Tums, Water and Vinegar
advertisement

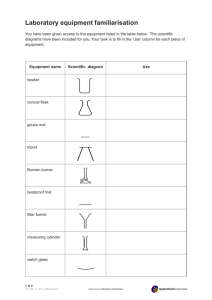
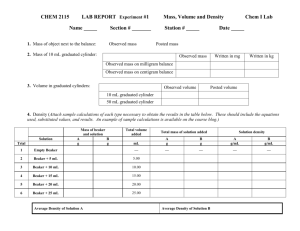
Name: ___________________ Class M2 (0 ) Date: ____________ Partner(s):____________________________________ LAB – TUMS, WATER AND VINEGAR Question: How will a Tums tablet react with water compared to vinegar? Prediction (Hypothesis): I think the Tums tablet will__________________ _________________________________________________________ because ___________________________________________________ _________________________________________________________. Materials: 30 mL vinegar in beaker 30 mL water in beaker pH paper and pH scale mortar and pestle 1 Tums Directions: 1. Put 30 mL of water in a beaker and put 30 mL of vinegar in a second beaker. 2. Dip pH paper in each beaker and compare to pH chart. 3. Crush a Tums tablet in the mortar. Place the powder on a paper towel. Do the same with another Tums tablet, keeping each pile of powder separate. 4. Get stopwatch ready. At the same time, drop one pile of Tums powder into each beaker. Gently swirl each beaker to mix the powder and liquid. Observe what occurs in each beaker. Record how long each fizzes. 5. Dip pH paper in each beaker and use the pH chart to determine the pH number. Observations: 1. Draw and label before and after pictures – 4 pictures total. 2. Write the pH under each of the 4 pictures. 3. Also under each picture, explain in words what happened. Including, which bubbled most and which bubbled longest. 4. Describe what is left in each beaker. Conclusion: 1. Create an outline for a paragraph that answers these questions: What’s an antacid? What’s it used for? How do you think it works? Why do you think the tablets reacted the way they did with the water and the vinegar? Was your prediction correct or incorrect? 2. Write the paragraph using complete sentences.