Midterm2Preview
advertisement

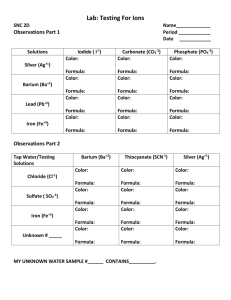
CHEM 209 Midterm 2 DATE ____________ NAME ___________________ NAvogadro = 6.02 1023 Activity Series of Metals Li>K>Ba>Ca>Na>Mg>Al>Mn>Zn>Fe>Cd>Co>Ni>Sn>Pb>H2>Cu>Ag>Pt>Au 1. Using the template below, give the electron configuration of Se in cell diagram notations 3d 3p 4p 4s 3s 2s 2p 1s 2. Name these compounds: Na2SO3 ____________________ I2O5 ____________________ Pb(NO3)2 _________________ 3. Write the chemical formulas of cadmium sulfide, lead(IV) oxide, silver nitrate, calcium carbonate, dinitrogen pentoxide, nitrite ion, nitrate ion, nitride ion, iron(III) hydroxide, potassium hydrogenphosphate 4. Compile the Lewis electron-dot formulas for the following: nitrite ion selenium trioxide 5. Identify polar molecular compound(s) out of the following: (a) SbCl5 (b) SOCl2 (c) SCl6 (d) SnCl2 For sulfite ion, SO32- : 6. Compile its Lewis electron-dot formula. 7. Determine & sketch its electron configuration: 8. Determine & sketch its molecular geometry: For potassium pyrosulfate, K2S2O7 9. Calculate its molar mass 10. Determine its mass percent composition %K = %S = %O = 11. Determine the oxidation number of sulfur in it. 12. How many moles of potassium pyrosulfate are there in 200.00 g of it? 13. Determine the molecular formula of a compound containing 37.5 % C, 12.5 % H, the rest being oxygen, if its estimated molar mass is about 60g/mol. C2H6O2 14. Determine the mass percent of nitrogen in ammonium nitrate, NH4NO3 15. How many moles of nitrogen are there in 39.0 g of calcium nitride? 16. How many moles of ions are released in a solution when 1 mole of Na2SO4 dissolves in water? 17. How many atoms are there in 3 moles of K2CO3? 18. Finish (including balancing) the chemical equation for a reaction between magnesium sulfate and barium chloride in aqueous solution. Indicate the physical state of the products. Compile the ionic & net ionic equations for that reaction. 19. Balance a chemical equation, such as: BH3 + O2 B2O3 + H2O 20. For the reaction in the previous problem determine which element(s) is (are) oxidized and reduced. 21. Finish (including balancing) the chemical equations for those reactions below that are possible. If the reaction would not go, state so ( X). Zn + H2SO4(aq) Cu + HCl(aq) Mg + CuCl2(aq) Cu + ZnSO4(aq) 22. Write the net ionic equation for the reaction between calcium chloride & silver nitrate in an aqueous solution. 23. Predict which reactions will go to completion & compile balanced chemical equation for each of them, indicating the physical state of products. Write the net ionic equations where appropriate. (a) Cobalt(III) nitrate + sodium sulfide (b) Magnesium chloride + sodium phoisphate (c) Potassium chloride + sodium nitrate (d) Sodium carbonate + hydrochloric acid (e) Copper(II) hydroxide + hydrobromic acid (g) Manganese(II) sulfate + sodium hydroxide 24. Sulfur is completely oxidized with oxygen producing sulfur dioxide. Haw many grams of sulfur are necessary to prepare 19.7 g of SO2? 25. How many moles of barium sulfate will be produced when a solution containing 0.41 mol of barium chloride is mixed with a solution containing 0.29 mol of sodium sulfate? 26. In an experiment described in the previous problem, after isolating, washing, filtering & drying the precipitate, its mass was measured as 61.32 g. What was the percent yield of that operation?