Document
advertisement

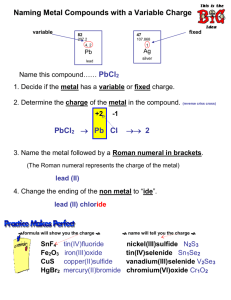
I. Naming Ionic Compounds (main group metal and main group nonmetal) Full name of metal change nonmetal ending to –ide (qwirky ones: sulfide and phosphide) 1. NaCl _________________________ 2. MgO ______________________________ 3. Sr3N2 _________________________ 4. AlF3 ______________________________ 5. Li2O __________________________ 6. CaS ______________________________ 7. K3P __________________________ 8. BeI 2 _____________________________ 9. Al2O3 _________________________ 10. Cs2Se ____________________________ 11. BaBr2 ________________________ 12. AlCl3 _____________________________ II. Write formula for Ionic Compounds (main group metal and main group nonmetal) Charge comes from the group number (+1, +2, +3, -3, -2, -1) 1. lithium iodide _______ 2. barium sulfide ______ 3. calcium phosphide _____ 4. aluminum chloride ______ 5. sodium oxide _____ 6. magnesium sulfide ______ 7. cesium nitride _____ 8. strontium fluoride _______ 9. aluminum sulfide ______ 10. rubidium oxide ______ 11. beryllium iodide _____ 12. aluminum fluoride _____ Because of d-orbitals, transition metals may form more than one type of cation. One cation will always results from losing both s-orbital electrons, for that reason MOST transition metals will form a +2 cation. For reasons known only to them, they will also lose some of their d-orbital electrons and form more cations. Iron forms an Fe2+ and an Fe3+. Transition metal cations require a roman numeral in the name to designate which charge you are talking about FeCl2 Iron (II) chloride FeCl3 Iron (III) chloride III. Write formula for Ionic Compound (transition metal and main group nonmetal) Cation charge comes from roman numeral in name, anion charge from group number. Do not put roman numeral in formula!!! 1. copper (II) oxide _____ 2. vanadium (III) fluoride ____ 3. Iron (II) sulfide ____ 4. scandium (III) nitride _____ 5. chromium (IV) iodide ____ 6. Manganese (VII) bromide _____ 7. Cadmium (II) phosphide _____ 8. molybdenum (III) oxide _____ 9. Nickel (II) chloride _____ 10. copper (I) sulfide _____ 11. niobium (III) fluoride ______ 12. palladium (II) phosphide _____ → Exceptions: zinc only forms +2 and silver only a +1 so they do not require roman numeral. Lead, Tin, and Bismuth are called “post transition heavy metals”, and they are ONLY main group metals that form two cations, so they must have roman numerals Silver chloride, zinc oxide, lead (II) chloride, Tin (IV) bromide, bismuth (V) nitride IV. Write the name of ionic compound (transition metal with main group metal) exceptions included this time!!! Determine the charge of transition metal by how it is bonded to the main group nonmetal. Example: CuCl 2 since ionic compounds are neutral, the copper must be +2 because I have two Cl- ions. So the name is: Copper (II) chloride 1. FeI2 _______________________ 2. VP ___________________________ 3. CrO _________________________ 4. Cr2O3 _________________________ 5. Fe2O3 ________________________ 6. FeO ___________________________ 7. MoBr3 ________________________ 8. MoBr2 ___________________________ 9. AgCl _________________________ 10. ZnO _____________________________ 11. PbCl2 _______________________ 12. PbI4 _____________________________ Polyatomic ions (PAI) list on page 226 of textbook you will be responsible for memorizing and knowing the rest of year! Here is a partial list to help us with the rest of the worksheet: NO 2- nitrite, NO3- nitrate, SO32- sulfite, SO42- sulfate, PO33- phosphite, PO43- phosphate V. Ionic compound name/formula (main group metal with PAI as well as transition metal with PAI) 1. sodium nitrate ________ 2. LiNO2 _________________ 3. calcium sulfate ____________ 4. CaSO3 ________________ 5. Aluminum phosphate ________ 6. AlPO3 _________________________ 7. potassium sulfite _______ 8. Cs2SO4 ________________________ 9. magnesium nitrate _____________ 10. Ba(NO3)2 _______________ 11. aluminum sulfate _____________ 12. Al2(SO3)3 _______________________ 13. iron (II) nitrate _________ 14. CuSO3 ______________________ 15. chromium (III) sulfate _____________ 16. silver nitrate ___________ 17. Zn3(PO4)2 _____________________ 18. lead (II) sulfate ________________ 19. Bi(NO3)5 _________________ 20. scandium (III) phosphite ______ 21. Ni(NO2)2 ____________________ V. Mix it up randomly write name or formula of any combination from parts I-V (remember main group anions end in –ide PAI anions end in –ite or –ate) Sodium chloride CaO CuSO3 Silver sulfide Iron (II) nitride CaBr2 Ni(NO2)2 Magnesium iodide Lead (II) sulfate Li2O BeO Aluminum nitrate Strontium chloride Cu3(PO4)2 NaCl Calcium sulfide Copper (II) nitrite VCl3 PbO Bi3N2 AlPO4 Cesium phosphate Bismuth (V) fluoride Lithium nitrate PAI Chart you will need to memorize (posted on web page): All have 1- charge NO2 nitrite NO3 nitrate ClO hypochlorite ClO2 chlorite ClO3 chlorate ClO4 perchlorate MnO4 permanganate C2H3O2 acetate HCO3 hydrogen carbonate (also called bicarbonate) HSO4 hydrogen sulfate H2PO4 dihydrogen phosphate All have 2- charge CO3 carbonate SO3 sulfite SO4 sulfate HPO4 hydrogen phosphate CrO4 Cr2O7 C2O4 All have 3- charge PO3 phosphite PO4 phosphate