Subject: CHEMISTRY
advertisement

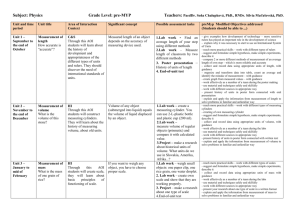
Subject: CHEMISTRY Unit: 4 – Periodic Table of Elements Teacher: Mgr. Barbara School year: 2008/2009 Kordíková LENGTH CONTENT+UQ+AOI OBJECTIVES/ ACTIVITIES OUTCOMES 14 lessons Content: Periodic law and changing properties of elements across PTE UQ: Why is PTE called periodic? What is periodicity and how is it applied in PTE? AOI: Human Ingenuity to understand and use scientific language relevant to the unit in a written lab report to formulate a simple hypothesis and explain it to design a simple investigation, materials/equipment needed, describe a method, comment on it and suggest improvements to carry out scientific investigation safely and skilfully and work effectively and resposibly as a member of a team to draw conclusions, discuss and explain the results students will revise writing a good lab report students will discuss their own formulations of a hypothesis as well as designing the investigation before the lab work students will be informed how to behave safely in a lab students will carry out the lab experiment Grade: MYP 3 ATL SKILLS how to write a lab report how to be organized in chemistry lab, time-management scientific-inquiry skills (hypothesizing, predicting explanation, planning, conducting experiment) collaboration (working in pairs) observation skills communication skills (writing lab reports) evaluation skills (using criticalthinking skills to evaluate information and make scientifically supported arguments) self -assessment analysing skills Number of lessons: 14 ASSESSMENT RESOUR CES WHAT HOW using scientific language and ability to write a good lab report ability to formulate simple hypothesis, to design a simple investigation with method, equipment needed ability to conduct the investigation safely and skilfully ability to draw conclusions and explain the results Lab work - reactivity of sodium and magnesium with water (B, D, F) Pat O’Brien: Target Science, Chemistry Tier, Oxford University Press Rose Marie Gallagher, Paul Ingram: Complete Chemistry, Oxford University Press Internet www.periodi cvideos.com describe and discuss properties of elements in selected groups and how they are applied in the society to provide scientific information in a poster clearly and effectively to demonstrate honesty when handling the data, acknowledging sources as appropriate to work effectively as members of a team collaborating and supporting others to recognize and recall scientific information relevant to the unit to explain and apply scientific information to solve problems in familiar and, with guidance, unfamiliar situations students will learn how to write a poster students will review acknowledging sources as appropriate students, with guidance, will develop their own assessment rubric students will divide their roles and responsibility within a team students will read pages 49-73 (text book) students will design and present their poster students will assess their common work students will write end-of-unit test learn how to write a poster acknowledging sources learn how to develop a good assessment rubric collaboration skills (team work) presentation skills self and peerassessment ability to provide and present scientific information clearly ability to show understanding of the information ability to appropriately acknowledge the sources ability to work effectively as members of a team knowledgeacquisition skills (understanding basic scientific concepts of bonds in covalent, ionic compounds and metals) application of knowledge self - and peer evaluation at the end of the unit knowledge the students acquire in the unit and their ability to apply it in familiar as well as unfamiliar situations Poster designing and presenting – Elements of Groups I, II, VII and VIII (A, B, F) End-of-unit test (C) On-going assessment