Bio 121 Final Exam (Session 1) - DLST
advertisement

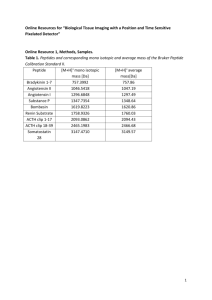
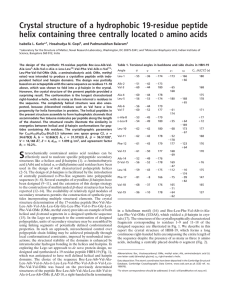
Université Joseph Fourier - Grenoble 1 Year 2012-2013 Bio 121 Final Exam (Session 1) Duration: 2h No document, no calculator, no cellular phone. Answers have to be written directly on the exam subject Total points: 100 PART C (25 points) (Time recommended - 30 min) A researcher wants to characterize the interaction between a protein A and a peptide B. The analysis of the purified protein A by acrylamide gel electrophoresis run in native conditions, shows a single band with an apparent molecular weight of 45 kDa. When the protein A is analyzed in the presence of SDS it gives this SDS-PAGE below: 1- What is the major difference between the two methods, (native versus SDS)? Justify your answer. (1.5 pt) 1 2- Describe the results of the SDS-PAGE gel and draw a schematic representation of the native structure of protein A after having estimated the size of the proteins on the electrophoresis profile. (5 pts) 3- If protein A (which is pure) had been analyzed by gel exclusion chromatography, how many elution peaks would you have expected on the chromatogram and what would be the estimated apparent molecular mass(es) ? (1.5 pt) The researcher then aims at isolating the region of protein A that interacts with peptide B. He thus attaches the B peptide (marked by a fluorescent probe on its Nterminal extremity) on the A protein using a reticulating reagent. This reticulating agent transforms any electrostatic interaction implicated in the association between two proteins, into a covalent bond. After this treatment, protein A is incubated with βmercaptoethanol, suspended in a buffer at pH 7 and loaded onto an anion exchange column. A pH gradient from pH 3 to pH 7 is then applied onto the column. The obtained elution profile is presented below. 2 wash Begining of the pH gradient wash Begining of the pH gradient 4- Describe the principle of the gel exclusion chromatography (1.5 pt) 5- Each of the 3 peaks contains a polypeptide chain X,Y or Z. Indicate the name of each peptide in the frames placed above the chromatogram peaks , knowing that pHi X = 4, pHi Y = 6 and pHi Z = 8. Comment on the basis of the given data and use a scheme for your explanation. (5.5 pts). 3 This protocol allowed the isolation of the polypeptide chain of protein A that is attached to peptide B. The tri-dimensional structure of protein A attached to peptide B is represented below: 6- Which polypeptide chain of protein A does interact with peptide B? What are your arguments to justify this conclusion? (1 pt) 7- What type of interaction (indicated by the arrows on the structural model above) does take place between the residues Asp7 and Glu 11 of peptide B and protein A, knowing that this interaction is dependent on the pH? Comment your answer (2 pts) 4 8- What kind of amino acid residues of protein A can interact with both residues Asp7 and Glu11? (1.5 pts) 9- Give the semi-developed formula of residues Asp7 and Glu11 at pH7. Would this interaction between peptide B and protein A be maintained at pH1.5? Comment your answer. (3.5 pts) Asp7 Glu11 10- What type of secondary structures do characterize peptide B and protein A? (2 pts) 5