DECOMPOSITION OF WATER
advertisement

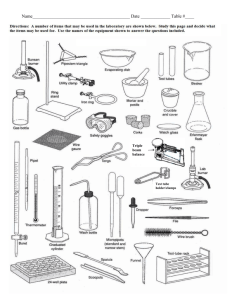
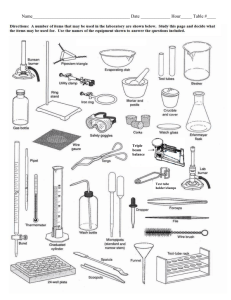
Water—What’s it made of? Today you will take the compound water and break it down into its elements (electrolysis). In order for this chemical reaction to take place you will need to add energy to break the bonds between the atoms. You will then test for the presence of hydrogen and oxygen gases. Afterwards, you will recreate water by chemically combining hydrogen and oxygen. In this experiment you will use the catalyst, NaOH (Sodium hydroxide). A catalyst is used to speed up a reaction but does not take part in it. This catalyst is a salt which, when mixed with water, will allow the electric current to pass through the water faster. Materials-get materials as you need them 600ml beaker 2 rubber stoppers for tubes matches water two equally sized test tubes goggles 2M NaOH power source w/ clips & wires wooden splint 2 electrodes test tube holder Procedure: Part A—Goggles on! 1. Fill a 600 mL beaker about ¾ full of water. 2. Fill 2 test tubes with water and seal them with rubber stoppers—NO AIR BUBBLES should be left in the test tubes! 3. Place the tubes upside-down in the beaker of water and remove the stoppers underwater! Let the stoppers sink to the bottom—you’ll need them later. 4. Carefully raise each tube slightly and place the long end of a wire electrode in each tube. Keep the mouths of each test tube underwater in the beaker while doing this so no air gets into the test tube. While holding the test tubes in one hand, carefully pour some of the water out of the beaker so it’s about half-full of water. 5. Arrange the electrodes so they are each resting on a rubber stopper on the bottom of the beaker. 6. Connect a wire to the end of each electrode that is out of the water and arrange the tubes so that no wires touch each other & that the electrodes do not touch each other. 7. Connect the opposite end of each wire to the AC power supply. Plug the power supply into a wall outlet and observe what happens. 8. Add about 50 mL of NaOH solution and slowly pour the solution down the side of the beaker so no bubbles are created. Observe how this affects the reaction. (NaOH is dangerous—do not touch it or any equipment that is wet with it). 9. Check for bubbles as they appear at the ends of both electrodes and collect in the test tubes as the water is separated into hydrogen and oxygen. If gas bubbles are not appearing, then recheck the wire connections and check to be sure no wires/electrodes are touching. 10. Wait about 15-30 minutes. You should observe the gases collecting at the tops of both test tubes. Be sure that the gas filled tubes do not float out of the water as they fill or the gas will be lost. The gases will collect at different rates. After 15 minutes, you will be able to get an approximate ratio. Answer question 1 on the lab data sheet. 11. Allow the reaction to continue until one of the test tubes is full of gas then disconnect the wires from the power source/ battery. Turn off the power source when no wires are connected to it. Observe which of the wires from the power supply was connected to it. 12. UNDERWATER…press each tube down on the rubber stopper to seal the test tube, remove from the beaker, and rinse with a slow stream of water. 13. Hydrogen Test: Take the test tube full of gas out of the water—keep it upside down! Light a wooden splint and place a burning splint into the mouth of the tube. Record the reaction AND what happens on Question 2. 14. Oxygen Test: blow the flame out from the Hydrogen test; put glowing splint into the test tube (avoid wet sides). Record the reaction AND what happens on Question 3. 15. Answer questions 3-6 on the lab data sheet. Procedure: Part B-CREATING WATER 16. Inspect test tube for breaks/cracks—Use 1 test tube! 17. To make water, repeat the experiment, placing both ends of the electrode into one test tube, being careful that the ends do not touch each other. After the test tube is over half full of gases, lift it out of the water with a test tube holder and quickly test it with a burning splint. Look for water created inside of test tube. 18. Answer questions 7-8 on the lab data sheet. Answer questions 9-10 after clean up. CLEANING UP Rinse glassware—put in dishwasher on last day Wash and dry electrodes so they do not corrode. Wrap up power supply and wires NEATLY!! Wet used matches/splints with water prior to disposal in trash can. Clear sinks of all debris. No solids in the sink. Wipe entire lab station TWICE with clean water to remove salt crystals. Do not use soap. Name ___________________ Date_____ “Water—What’s it made of?” NB Lab Sheet Part A—ELECTROLYSIS OF WATER 1. Draw the test tubes showing the volumes of the two gases: a. What is the ratio of the hydrogen to the oxygen?____________ b. How do you know which test tube contains hydrogen at this point in the lab? 2. How does hydrogen react to the burning splint? Write the reaction that occurred. 3. How does the oxygen react to the glowing splint? Write the reaction that occurred. 4. Write the balanced chemical reaction you observed in Part A. (reactants → product(s)) What type of reaction is this? (Hint: think of what you started with & what you ended with, THEN balance the equation!) 5. What type of reaction was this? Was it exothermic or endothermic? WHY? 6. Explain what conservation of mass is/means? 7. Prove mass have been conserved in this reaction? (list atoms & quantities) Part B—CREATING WATER 8. Compare the reaction of the combined gases versus the hydrogen only. Explain the difference. 9. Write the chemical reaction that took place in Part B. (reactantsproducts), then balance! 10. What type of reaction is this? Is this an endothermic or exothermic reaction? WHY? 11. What was observed inside the test tube after you put the burning splint in? 12. If you added more NaOH to the water, how would it affect the reaction? Conclusion: (For Part A & B) Describe in your own words the two reactions that took place. How are Parts A & B related?