Name
advertisement

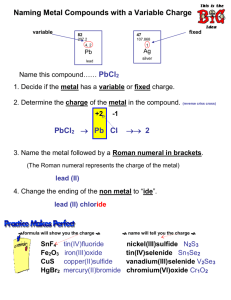
Honors WKSHT 4-4 Naming Type 2 Ionic Compounds - Notes Ionic Bond ~ Transition Metal + Nonmetal Transition metals can have more than one charge, so the name and formula has to reflect the charge of the metal in the compound name. Naming Write the name of the metal, Determine the charge on the metal cation from the anion in the formula and the ratio of anion to cation, Put the charge on the metal in roman numerals inside parentheses after the metal name, Name the nonmetal anion with “ide” ending. CoBr2 = CoBr3 = PbO = PbO2 = Cr3N2 = Cr2S3 = FeI2 = Hg3P = GaCl3 = MnO2 = MoCl2 = MnO2 = FeBr3 = CuCl = Naming Type 2 Ionic Compounds - Notes Ionic Bond ~ Transition Metal + Nonmetal Transition metals can have more than one charge, so the name and formula has to reflect the charge of the metal in the compound name. Formulas Write symbol for the metal with charge (from roman numeral!) and nonmetal with charge from periodic table. Balance charges between metal and non-metal so the compound is neutral. (or use cris-cross) Sum of charges for an ionic compound must = zero! Iron(II) oxide Iron(III) oxide Manganese(II) sulfide Lead(IV) phosphide Copper(II) Selenide Iron(III) nitride Iron(III) selenide Iron(III) fluoride Nickel (II) bromide Copper (I) oxide Mercury (I) oxide Mercury (II) phosphide