
described here is a150 useful fay the determination of the vapour~ liquid
equilibrium of mixtures(3,4,S) since both the liquid and vapour phases
may be sampled to determine
their composition
through H and F, respectively.
67
3.
The measurement
manometer
AlJJ(ILIARY EQUIINENf
oí pressure was carried out with a mercury
used in the absolute
continuosly
evacuated.
a Gaertner
eathetometer
type oí manometer
approximately
fashion, i.e.,tbe reference
The height
(morlel M-9I2) with a precision oí iD.OI mm.
is wel1 suited far pressure measurement
130 kPa as discussed
by Ambrose(6).
A
was used to control the pressure in the ebulliometer
system composcd
thermal1y
manostat
through a surge
This surge system was connected
and to the mercury manometer
pump was used for measurements
oí vapour pressures
A dircct-drive
of equi1ibrium
and yapour
in the equi1ibrium
Thermometer
(Hew1ett-Packard
iO.04° K as reported
vacuum
below the atmospheric
dry nitrogen was used as the pressurizing
aboye the latter.
The measurement
each
to the ebulliometer
through a liquid ni trogen trap and connected
systern through the manostat.
to the pressure-vacuum
This
up to
pelID ••..
¡alt
amongst othcr things oí two glass spheres oí 20 1
insulated.
va1ue whereas
limb was
oí thc mercury colurnn Has measured with
temperatures
gas for pressures
be~~en
the liquid
chamber was carried out with a Quartz
mode1 2804 A) with an absolute
by the makers
for the temperature
accuracy of
range studied
in
this work.
4.
EXPERIMENTAL TEQiNIQUE
The pure liquid to be studied was p1aced in the ebu11iometer
through
slight1y
the tef10n va1ve G (see Fig. 1) until the 1iquid level was
aboye the mixing chamber
mixing-boi1ing
has been estab1ished
state has to be defined
of drops of condensate
three quantities
l.
After the cyc1e boi1ing-condensationin the ebu11iometer
in terms of the pressure,
the stationary
temperature
and amount
flowing through the drop counter F since these
should be independent
oí time.
rule there exists only one degree of freedom
According
to the phase
for a system of one
component and two phases, thus we decided to control the pressure within
the ebulliameter in order to automatically define the thermodynamic state
of the substance
being studied,
that is, the temperature
is inmediate1y
68
obtained
far a given constan! pressure.
previous
section the amount oí drops that we used as a criterion
blish the stationary statc.
Several corrections
height of the mercury
Thesc corrections
however,
in a
to esta-
have to be applied to the directly measured
column in the manometer
fer the vapour pressurc.
elsewhcre(6,7),
We have already mentioned
far completeness
to finally obtain a value
are discussed
in detail
we give here the equation
that
was used to calculate values of vapour pressure from the manometric
readings:
.
= 0.9972
p/mm Hg
where
lJl
óh(l - 1.634x10
is the difference
-,
t)
(1)
Po •
+
in height oí the rnercury
collUTUlS
between
the
rncasuring and reference 1imbs of the manometer, Po is
fhe
pressure
is the tcmperature
in the reference
1 imb oí the manometer
and
t
residual
oí the manometer.
S.
RESULTSAND D1SruSSlON
The experirnentally measured vapour pressures
ven substances
residuals p
=
studied are listed in Tables 11 to VIII together with their
(p
exp
log,. (p/kPa)
=
- p
A
The constants
cal
) from the Antoine
B/{(T/K)
+
+
equation:
(2)
el
of Eq. (Z) were obtained
from a least - square fit
ad are given in Table IX.
The residuals in Table 11 to VIII eorrespond
fferences no greater than
furthenmore
for each of the se-
!O.OZ-K indieating
to temperature
di-
the goodness of the fit,
the óp values are randomly distributed.
Figures 2 to 8 show the pressure residuals ~p based on Eq. (2).
Also included en these figures are the limits of pressure changcs
corresponding to changes in the temperature of !O.02° K and !O.04° K.
In order to ecmpare wi th the present work we have used published
values for eaeh substanee
te calculate
from Eq. (2) using the eonstants
óp where now pare
in Table IX and pare
exp
cal
the values
the literature
69
values.
These residuals
is observed
literature
far
OUT
values.
FroID this comparison
is possible
A further test on the quality
oí the thermometer.
oí the results oí this work and
out by calculating
fitted to them may be carried
quantities
such as the nonmal boiling
saturatian
curve dp/dt and molar heats oí vaporizatian
boiling
temperature
obtained
temperature
tJI. The nonnal
in this work far each substance
extrapolatian
since the experllncntal values, with exception
below 101.325 kPa.
in a11 cases.
with the Clapeyran
vaporization
2.303
(T!K
rabIe X compares
and dp/dt with 1iterature
temperatures
Differentiation
equation
derived
Tb, the slape oí the
were obtained
excellent
with the
to give an accuracy
in each case and very close to the accuracy
oí the eguatian
it
in Figs. 2 to 8, from which
far each substance
oí fD.OSo K, which is in faet the largest deviation
measurements
observed
are a150 plotted
that there is good agreement
is and
oí n-pentane,
calcu1ated
boiling
values and the agreement
oí the Antoine
allows the calculation
is
equatian
tagether
oí the molar heats oí
~:
pB
+
e)
2 _ 6H
(3)
- wr
where ~V is the difference
oí the molar volume oí the vapaur
(Vv) and
1iquid (VL) phases.
\Ie have calculated VL froID density values from the literature(17,18,19)
and Vv from the viria1 expantion.
The second virial
coeíficient
B(T) used in the 1atter equation was calculated
correlation
of Tarakad
and Danner(20)
Tabla XI campares
calculated
in order to keep cansi~tency.
and experimental
vaporization
at 298.2° K and at the boiling
The relative
error in each case is always smaller
reliability
of the calculated
It is cancluded
temperature
calibration
íor each substance.
than 3% indicating
for detenmining
in relatively
and technique
vapour-liquid
short periods
relatively large range oí pressure and temperature.
that the accuracy of the measurements may be further
rigorous
molar heats of
óH values from the fitted Antaine
that the ebulliometer
this study are very conyenient
data of pure substances
írem the
of the quartz thermomcter
the
equation.
used in
equilibrium
of time and in a
It is clear, howcver,
improved through a
at several points of the
70
temperature
range used in this work.
TABLE 1I
T/K
Pexp /kPa
T/K
/lp/?"
P
exp
/kPa
325.42
26.419
-78
340.12
52.957
-22
327.39
29.152
-45
342.30
58.347
-22
329.0b
31.659
-9
3..13.25
60.904
45
330.77
34.462
82
34.L 26
63.625
20
332.28
36.91:':
-26
345.15
66.1:>7
27
333.8~
39.773
25
346.13
68.938
-27
335.19
42.310
-1 S
3.17.Só
74.223
330.57
45.151
46
3~9.08
78.190
-3b
- S
337.76
47.067
41
Tabla n.
Experimental vapour pressure Pexp of 2-propanol and pressure
residuals
6p : P
exp - Peal; where Peal has been obtained fram
Eq.
(2).
TABLE
III
t,p/Pa
T/K
19.530
-76
351.02
53.022
52
331.1 S
26.530
-58
352.49
55.603
333.76
29.220
-41
354.01
58.347
T/K
323.15
P
exp
/kPa
P
exp
/kPa
tI'/Pa
336.28
32.051
S
355.20
bO.599
54
2b
32
338. SO
34.692
17
356.79
63.b99
23
340.34
37.017
33
357.94
65.991
-13
342.37
39.678
3
359.34
68.932
4
344.22
42.282
20
360.52
71.447
-26
346.10
45.068
38
362.10
75.000
4
3H.80
47.689
32
3b3.óS
78.538
-48
349.07
50.717
29
Table II!.
Experimental vapour pressure Pexp of 2,2,4-tr~methylpentane
and pressure residual 6p = Pexp - Peal; where Peal has been
obtained
fraro Eq. (2).
TABLE
T/K
Pexp /kPa
71
IV
6p/Pa
T/K
pexp /kPa
6p/Pa
275.06
26.653
-
7
303.53
82.991
- 6
275.21
26.800
-39
304.70
86.578
49
276.09
27.880
-25
306.15
91.137
65
277 .66
29.926
33
308.34
98.263
-28
-11
279.81
3l.789
309.06
100.750
l82.08
36.154
36
310.22
104.763
-80
283.82
38.868
27
311.29
108.680
-45
286.14
42.734
6
312.31
112.565
36
288.07
46.231
34
312.73
114. 144
292.09
54.165
10
313.17
115.797
29.L 41
59.2b7
313.99
119.029
4
296.80
64 .839
-
315.15
123.646
34
299.27
71.072
-23
Table IV.
-12
39
8
18
-22
Experimental
vapour pressure Pexp of n-pentane and pressure
residual s !'Jp = p
- PI;
where Peal has been obtained fram
exp
ca
Eq.
(2) •
TABLE
T/K
P
exp
/kPa
tlp/lla
l'
T/K
p
exp
/kPa
6p/Pa
306.98
26.392
42
323.34
55.906
- 21
309.18
29.316
8
324.36
58.418
-31
314.16
37.024
-37
325.33
60.952
16
315.76
39.918
25
326.42
63.872
36
317.07
42.342
- 4
327.29
66.235
2
318.45
45.046
- 21
328.24
68.898
-39
319.75
47.737
-27
329.21
71.806
14
320.97
50.392
-25
330.01
74.218
2
322.18
53. I 54
-16
331. 32
78.340
5
Tabla V.
Experimental vapour pressure P
exp of methanol and pressure
residuals ll.p ::P
exp - Peal; where Peal has been obtained frem
Eq.
(2) •
72
TABLE
TIK
304.76
309.86
313.51
316.34
318.54
320.70
323.75
324.96
326.40
328.07
329.46
331.15
332.67
Pexp IkPa
11.460
15.089
18.215
21.054
23.481
26.157
29.576
32.099
34.352
37.186
39.594
42.795
45.814
6p/Pa
VI
Pexp IkPa
TIK
1
12
334.08
335.48
48.816
51.921
-20
- 6
336.88
338.01
339.06
340.18
341.13
341.94
342.81
343.88
55.202
57.978
60.670
-26
14
O
II
10
62
10
35
16
63.656
66.268
68.582
71.129
74.389
77.614
77.820
344.90
344.98
6p/Pa
37
21
14
8
II
12
4
7
-15
-13
- 9
-61
Table VI. Experimental vapour pressure Pexp oE ethanol and pressure
residual ~p ~ p
- p 1; where p 1 has been obtained fram
Eq. (2).
exp
ca
ca
l'Al:L!:
-------'1'/1\
--- --_._--- ----~~---_.
P
exp
/\..1':1
29S.117
20.927
297.U3
29.339
298.87
31.793
301.Ul
34. -3.1
302.83
37 .. 19l1
304..15
30S. ib
4(J.078
-.\2. lBS
S •. 11.1
307.5'"
..¡
308.78
.1i.llS3
Table VII.
VII
--------TIK
6p/Pa
-17
- 4
40
-25
311.
p
exp
/kP3
53.078
SO
,5.693
312.77
2
17
11
-44
-
313.91
S8.249
315.18
(¡1.O70
6p/Pa
17
-36
34
-20
46
316.35
63.885
317.43
b6.487
318.43
ú8. %9
-
319.52
71. 793
322.03
78.598
- 3
-51
22
S
oE acetona and pressure
~~~~~~~~ :a~our:r:s~:ew~~~g
p
Eq.
(2).
exp
ca
cal
has been obtained
fraro
73
TABLEVIII
T/K
P
exp
/kPa
óp/Pa
T/K
Pexp /kPa
op/ll;l
303.17
15.947
9
328.90
44.759
- 7
306.08
18.101
O
332.64
51.308
101
309.75
21.158
8
334.36
5,1, 38:;
-22
312.92
24.130
-I2
335.98
57.559
315.58
-
"
26.892
- 2
337.71
61.136
318.33
29.979
-23
339.01
63.833
-32
320.42
32.548
5
340.79
67.7sn
-{l3
322.72
35.566
5
324.33
37.786
-11
341. 93
7(1.-1:,(}
344.88
77.ú();
38
,
-15
--_._------ _.
Table VIII.
Experimental
vapour pressure Pex
residuals lJp '" p
wherE
exp - PI;
ca
Eq.
(2) •
-_.
------
of benzene and pressure
p ca1 has been obtained from
TABLE IX
SUBSTA~CE
e
A
B
acetone
6.384897
1288.8889
-34.9215
bcnzene
6.048137
1222.364
-50.9121
2 - propanol
7.435340
1677.407
-46.4170
mcthanol
7.211740
1583.890
-33.4690
2,2,4 - trimethylpentane
6.146780
1386.248
-37.5840
n-pentane
6.206773
1183.321
-27.5503
ethanol
6.404289
1690.380
-38.3550
Table
IX.
Constants
of the Antaine
equation.
74
TABLE
X
(dp/dt)/kPa K-'
Tb/K
SUBSTANO,
(a)
a
1
(b)
ReL
acctone
329.24
329.28
3.472
3.403
bcn:ene
353.30
353.25
3.119
3.122
4.102
2 - propanol
355.35
355.48
4.101
mcthanol
337.71
337.66
3.992
3.992
2,2,4 - trimcthylpcntanc
372.34
372.39
2.886
2.886
3.458
4.003
n - pentanc
309.22
309.22
3.480
cthanol
351. 47
351. 48
4.023
Table
b
Re£.
X.
Comparison
of ealculated
and literature
values
point and the first differential
coefficient.
values
froro the Antaine equation
values
froro the Antaine
oi normal
1
beiling
at p = 101.325 kPa.
equation
at the normal
beiling
point.
TABI.E Xl
Ml/kJ mol-'
at 298.2 ti:
acctone
bcn:enc
31.84(17)
33.8
33.47(18)
(1 error)d
298.2 R Tb
26.9
30.44(17)
2.70
-2.84
31.1
31.06(18)
-0.98
0.13
44.7
44.98(19)
40.7
39.83(19)
0.63
2.14
mcth,¡¡nol
37.9
37.96(18)
35.9
35.92(18)
0.16
-0.06
2,2,4-trimethylpentanc
35.1
35.52(18)
30.4
31.12(18)
1. 20
-2.40
n-pcntane
26.8
26.,,(18)
26.4
25.84 (18)
-1.34
- 2.12
41.84(17)
39.1
39.42(17)
-0.85
.2-propanol
cthanol
Tabla
d
31.0
Ilfl/Kj mol-'
at lb
XI.
0.82
Comparison of ealculated and literatura values of the molar
heat of vaporization
at 298.2° K anó at the normal boiling
poiot.
ÓH
- 6Hca1
1it
(\ error) s --~~--~~
• 100
6H
ca1
75
- 0.04
K
120
100
90
-0.02
K
o
o
O
<O
•
20
O
d!.
O
"
•
O
O-
O
<J
•
O
•
-20
•
•• •
-<O
•
-60
-80
0.02
•
K
-100
-120
004
320
330
K
3<0
360
TI K
Fig. 2.
Plot ai pressure residuals ~p ter 2-propanol- frorn Eq. (2) fer
values af this work and values reported by other authors.
The
curved lines correspond to 6p vaJues ter the stated
liT
oi temperature,
, Hirata120,
Ambrose13
O,
Le.,
Ohe1l+
l.
óp ""-(dp/dT)
liT.
This work. •
valu~s
76
ea
o
O
'"
"-
O
•
a.
Q.
•
O
•
•
•
•
0_02
K
004
K
O
o
• •
•
o
K
~_O_02K
o
o
<1
•
_0_04
•
-'"
o
•
•
330
340
360
T/K
Fig. 3.
Plot of pressure residuals Óp for 2,2,4-trimethylpentane iram
Eq. (2) for values of this work and values reported by other
authors. The curved lines correspond to Óp values fer the stated
AT values of temperature, i.e. Ap = -(dp/rlT) AT. This work .,
Hirata 12 O.
77
-004
1(
"fl "
"
ro
"
+
"
11"
-002
~
"
60
40
20
•
+
•
•
."
~
•
"
• •
~
•
."
•
•
•
•
•
~"
•
• •~"
•
•
+
-EO
~"
•
+
00,"
'+"
-100
-IZO
"
..
,
•••
T/K
Fig. 4.
Plot ai pressure
residuals
óp fer n-pentane
frorn Eq.
(2) for
values of this wark and values reported by other authors.
The
curved line~ correspond to Ap valu€s fer the stated óT values
temperature,
Le. 6p'" -(dp/dT)
AT. This work ., A.P.I.1rl ,
Willingham10+, Ohe14•
af
78
140
-0,04 K
120
00
00000000
-0021(
000
60
•
20
r
°
"-O<:l
•
•
•
•
•
-20
•
•
••
•
-40
•
-60
0.021(
-80
-100
-1
0.04
1(
~
'0.
'lO
'"
TI K
Fig. 5.
PIot oi pressure residuals ~p for methanol irom Eq. (2) for values
oi this •..•••
ork and values reported by other autl'ioTs.
The curved
lines correspond to ~p values fer the stated 6T values oi
temperature.
Le.
IIp '" -(dp/dT)
liT.
This work ••
Ambrose13D.
79
120
10
80
60
40
20
&
O
"c.
"
<J
-20
-40
-60
•
-80
-100
-~O
0.04 K
-140
30'
",.
"'"
"4>
T/K
Fig.
6.
Plot of pressure residuals Óp far ethanol fraro Eq. (2) fer values
of this work and values reported by other a.uthors. ThE; curved
lioes correspond to Óp values fer the stated ÓT values of
tewperature, l.e. 6p - -(dp/dT) ÓT. This work • , Ambrose13(J
A.P.1.1
fI, Reid9 0,
Kretschmer1S X,
HOlmes16
m
80
120
-0.04
O
o
DO
K
O
O
O
80
60
40
.0
&
"-
c.
<:l
O
-'0
-40
-60
-80
-~O
0.04 K
~120
-~O
'"O
"O
'00
TI
Fig. 7.
K
Plot of pressure residuals Óp fer acetone from Eq. (2) fer values
oí this work and values reported by other authors.
The curved
lines correspond to óp values for the stated ÓT values oí
ternperature,
Le.
6p :> - (dp/dT) 6T.
This work.,
Ambrose8 D,
A.P.r.1r:!.
81
'40
"O
-004 K
,"
80
<o
l'
-002 K
"f
20
O
"-"-
o.
@I
O
<l
~
-,o
+
t
•
---------o
~ +
+
•
•
•
•
•
•
•
•
-00
•
002 K
-'"
-'0
~,~.
-'"O
-120
-""
""
Fig. 8
'"
T/K
'"
'"
PIot Di pressure residuals ~p ior benzene from Eq. (2) ior values
oi this work and values reported by other authors.
The curved
lines correspond to 6p values for the stated liT values oE temperature,
Le.
IIp = -(dp/dT)
liT. This work.,A.P.I.1
fl, Reid9G,
Willingharn10
+, Forziatti11
82
REFERENCES
1.
Selected
Values
of Properties
American Petroleum
of Hydrocarbons
and Re'ated
Compounds.
Institute Research Project 44, Pittsburgh,
4.
Pennsylvania
(1959).
Handbook of Chemistry
and Physics,
R.C. \least (editor),
The Chemical
Rubber Co., 52nd Ed., CJeveland
(1971).
N. R09alski,
K. Rybakiewicz,
and S. Na'anowski,
Ber. Bun. Phys. Chem.,
81 (1977) 1070.
M. R09alski and S. ~alanowski,
F'uid Phase Equilibria,
5 (1980) 97.
5.
A. Olivares
Fuentes,
6.
Unpublished
D. Ambrose,
results.
in:
M.L. /ole Glashan
2.
3.
A Spec;al ;st
7.
8.
Periodical
Society,
London
A. Treja
Rodrfguez,
Report
Report,
and A. Treja Rodríguez.
(editor),
Chemica'
volume 1
t
Thennodynamics,
chapter 7, The (hemical
(1973).
F-3217.2;
D. Ambrose,
J. Su~rez Cansino
C.H.S.
J. Suárez Cansino and J. P. ~lonfort. Techinal
Instituto
$prake
~~xicano
del
Petroleo,
and R. Townsend,
December
of
1981.
J. Chem. Thennodynamics,
15.
16.
17.
18.
6 (1974) 693.
R.C. Reid, J.M. Prausnitz
and T.K. Sherwood, The Properties
of Gases
_Li9uids.
Me Graw Hil] Book, CO.,3rd
Ed. (1977).
..
C.B. Ihlhngham,
W.J. Tay'or,
J.~,. P19nOCCO and F.D. ROSS1n1; Ilat. Bur.
Stds.,
35 (1945) 219.
AJ. Forziati,W.R.
Norris and F.D. Rossini,
Nat. Bur. Stds .• 43 (1949)
555.
M. Hirata.
S. Ohe and K. Nagahama, Computer aided data book of vaporliguid
eguilibria.
Kidacsha Ltd.,
Tokyo, 1975.
D. Arnbrose and C.H.S. Sprake,
J. Chem. Thennodynamics.
2 (1970) 631.
S. Ohe, Computer aided data book of vapor pressure,
Data book
publishing
Co., Tokyo (1976).
C.B. Kretsc~r
and R. Wiebe, quoted in Ref• 14.
lí.J. Holmes and M. van Winkle,
Ind. Eng. Chem.• 62 (1970) 21.
Internationa'
Critica'
Tables,
vol. S, Me Graw HITl Co., (1929).
N.B. Var9aftik,
Tables on the thermo h sica]
ro erties
of li uids and
19.
20.
C.F.
R.R.
9.
10.
11.
12.
13.
14.
gases,
John lH ley an
Spencer
Tarakad
and S.B.
and R.P.
Sans,
lnc.,
New Vor,
1
.
Adler, J. Chem. Eng. Data, ~3 (l97B)
Danner, AIChE J., 23 (1 977T685.
82.