Hormonal Control of Gametogenesis: Spermatogenesis & Oogenesis
advertisement

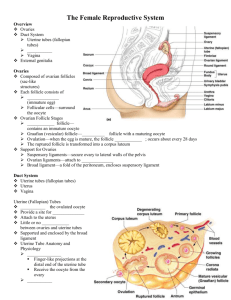
HORMONAL CONTROL OF GAMETOGENESIS © Αnastasios A. Argyriou PhD Kλινικός Εμβρυολόγος 1 I. INTRODUCTION Gametogenesis is the general process of gamete formation in both males and females. Meiosis, the process by which gametes are formed, can also be called gametogenesis, literally “creation of gametes”. The type of meiosis in male organisms from a spermatogonium to a primary spermatocyte, a secondary spermatocyte, a spermatid and finally, a spermatozoid, is called spermatogenesis, while the process of meiosis in female organisms from an oogonium to a primary oocyte , a secondary oocyte and then an ovum (egg cell), is called oogenesis. Primordial germ cells, once they have populated the gonads, proliferate into sperm (in the testis) or ova (in the ovary). The decision to produce either spermatocytes or oocytes is based primarily on the genotype of the embryo. In rare cases, this decision can be reversed by the hormonal environment of the embryo, so that the sexual phenotype may differ from the genotype. A tabular comparison of spermatogenesis and oogenesis furnishes evidence for major differences in the timing of production, number and size of gametes (Table 1) Spermatogenesis Oogenesis Number of gametes Continuous production. Although from Using up the oocytes generated before birth puberty to old age, sperm cells are Continual decrease of the oocytes, beginning constantly being engendered, the with the fetal period. production is subject to extreme Exhaustion of the supply at menopause. fluctuations regarding both quantity and quality Meiotic output Four functioning, small (head 4μm), One large immotile oocyte (d=120μm) and motile spermatozoids at the end of the three shriveled polar bodies are left at the end meiosis of the meiosis Fetal period No meiotic divisions Entering into meiosis (arrested in the dictyotene No germ cell production stage). Production of the entire supply of germ cells Table 1: Tabular comparison of spermatogenesis and oogenesis 2 A comparative outline of Oogenesis and Spermatogenesis is shown on Table 2 . OOGENESIS Oogonium (female germ cell) Germ cells committed to Meiosis ↓ Primary Oocyte ↓ Secondary Oocyte SPERMATOGENESIS Spermatogonium (male germ cell) ↓ Primary Spermatocyte First Meiotic Division First Polar Body ↓ ↓ ↓ ↓ Secondary Spermatocyte Secondary Spermatocyte Second Meiotic Division Ovum and Second Polar Body 4 Spermatids ↓ ↓ 1 Ovum (1Viable gumete) 4 Spermatozoa ( 4 viable gametes) Table 2: Differences between Oogenesis and Spermatogenesis II. SPERMATOGENESIS The male testes have tiny tubules (seminiferous tubules) containing diploid cells called spermatogonia that develop into mature spermatozoa. Spermatozoa are the mature male gametes in many sexually reproducing organisms. Thur , spermatogenesis is the male version of gametogenesis. Spermatogenesis is a complex process , during which spermatogonia (stem cells) multiply giving rise to other spermatogonia restoring their population, and to other which mature to spermatocytes. The spermatogenetic cells are grouped and form the spermatogenetic generations. All the cells which belong to the same generation, correspond to the same maturation stage, e.g. to the some stage of a cycle. . In man, six different stages per cycle are distinguished, with a duration of 16 days per stage. In order to procure mature 3 spermatogenetic cells, 4,6 cycles are necessary. Thus, the total duration of one spermatogenetic cycle in 74 days. Spermatogenesis is highly dependent upon optimal conditions for the process to occur correctly and is essential for sexual reproduction. It starts at puberty and usually continues uninterrupted until death although a slight decrease can be discerned in the quantity of produced sperm with increase in age. The basic function of spermatogenesis is to turn each one of the diploid spermatogonium into four haploid spermatozoa. This is achieved through the meiotic cell divisions (meiosis I and II). During interphase before meiosis I, the spermatogenium’s 46 single chromosomes are replicated to form 46 pairs of sister chromatids, which then exchange genetic material through synapsis before the first meiotic division. In meiosis II, the two daughter cells go through a second division, giving rise to four cells containing a unique set of 23 single chromosomes that ultimately mature into four sperm cells (spermatozoa). Thus, a male will produce literally millions of sperm every single day for the rest of his life. The spermatozoa are able to fertilize the counterpart female gamete, the oocyte, during conception to produce a single – celled individual known as a zygote. This is the cornerstone of sexual reproduction and involves the two gametes both contributing half the normal set of chromosomes (haploid) to result in a chromosomally normal (diploid) zygote. To preserve the number of chromosomes in the offspring (which differs between species), each gamete must have half the usual number of chromosomes present in other body cells. Otherwise, the offspring will have twice the normal number of chromosomes , and serious abnomalities may result . In human beings, chromosomal abnormalities arising from incorrect spermatogenesis can result in Down Syndrome, Klinefelter’s Syndrome and 4 spontaneous abortion. Most chromosomally abnormal zygotes will not survive for long after conception. The process of spermatogenesis is divided into the following phases, while a part of testis enlarged to show different stages of Spermatogenesis and spermiogenesis is shown on (Figure 1): Figure 1: 1. Multiplication phase 2. Growth phase 3. Maturation phase 4. Metamorphosis of spermatid 1.Multiplication Phase Multiplication phase is also known as Spermatocytogenesis. The sperm mother cells present in the germinal epithelium of the seminiferous tubules divide repeatedly by mitosis to form large number of diploid rounded sperm mother cells which are called as spermatogonia. Some of these sex cells move towards the lumen of seminiferous tubules 5 and enter the growth phase. These cells are called primary spermatocytes. The primary spermatocytes are diploid and contain (44+XY) chromosomes. Some of the sex cells produced by the division of spermatogonia remain in their original condition and continue to divide giving rise to primary spermatocytes. Such cells are known as stem cells. 2.Growth Phase During this phase, the spermatocyte as well as its nucleus enlarges in size. It gets ready to undergo maturation division. 3. Maturation Phase Each diploid primary spermatocyte undergoes meiosis I, which is a reduction division. Two daughter cells are formed each with ’n’ number of chromosomes. The daughter cells are called secondary spermatocytes. The secondary spermatocytes are haploid and much smaller comparatively, containing (22+X) or (22+Y) chromosomes. The secondary spermatocytes undergothe second meiotic division (equational). This results in the formation of four daughter cells known as spermatids (Figure 2). Figure 2: 6 Ιn men, there are two kinds of spermatogonia, types A and B. The nuclei of A spermatogonia do not show heterochromatin. Cells with nuclei which stain less heavily with hematoxylin are named A-pale spermatogonia, while others which stain more heavily are called A-dark (1) A-pale spermatogonia divide mitotically and give rise to B spermatogonia and following another mitosis, become meiotic spermatocytes. B spermatogonia divide and transform initially into preleptotene, and then leptotene, zygotene and pachytene primary spermatocytes. Diakinesis of secondary spermatocytes completes meiotic division and initiates spermiogenesis. A-dark spermatogonia do not divide and are quiescent. However, when the number of A-pale spermatogonia is diminished, for example after irradiation, A-dark spermatogonia become active and transform into A-pale, and thereafter start to proliferate. So, A-dark spermatogonia act as reserve of stem cells. They are renewed by redifferentiation of A-pale spermatogonia when it is needed (2) (Figure 3) Figure 3: 7 4. Metamorphosis or Spermiogenesis The spermatids formed as a result of maturation division is a typical animal cell with all the cell organelles present in it. In this form, it cannot function as a male gamete. So, many changes take place to change the non-motile spermatid into motile spermatozoan. The main aim of the changes is to increase the motility of the sperm. The changes are: • The nucleus shrinks by losing water and DNA becomes closely packed. • An acrosome is formed from the golgi complex • An axial filament of the tail of the spermatozoan is formed from the distal centriole of the spermatid • A mitochondrial ring is formed from the mitochondria around the distal centriole and is called as nebenkern • Much of the cytoplasm of the spermatid is lost and the remaining cytoplasm forms a sheath around the mitochondrial spiral. This sheath is known as manchette. During the process of differentiation, the developing sperms have their head embedded in the Sertoil cells, which are thought to provide nutrition for the developing sperm, because their cytoplasm contains large stores of glycogen which diminish as spermatids mature. There is no direct evidence for this nutrive function, but some forms of male sterility are associated with the failure to produce normal Sertoli cells. Electron microscopy has revealed distinct plasma membrances surrounding the two cell types at the points of revealed distinct plasma membrances surrounding the two cell types at the points of contact, and thus the Sertoli cell-spermatid relationship is not syncytial,as once thought. At all stages of differentiation, the spermatogenic cells are in close contact with Sertoli cells. A single Sertoli cell extends from the basement membrance to the lumen of the 8 seminiferous tubule, although the cytoplasmic processes are difficult to distinguish at the light microscopic level (Figure 4) Figure 4: Cellular events in human spermatogenesis Sertoli cells support the developing gametes in the following ways: • Maintain the environment necessary for development and maturation via the bloodtestis barrier • Secrete substances initiating meiosis • Secrete supporting testicular fluid 9 • Secrete the Androgen-Binding Protein (ABP), which concentrates high quantities of testosterone in close proximity to the developing gametes. Testosterone is produced by interstitial cells (Leydig cells), which reside adjacent to the seminiferous tubules. • Secrete hormones effecting pituitary gland control of spermatogenesis, particularly the polypeptide hormone, inhibin • Phagocytose residual cytoplasm left over from spermiogenesis • Release Antimullerian Hormone (AMH), which prevents formation of the Mullerian Duct/Oviduct Seminiferous epithelium is sensitive to elevated temperature in humans and will be adversely affected by temperatures as high as normal body temperature. Consequently, the testes are located outside the body in a sack of skin called the scrotum. The optimal temperature is maintained at 20 C (man) – 80C (mouse) below body temperature. This is achieved by regulation of blood flow and positioning towards and away from the heat of the body by the cremasteric muscle and the dartos smooth muscle in the scrotum. Dietary deficiencies (such as vitamins B,E and A), anabolic steroids, metals (cadmium and lead), x-ray exposure, dioxin, alcohol, and infectious diseases will also adversely affect the rate of spermatogenesis. The hormonal control of spermatogenesis varies among species. In humans, the mechanisms are not completely understood, however it is known that initiation of spermatogenesis occurs at puberty due to the interaction of the hypothalamus, pituitary gland and Leydig cells (Figure 5). 10 Figure 5: Hormonal interactions in the hypothalamus-pituitary-testis axis The hormones that are classicly related to spermatogenesis is the luteinizing hormone (LH), the follicular stimulating hormone (FSH) and testosterone (T). LH controls spermatogenesis via the secretion of T by the Leydig cells. (3,4,5). Testosterone mainly acts onto Sertoli cells by increasing their respronsiveness to FSH and simultaneously, inhibits the secretion of LH by the mechanism of negative feedback upon the hypothalamus and the pituitary. FSH controls the maturation of the spermatic epithelium, by acting directly on the Sertoli cells. Finally, the protein which binds to the androgens (ABP) is produced by the Sertoli cells. 11 In the absence of the pituitary gland, spermatogenesis can be initiated by FSH and T. FSH is necessary to develop the ABP production by the Sertoli cells and to develop the blood-testis barrier and other functions of these cells. Once the Sertoli function is developed, testosterone alone will maintain sperma togenesis. The yield of spermatozoa, however, is increased if FSH is present. FSH is known to increase the yield of spermatogonia by preventing the atresia of differentiating A spermatogonia. Normally, 50% of A spermatogonia can also be reduced by increased sexual activity. FSH levels in males are environmentally influenced, increased by sexual activity and decreased by inhibin. Androgens are transported from the site of production (Leydig cells) to influence the developing germ cells. ABP produced by the Sertoli cell and shed into the adluminal compartment, assists in this role as well as transporting large amounts of androgens to the caput epididymis. Synthesis of ABP is dependent on FSH stimulation but only after the Sertoli cell has been under androgen influence. Testosterone participates in the induction and maintenance of spermatogenesis, acting through Sertoli cell’ s androgen receptor (6,7) or through spermatogenetic cell’ s androgen receptor (8,9) The testis also secretes some other hormones, that participate in the regulation of spermatogenesis, but their roles are not clearly understood; one of them is estradiol (E2). Since its discovery, etradiol was recognized as “female” sex hormone. However, estrogen receptor (ER) is widely distributed in testicular cells, suggesting a role of estrogens in the regulation of testicular function. In human testis, ERb is probably the main receptor of the effect of estrogens. ERb is localized in the nuclei of spermatogonia, spermatocytes and early developing spermatids of 12 adult men (10). The demonstration of abundant ERs in human spermatozoa (11), supports the possible involvement of estrogens in the male reproductive function. Except from the above classical mechanisms of control of spermatogenesis, some other important autocrine and paracrine interactions have described between the different cellular compartments of the the testis (12). Among them, the Inhibin-b (Inh-b) and the Antimullerian Hormone (AMH) have a dinstinguished position. In man, Inh-b is mainly produced by the Sertoli cells and controls the secretion of FSH from the pituitary and consequently the spermatogenesis, via a negative feedback mechanism (13). In general, low blood concentration of Inh-b often reflects on a disorder of spermatogenesis (14). AMH is exclusively secreted by Sertoli cells and represents a precocious hormonal index of their function. Its production is influenced by transcriptional factors, testosterone, FSH and spermatocytes at prophase I (15) III. OOGENESIS Oogenesis begins soon after fertilization, as primordial germ cells travel from the yolk sac to the gonads, where they begin to proliferate mitotically. The germ cells multiply from only a few thousand to almost 7 million. They become oocytes once they enter the stages of meiosis several months after birth. Now called primordial follicles, they are made up of oogenic cells from the primordial germ cells surrounded by follicle cells from the somatic line. The oocyte is then arrested in the first meiotic prophase until puberty. At puberty, between 4 to 10 follicles begin to develop although only 1-2 are actually released. Surrounding each oocyte is a zona pellucida, membrana granulosa, and theca cell layer. Each oocyte finishes its first meiotic division, creating a secondary oocyte and polar 13 body, which serves no further function. It begins the next meiosis cycle and is arrested in its second metaphase, at which point it is released from the ovary in ovulation. It will not finish the meiosis cycle until it encounters the stimuli of a sperm. Formation of the ovum most often involves substantial increases in cell volume as well as the acquisition of organellar structures that adapt the egg for reception of the sperm nucleus, and support of the early embryo. In histological sections, the structure of the oocyte often appears random but as the understanding of its chemical and structural organization increases, an order begins to emerge. Among lower vertebrates and invertebrates, mitotic divisions of the precursor cells, the oogonia, continue throughout the reproductive life of the adult; thus extremely large numbers of ova are produced. In the fetal ovary of mammals, the oogonia undergo mitotic divisions until the birth of the fetus, but a process involving the destruction of the majority of the developing ova by the seventh month of gestation reduces the number ofoocytes from millions to a few hundred. Around the time of birth, the mitotic divisions cease altogether, and the infant female ovary contains its full complement of potential ova (Table 3) Week of gestation 3/4 5-6 8 8-20 20-40 Birth to puberty Stages Number of germ cells Primordial germ cells in the entoderm of the yolk sac Premeiotic cells: oogonia ∼0000 Propagation by mitosis Mitosis, meiosis, atresia, Maximum at week 20 Reduction of oocytes (80% of germ cells are lost) Further oocytes are lost by atresia 600000 6-7000000 1-2000000 300000 14 One important feature of oocyte differentiation is the reduction of the chromosome complement from the diploid state of the somatic cells to the haploid state of gametes. Fusion with the haploid genome of the sperm will restore the normal diploid number of chromosomes to the zygote. The meiotic divisions which reduce the chromosome content of the oocyte occur after the structural differentiation of the oocyte is complete, often after fertilization. Unlike the formation of sperm, in which the two divisions of meiosis produce four equivalent daughter cells, the cytoplasm of the oocyte is divided unequally, so that three polar bodies with reduced cytoplasm and one oocyte are the final products. Generally, each fertilized oocyte produces a single embryo, but there are exceptions. Identical twins, for example, arise from the same fertilized egg. Egg cytoplasm also contains large stores of ribonucleic acid (RNA) in the form of ribosomal messenger, and transfer RNA. These RNAs direct the synthesis of proteins in the early embryo and may have a decisive influence on the course of development. Development of the germ cells in the ovary Following the immigration of the primordial germ cells into the gonadal ridge, they proliferate, are enveloped by coelomic epithelial cells, and form germinal cords that, though,keep their connection with the coelom epithelium In the genital primordium, the following processes then take place: • Α wave of proliferation begins that lasts from the 15th week to the 7th month: primary germ cells arise in the cortical zone via mitosis of oogonia clones, bound together in cellular bridges, that happens in rapid succession. The cell bridges are necessary for a synchronous onset of the subsequent meiosis. • With the onset of the meiosis (earliest onset in the prophase in the 12th week) the designation of the germ cells changes. They are now called primary oocytes. The 15 primary oocytes become arrested in the diplotene stage of prophase I (the prophase of the first meiotic division). Shortly before birth, all the fetal oocytes in the female ovary have attained this stage. The meiotic resting phase that then begins is called the dictyotene and it lasts till puberty, during which each month (and in each month thereafter until menopause) a pair of primary oocytes complete the first meiosis. Only a few oocytes (secondary oocytes plus one polar body), though, reach the second meiosis and the subsequent ovulation. The remaining oocytes that mature each month become atretic. The primary oocytes that remain in the ovaries can stay in the dictyotene stage up to menopause, in the extreme case without ever maturing during a menstrual cycle. • While the oogonia transform into primary oocytes, they become restructured so that at the end of prophase I (the time of the dictyotene) each one gets enveloped by a single layer of flat follicular epithelial cells (primordial follicle) From birth, there are thus two different structures to be distinguished that, at least conceptually, do not develop further synchronously: • On the one hand, the female germ cell that at birth is called the primary oocyte, and which can develop further only during (and after) puberty (hormonal cycle is necessary) • On the other hand, the follicular epithelium that can develop further from the primordial follicle via several follicle stages while oocytes remain in their primary state. The developmental sequence of the female germ cells is as follows: • Primordial germ cell – oogonium – primary oocyte – primary oocyte in the dictyotene 16 The continuation of the development / maturation of the oocyte begins again only a few days before ovulation The developmental sequence of a follicle goes through various follicle stages: • Primordial follicle –primary follicle – secondary follicle – tertiary follicle (graafian follicle) Since a follicle can die at any moment in this development (=atresia), not all reach the tertiary follicle stage. Maturation of the oocyte in the dominant follicle shortly before ovulation (Figure 6). Figure 6 Stages of follicle development 17 The primary oocyte In the first week of the cycle, the maturation of the oocyte in its associated follicle depends on the progress of the maturation of the surrounding follicle cells. The fittest follicle with its oocyte becomes the dominant follicle in the second cycle week and later a graafian follicle Up to just under two days before ovulation, the maturation of the oocyte consists in its ingestion of substances (growth of the yolk) that are supplied by the surrounding granulosa cells. This exchange of substances is mediated through cytoplasma processes of the granulosa cells that are anchored through the pellucida zone at the oocyte surface. The oocyte nucleus (2n,4C) is also matured in the last days before the LH peak. Up to that point it was arrested in the extremely elongated prophase (=dictyotene) of the first meiosis (the arrested condition that has existed since the fetal period). Through the “maturation” the nucleus changes in the diakinesis (of the prophase) and prepares itself for the completion of the first meiosis which is triggered by the LH peak. With the LH peak, the following maturation steps are now triggered in and around the oocyte-up to ovulation: In the oocyte: • Termination of the first meiosis with ejection of the first polar body • Begin of the second meiosis with arrest in the metaphase • Maturation of the oocyte cytoplasme by preparing molecules and structures that will be needed at the time of fertilization. In the follicle: 18 • Τhe granulosa cells that sit just outside on the pellucida zone withdraw their processes from the oocyte surface back into the pellucida zone. Those processes were in charge of transferring substances to the oocyte • The perivitelline space forms between the oocyte and the pellucida zone. This space is necessary for allowing division of the oocyte and for harboring the first polar body formed in the division. • Loosening of the granulosa cells in the vicinity of the cumulus oophorus and proliferation of the granulosa cells. • Increasing the progesterone concentration in the follicle fluid via increased production in the granulosa cells. The results of these processes are: • The correct placement of the uterine tube infundibulum upon the ovarian surface • The rupture of the follicle wall and the flow of the follicle fluid with the oocyte into the infundibulum • The inhibition of the maturation of further follicles Termination of the first meiosis The spindle apparatus for dividing the chromosomes has formed and oriented itself radially to the cellular surface. The first polar body will arise at the spot where the spindle apparatus is anchored on the cellular surface. Further, the processes of the granulosa cells have retracted from the oocyte surface into the pellucida zone. They have released themselves from the oocyte and this leads to the formation of the perivitelline space, In this 19 space, the ejection of the first polar body takes place as a sign that the first meiosis has ended. With the end of the first meiosis, the name of the oocyte changes from primary oocyte to secondary oocyte The secondary oocyte Through the effects of LH on the granulosa cells, these have begun to loosen their cellular bonds and to multiply. They now also produce progesterone that is released into the follicle fluid. Through the separation of the homologous chromosomes in the first meiosis a haploid (reduplicated) set of chromosomes (1n,2C) is now to be found in the secondary oocyte. The first polar body also contains 1n, 2C. Via a fine cytoplasmic connection, the polar body and oocyte remain bound together following the meiotic division, similar to what takes place when male gametes are formed. The role of progesterone in the follicle fluid Progesterone has following two main tasks in the follicle fluid: • It stimulates the further maturation of the oocyte • During ovulation, it enters the fallopian tube and guides the formation of a concentration gradient for attracting the sperm cells The follicle that is about to rupture Besides the hormones, the granulosa cells also secrete an extra – cellular matrix, mainly hyaluronic acid, into the follicle fluid. Before ovulation, the follicle fluid volume increases markedly. The cumulus cell bonds loosen further. In this way, together with the enclosed oocyte, they free themselves from where they were attached to the follicular wall 20 and now swim in the follicle fluid.The wreath of granulosa cells that enclose the oocyte is called the corona radiate. The oocyte has now ended all the steps of maturation that were set into motion by the LH peak. The molecular and structural preparations for the time following the penetration by the sperm cell have now been made in the cytoplasma. A spindle apparatus (2nd meiosis) has again been able to form with the chromosomes in the equatorial level (metaphase plate). The spindle is once more anchored radially to the cell membrane near the polar body. The same processes of spindle formation also take place in the polar body. The second meiosis is arrested in this position. The final steps of the maturation, namely the freeing for the second meiosis are first completed by the secondary oocyte when the spermatozoon has penetrated the oocyte. The follicle and the oocyte are now ready for ovulation that takes place roughly 38 hours after the LH peak. The ovarian cycle At the onset of puberty, there are approximately 400.000 primordial follicles and single follicles in all stages of maturity in the ovary. Oocytes contained in the primordial follicles migrate out of the extragenital structures of the celomic epithelium into the stroma of the primary bipotent gonads as oogonia during embryonic development. These then divide further mitotically. Of the roughly 400.000 follicles that are present in the two ovaries at the beginning of sexual maturity, only around 480 reach the graafian follicle stage and are thus able to 21 release oocytes (ovulation). This number is simply derived by multiplying the number of cycles per year (12) and the number of years in which a woman is fertile (40). Cyclic ovarian function (entailing follicle maturation, ovulation, corpus luteum development and luteolysis) is regulated by the hypothalamic – pituitary system as well as by intraovarian mechanisms (16). Hypothalamus, pituitary and ovary are thereby in dynamic interaction (Figure 7). Figure 7 Μorphological and endocrinological changes during the various phases of the cycle. 22 Cyclic changes in the hormone household (hormonal cycle) governed by the hypothalamic – pituitary system are responsible for the periodicity of the ovulation. In a woman, the rhythmic hormonal influence leads to the following cyclic events: 1. the ovarian cycle (follicle maturation) that peaks in the ovulation and the subsequent luteinization of the granulose cells 2. cyclic alterations of the endometrium that prepare the uterine mucosa so fertilized oocytes can “nest” there As a rule, the ovarian cycle lasts 28 days. It is subdivided into two phases: 1. Follicle phase: recruitment of a so-called follicle and, within this, the selection of the mature follicle. This phase ends with ovulation. Estradiol is the steering hormone; normally, it lasts 14 days, but this varies considerably. 2. Luteal phase: progesterone production by the corpus luteum. The control circuit of the hormonal cycle has two essential control elements (Figure 8). 23 Figure 8 Interactions between hypothalamus, pituitary and ovary. Representation of the negative and positive feedback mechanisms 24 1. Τhe pulsatile liberation of GnRH, as well as FSH and LH 2. The long-loop feedback – effect of estrogen and progesterone on the hypothalamic – hypophysial – system Early hormonal control helps the follicle to develop and forces oogenesis to occur in a cycle in a certain time period. The control begins in the hypothalamus which produces gonadotropin-releasing hormone (GnRH). GnRH is received by receptors in the anterior pituitary gland, which responds by releasing Foliclle Stimulating Hormone (FSH) and Luteinizing Hormone (LH), in a pulsatile manner approximately every 90 minutes (17) . At the beginning of development, the granulosa cells express FSH receptors, which stimulate growth of the follicle. Theca cells express receptors for LH, which stimulates growth of the corpus luteum. Theca cells also produce androgens, which the granulosa cells convert to estrogen. Estrogen act back on the anterior pituitary gland to further FSH and LH surges, and also supports the growth of the endometrium. At some point, the dominate follicle begins to secrete inhibin, which acts back on the anterior pituitary gland to stop producing FSH. Only the dominant follicle, which is now FSH independent, will continue to grow. During further development, the granulose cells increase their FSH receptors and express LH receptors, while the theca cells increase their LH receptors. This surge in hormone reception results in ovulation. The mean interval between maximal E2 production of the Graafian follicle and maximal pituitary LH release is approximately 24 hours. Ovulation follows on average 8-10 hours later. Midcycle serum E2 concentration is approximately 250 pg/mL. After ovulation, if fertilization occurs, the corpus luteum secretes progesterone that supports the further growth of the endometrium. If, however, fertilization does not take place, then the hormone levels drop, the corpus luteum breaks down, no longer secreting progesterone, so that the endometrium sloughs off producing menstruation. It is estimated that less than 1% of all follicles reach the stage of the Graafian follicle, with 99% of follicles degenerating by apoptosis. Programmed cell death is an energy-dependent process accompanied by DNA degradation (18). 25 In addition to an adequate FSH level, survival of a follicle also depends on growth factors such as epidermal growth factor (EGF), transforming growth factor b (TGF-B), basic fibroblast growth factor (b FGF), insulin-like growth factor (IGF-I) and estrogens (19). Besides the sex steroids (estradiol and progesterone), which exert a regulatory influence on the function of GnRH producing nerve cells, catecholamines and endogenous opiates are also involved in the regulation of GnRH secretion (Figure 9). Figure 9 Inhibitory or stimulating influences on the function of GnRH neurons. GABA, gamma-aminobutyric acid; VIP, vasoactive polypeptide; 5-HT, 5-hydroxytryptamine; NA, noradrenaline(norepinephrine);OP, opioids; ACh, acetylcholine; DA, dopamine; A, adrenaline(epinephrine); E2, estradiol; P4, progesterone. 26 The corpus luteum develops out of the ruptured follicle immediately following ovulation. The most important morphological characteristic of the corpus luteum is the vascularisation of the previous avascular follicular epithelium. With its integration into the circulatory system and the expression of low-density lipoprotein (LDL) receptors, the follicular epithelial cells are able to take up cholesterol from the periphery and use it for progesterone biosynthesis. Serum progesterone values reach a peak of approximately 15 ng/mL at 6-8 days post-ovulation. Co n c l u s I o n The origin of germ cells (gametes) is of special interest because the differentiation of these cells is responsible for continuing the life cycle. The initial determination of cells as primordial germ cells occurs very early in mammals, where all of the meiotic divisions and the differentiation into oocytes occur before or just after birth, but ovulation does not take place until much later. In any case, the final production and delivery of the fully competent eggs or sperm require complex hormonal stimulation that occurs in adults, after the reproductive organs are fully mature. 27 REFERENCES 1. Clermont Y. Renewal of spermatogonia in man. Am J ANat 1966; 118: 509-524 2. De Rooij D.G., Russel L. All you wanted to know about spermatogonia but were afraid to ask. J Androl 2000; 21: 776-798 3. Lostroch A.J. Regulation by FSH and ICSH (LH) of reproductive function in the immature rat. Endocrinology 1969; 86: 438-445 4. Russel L.D., Clermont Y. Degeneration of germ cells in normal, hypophysectomized and hormone-treated hypophysectomized rats. Anat Rec 1977; 187: 347-366 5. Sharpe R.M. Intratesticular factors controlling testicular function. Biol Reprod 1984; 30:29-37 6. Bremner W.J., Millar M.R., Sharpe R.M., Saunders P.T.K. Immunohistochemical localization of androgen receptors in the rat testis:evidence for stage-dependent expression and regulation by androgens. Endocrinology 1994; 135: 1227-1234 7. Aoki A., Fawcett D.W. Is there a local feedback from the seminiferous tubules affecting activity of the Leydig cell? Biol Reprod 1978; 19: 144 8. Steinberger E. Hormonal control of mammalian spermatogenesis. Physiol Rev 1971; 51: 1 9. Sanborn B.M., Steinberger M., Meistrich M., Steinberger E. Androgen binding sites in testis cell functions as measured by a nuclear exchange assay. J Steroid Biochem 1975; 6: 1459 10. Μakinen S., Makela S., Weihua Z., Warner M., Rosenlund B., Salmi S., Hovatta O., Gustafsson J.K. Localization of oestrogen receptors alpha and beta in human testis. Mol Hum Reprod 2001; 7: 497-503 11. Aquilla S., Sisci D., Gentile M., Middea E., Catalano S., Caprino A., Rago V., Ando S.. 28 Estrogen receptor (ER) alpha and ER beta are both expressed in Human ejaculated spermatozoa: evidence of their direct interaction with phosphatidylinositol-3-OH kinase/Akt pathway. J. Clin Endocrinol Metab 2004; 89: 1443-1451 12. Bergh A. Paracrine control of Leydig cells by the seminiferous tubules. Int J Androl 1983; 6: 57 13. Anderson R.A., Irvine D.S., Balfour C., Groome N.P., Riley S.C. Inhibin B in seminal plαsma: Testicular origin and relationship to spermatogenesis. Hum Reprod 1998; 13:920-926 14. Radicioni A.F., Anzuini A., De Marco E., Nofroni I., Castracome V.D., Lenzi A. Changes in serum Inhibin B during normal male puberty. Eur J Endocrinol 2005; 152: 403-409 15. Josso N., Racine C, di Clemente N. et al. The role of anti-Mullerian hormone in gonadal development. Mol Cell Endocrinol 2003; 145: 3-7. 16. De La Iglesia HO, Schwartz WJ. Timely ovulation: circadian regulation of the female hypothalamo-pituitary-gonadal axis. Endocrinology 2005; 147: 1148-1153. 17. Lanzone A, Fulghesu AM, Cucinelli F et all. Evidence of a dinstinct derangement of opioid tone in hyperinsulinemic patients with PCOS. J Clin Endocrinol Metab 1995; 80: 3501-3506 18. .Rolaki A, Drakakis P, Millingos S et all. Novel trends in follicular development, atresia and corpus luteum regression: a role for apoptosis. Reprod Biomed Online 2005; 11: 93-103 19. Chun SY, Einsenhauer KM, Minami S et all Hormonal regulation of apoptosis in early antral follicles: FSH as a major survival factor. Endocrinoogy 1996; 137: 1447-1456. 29