Review for second exam:
advertisement

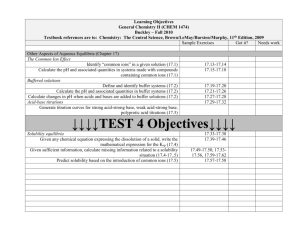
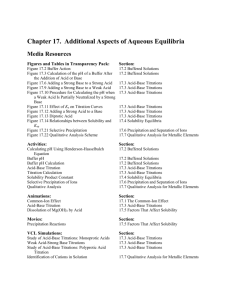
Review for Third Exam: Chapter 16 Neutralization (acid-base) reactions; strong acid-strong base; strong acid-weak base, and weak acid-strong base neutralization Buffers; definition; how buffers work; buffer reactions Calculations involving buffers The Henderson equation and its uses Titration and titration calculations; equivalence point Indicators; acid-base properties of indicators; end point of titration; choice of indicators in titrations Strong acid-strong base titration; strong acid-weak base titration; strong base-weak acid titration Relationship between pH at half-equivalence point and pKa or pKb Titration curve for polyprotic acids Soluble and insoluble Solubility product (Ksp), definition Relationship between solubility product, molar solubility, and solubility by mass Use of the solubility product in calculations Factors affecting solubility (temperature, pH, common ion effect) Solubility and the reaction quotient; unsaturated, saturated, supersaturated Selective precipitation Complex ion formation Chapter 17 Review of thermodynamics - work, heat, internal energy, enthalpy Sign conventions; state functions First law of thermodynamics Spontaneous processes; the need for a second law for thermodynamics Entropy (S), properties and interpretation of entropy Ssyst, Ssurr, Suniv The second law of thermodynamics and its meaning Finding Ssyst, Ssurr, Suniv for chemical reactions Entropy for solids, liquids, and gases Qualitative prediction of Ssyst for chemical reactons Trends in entropy based on size of molecules, dissolving substances in solvents The third law of thermodynamics Free energy (G); definition of free energy Relationship between G and spontaneous processes (for T, p constant) Method for calculating Grxn Find Teq for processes; finding the range of temperatures for which processes are spontaneous Definition of the thermodynamic reaction quotient (Q) and equilibrium constant (K) Free energy change for non-standard conditions Use of thermodynamic data to find the value for K (equilibrium constant)