Properties of alcohols
advertisement

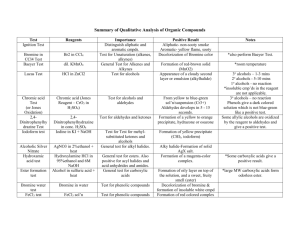
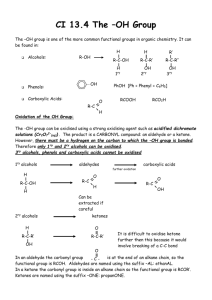
Org Chem II Laboratory (Non-Majors) The Properties of Alcohols I. Kelly INTRODUCTION Alcohols are classed into three categories: primary (1˚), secondary (2˚) and tertiary (3˚); the classification is based on the type of carbon to which the hydroxyl group is attached. If the carbon is attached to only one other carbon, it is a primary carbon. If the carbon is attached to two other carbons, it is a secondary carbon and the alcohol is a secondary alcohol and so on. H R C OH H H R C OH R' 1˚ R'' R C OH R' 2˚ 3˚ Alcohols have a much higher boiling point than ethers or alkanes of similar molecular weight. This is due to the association of alcohols in the liquid phase through hydrogen bonding. For example, the boiling point of butyl alcohol is 118˚C whereas the boiling point of the isomeric diethyl ether is 36˚C. Hydrogen Bonds R -∂ O +∂ H II. H +∂ O -∂ R Procedure Before the advent of modern spectroscopic methods, the determination of chemical properties was of paramount importance for the identification, characterization and determination of structure of pure substances. Many reagents or conditions were found to be specific with substances that contained certain functional groups. Thus these reactions can be used to confirm or deny the presence of a specific functional group. The following experiment is designed to acquaint you with the chemical properties of alcohols and to determine the structure of an unknown that you will receive. You will examine the chemical and physical properties of various alcohols. From this data you will arrive at conclusions as to the Structure-Chemical Activity relationship of alcohols. You will then draw on these conclusions to deduce the structure of an unknown alcohol sample. I. Solubility of Alcohols Because of the hydrogen bonding possible between the polar hydroxyl group of an alcohol and water, many alcohols are soluble in water. However as the carbon content of an alcohol goes up, the solubility decreases. This is often described as the 5-Carbon Rule, since the point at which an alcohol is no longer soluble in water is often (but not always) reached when it contains 5 or more carbons. However, alcohol structure (either 1˚, 2˚ or 3˚) also has an effect on solubility. PROCEDURE 1. Add each of the following alcohols drop wise to 1 mL of water. Count the number of drops needed to cause a phase separation, but do not use more than 10 drops. If you have added 10 drops of alcohol, and no phase separation has been observed, the alcohol should be classified as completely miscible in water. Record your observations as very soluble (6-9 drops), soluble (2-5 drops), or insoluble (1-drop) 1. absolute ethyl alcohol 2. n-butyl alcohol 3. sec-butyl alcohol 4. tert-butyl alcohol 5. cyclohexanol 6. Unknown II. Alcohols as a source of protons This is a test for reactive hydrogens that is hydrogens that are sufficiently acidic that they will react with sodium metal. Note that many compounds will give a reaction with sodium metal even if that compound does not have a reactive hydrogen such as alcohols do. This is often do to small amounts of water in the sample. CAUTION SODIUM IS VERY REACTIVE AND MUST BE HANDELED WITH CARE. USE ONLY SMALL PIECES AND HANDLE IT WITH FORCEPTS. SODIUM REACTS VIOLENTLY WITH WATER! CARRY OUT ALL REACTIONS IN A HOOD. IT IS NORMALLY STORED UNDER VISCOUS OIL THAT MUST BE CAREFULLY BLOTTED OFF WITH PAPER TOWELS. PROCEDURE (in the Hood) 1. Cut a small piece of sodium metal about half the size of a pea and add to a beaker containing about 50 mL of water. Do this in a hood! Record you observations on this reaction. After the reaction is complete, add 1-2 drops of phenolphthalein indicator to the aqueous solution. Write an equation for the reaction of sodium with water. 2. Cut small pieces of sodium metal about half the size of a pea and add, in the hood, to 3 mL of each of the alcohols, below, in a 6-inch test tube. Devise a method to describe the rate of the reaction (such as the rate of gas bubbles evolved over time). Test the resulting solution with phenolphthalein. Write an equation for the reaction of the alcohol with sodium. If all of the sodium doesn’t react in any one of the alcohols, transfer this remaining sodium to the alcohol that reacts fastest. DO NOT POUR THE ALCOHOL DOWN THE DRAIN UNTIL YOU ARE SURE NO SODIUM REMAINS. 1. absolute ethyl alcohol 2. n-butyl alcohol 3. sec-butyl alcohol 4. tert-butyl alcohol 5. cyclohexanol 6. Unknown III. Alcohols in Substitution Reactions (Lucas Test) This test relies on the fact that the ease of formation of carbocations from the corresponding alcohols is highly dependent on the structural features in the molecule. Treatment of the alcohol with a solution of zinc chloride in hydrochloric acid can serve as a means of distinguishing between those alcohols that are quickly and easily converted to the corresponding alkyl chlorides (via the carbocation) and those that are not. Coordination of the zinc chloride with the hydroxyl function results in the production of a sufficiently good leaving group the carbon-oxygen cleavage can now occur…if a reasonable stable carbocation can form. RO H ZnCl2, HCl RO ZnCl2 H R + Zn(OH)CL2 Cl R Cl If R is tertiary, the reaction takes place almost immediately, whereas secondary alcohols require several minutes to undergo reaction and primary alcohols do not react over a period of 15-20 min. The test depends on the observing the formation of an insoluble liquid, alkyl chloride as a cloudy suspension that later separates into a distinct upper layer. This test requires the alcohols to be soluble in the test solvent (namely water) and is thus restricted to simple alcohols below hexyl (6 carbons). PROCEDURE 1. Add 5-7 drops of each of the following alcohols to each of 6-inch test tubes and add to each test tube 2-3 mL of Lucas Reagent prepared by dissolving 16 g of test to be valid, the alcohol must be initially soluble in the Lucas Reagent. The formation of a milky or opaque solution is evidence for formation of the insoluble alkyl chloride. Note the time required for the mixture to become cloudy or to separate into two layers. Place test tubes that have not reacted after 10 minutes in a beaker of boiling water for 15 minutes. Observe any changes. Write an equation to describe the reaction, if any. 1. 2. 3. 4. IV. n-butyl alcohol sec-butyl alcohol tert-butyl alcohol Unknown Oxidation of Alcohols Primary and secondary alcohols are oxidized rapidly to acids and ketones, respectively, by Cr+6 oxidizing agents. Oxidation is readily detected by the loss of the red-orange color of Cr+6 and the appearance of green color due to the reduced Cr+3 ion. Tertiary alcohols are not easily oxidized, (however, over time in an acidic environment, they are dehydrated to alkenes). This is the chemical test that is used in the common Breathalyzer Test used to detect alcohol on the breath of drunken drivers, PROCEDURE 1. Prepare the Jones Reagent by adding 2 grams of sodium dichromate to 10 mL of water. Then add, slowly, with stirring, 2 mL of Conc. sulfuric acid. Pour 2 mL each of this solution into four 6-inch test tubes and add 2 mL of the following alcohols to different test tubes. 1. n-butyl alcohol 2. sec-butyl alcohol 3. tert-butyl alcohol 4. Unknown 2. Stir each tube and note any rise in temperature (use you hand as a guide) or change in color. Report your observations. In an acidic solution, the dichromate ion, which is usually an orange-red color, is oxidized to the Cr+3 ion. This ion is usually a dark green color but may form a dark precipitate with the carboxylate group. V. Iodoform Test for Methyl Carbinols A methyl carbinol is a 2˚ alcohol in which at least one of the R groups attached to the 2˚ carbon is a methyl group. A methyl carbinol can be easily oxidized to a methyl ketone that is then able to undergo a process known as the iodoform reaction. OH R C CH3 H O R C CH3 O R C CH2I O R C CHI2 O R C CI3 I2 as oxidising agent - OH / I2 - OH / I2 - OH / I2 - OH O R C CH3 + 2 HI Oxidation to methyl ketone O R C CH2I O R C CHI2 O R C CI3 Successive iodination leading to triiodomethyl substituent O R C O + CHI3 Formation of iodoform PROCEDURE Add 4 drops of isopropyl alcohol to 5 mL of dioxane in a test tube and shake until all the sample has gone into solution. You may need to stopper the test tube to get adequate shaking. Add 1 mL of 10% sodium hydroxide solution, and then iodine-potassium iodide solution, with shaking, until a slight excess yields a definite dark color of iodine. If less than 2 mL of the iodine solution is decolorized, place the test tube in a water bath maintained at a temperature of 60°. If the slight excess of iodine already present is decolorized, continue the addition of iodine solution with shaking until a slight excess of iodine solution again yields a definite dark color. The addition of iodine is continued until the dark color is not discharged by 2 minutes’ heating at 60°. This excess of iodine is removed by the addition of a few drops of 10% sodium hydroxide solution with shaking. Now fill the test tube with water and allow to stand for 15 minutes. Observe the results. Iodoform is a light yellow solid. Carry out the same reaction with your unknown alcohol. The iodine-potassium iodide solution has been prepared by adding 200!g of potassium iodide with 100 g of iodine to 800 mL of distilled water and stirring until solution is complete. Reaction with Carboxylic Acids (Esterification) Acid catalyzed esterification is an equilibrium in which water is a bi-product (see reaction a). If we use a concentrated acid to tie up the water produced (through heat of solvation) and add an excess of carboxylic acid, we can drive the equilibrium toward the right. After a few minutes of reaction we can convert all the excess acid to its non-volatile salt (reaction b) and the only volatile material left will be the ester. We will use this technique to form some common flavor components. RCH2OH + R'CO2 H R'CO2H + NaOH H+ RCH2OCOR' + H2 O R'CO2-Na+ + H 2O (a) (b) Procedure (IN THE HOOD) 1...Place 1 mL (≈5 drops) of acetic acid in one test tube, 1 mL of butyric acid in a second, and about 0.5 g of salicylic acid in a third. Note the different odors. 2. Add 5 drops of amyl alcohol to the acetic acid, 5 drops of ethyl alcohol to the butyric acid and 20 drops of methyl alcohol to the salicylic acid. 3. Add 2 drops of concentrated sulfuric acid to the first and second tubes and 5 drops to the third tube. 4. Place all three tubes in a beaker of water warmed to 60° and let stand for 10 minutes. 5. At the end of this time remove the tubes from the water and add 5 mL of 10% NaOH to each tube, stopper and shake. 6. The ester should float on the water solution as an insoluble oil. Note the odor in each tube. After you have recorded your results, pour the contents of each tube down the drain IN THE HOOD. Rinse all glassware well in the hood before taking anything back to your desk. ORG II LABORATORY (NONMAJORS) NAME_________________________ PROPERTIES OF ALCOHOLS DATA SHEETS Include these Data Sheets in with your report I. SOLUBILITY TESTS Alcohol ethyl alcohol n-butyl alcohol sec-butyl alcohol tert-butyl alcohol Cyclohexanol Unknown A. Drops of H2O Account for the differences in solubility of the different alcohols. II. ALCOHOLS AS A SOURCE OF PROTONS Alcohol ethyl alcohol n-butyl alcohol sec-butyl alcohol tert-butyl alcohol cyclohexanol Unknown Rate of reaction with Sodium A. Show below the general reactions of the sodium with water and the various alcohols (except the unknown). B. Explain why the order of reactivity is as you observed. III. ALCOHOLS IN SUBSTITUTION REACTIONS Alcohol n-butyl alcohol sec-butyl alcohol tert-butyl alcohol Unknown Rate of Reaction with HCl A. Show, below, the complete mechanism for the reaction of Lucas Reagent with the various alcohols (except the unknown). B. Offer an explanation of the relative rate of reaction for each of the alcohols. IV. OXIDATION OF ALCOHOLS Alcohol n-butyl alcohol sec-butyl alcohol tert-butyl alcohol Unknown A. Rate of Reaction with Jones Reagent Show a balanced equation for the reaction of the Jones Reagent with each of the alcohols. V. IODOFORM TEST A. Show a balanced equation for the reaction of isopropyl alcohol with iodine in a base solution. VI. FORMATION OF ESTERS ACID Acetic acid Butyric acid Salicylic acid A. ALCOHOL amyl alcohol ethyl alcohol methyl alcohol SMELL Show the reactions for the formation of ester for each of the following. a. Acetic acid + amyl alcohol b. Butyric acid + ethyl alcohol c. Salicylic acid and methyl alcohol VI. CONCLUSION What type of alcohol is your unknown? Give a detailed explanation.