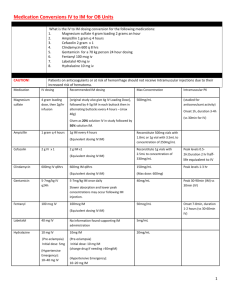
3.3 process for remote dosing program
advertisement

Proposal for Remote Dosing Program 1 PROPOSAL FOR REMOTE DOSING PROGRAM By Dr Clive Stack and Dr Gwenyth James Humanitas Trust, Tasmania © STACK & JAMES 2010 Proposal for Remote Dosing Program 2 Table of Contents ACKNOWLEDGEMENT ............................................................................ 2 1. INTRODUCTION ........................................................................ 3 1.1 METHADONE TREATMENT IN AUSTRALIA .......................... 5 1.1.1 HISTORY OF METHADONE TREATMENT IN AUSTRALIA .... 5 1.1.2 THE AUSTRALIAN METHADONE PROGRAM ........................ 5 2. PROBLEM IDENTIFICATION ................................................ 11 2.1 STATISTICAL ANALYSIS OF DRUG DEPENDENCE ....... 11 2.2 ECONOMIC ISSUES ............................................................... 14 2.3 COST TO PATIENT OF METHADONE MAINTENANCE THERAPY ................................................................................ 17 2.4 SUPERVISION OF METHADONE TREATMENT .................. 17 3. PROPOSED REMOTE DOSING PROGRAM .......................... 19 3.1 INTRODUCTION ..................................................................... 19 3.2 AIMS OF PROGRAM .............................................................. 19 3.3 PROCESS FOR REMOTE DOSING PROGRAM .................. 20 3.3.1 CRITERIA FOR INVOLVEMENT IN REMOTE DOSING PROGRAM ............................................................................... 20 3.3.2 INITIAL ASSESSMENT ............................................................ 21 3.4 THE DOSING MACHINE ........................................................ 27 3.5 DUTIES OF PARTICIPATING PHARMACISTS ..................... 29 3.6 PATIENT AGREEMENT ......................................................... 29 3.7 COMPUTER NETWORK ........................................................ 31 3.8 ADVANTAGES OF REMOTE DOSING PROGRAM .............. 31 3.9 DISADVANTAGES OF REMOTE DOSING PROGRAM ........ 35 4. PRELIMINARY RESEARCH ................................................... 36 4.1 TRIAL OF REMOTE ASSESSMENT TECHNOLOGIES ....... 36 4.2 TRIAL OF DOSING MACHINE ................................................ 37 5. PROPOSED RESEARCH – PILOT STUDY .......................... 38 5.1 AIM........................................................................................... 38 5.2 METHODOLOGY ..................................................................... 38 5.3 RESOURCES .......................................................................... 40 5.4 PROPOSED BUDGET ............................................................. 40 6. CLINICAL TRIAL ..................................................................... 41 7. APPENDIX 1 – MEDICAL TERMS ........................................... 43 7.1 OPIOIDS .................................................................................. 43 7.2 METHADONE .......................................................................... 43 8. APPENDIX 2 - LITERATURE REVIEW .................................... 45 REFERENCES ..................................................................................... 47 ACKNOWLEDGEMENT The assistance of Dr Tim Gale, Department of Engineering, University of Tasmania in development of the dosing machine, pupillometry goggles and remote assessment software has been fundamental in the development of the proposed remote dosing program. © STACK & JAMES 2010 Proposal for Remote Dosing Program 3 1. INTRODUCTION Over the last 50 years, opioid dependence has been steadily increasing in Australia. Drug replacement programs, or pharmacotherapy programs, are the most common treatment for opioid dependence with guidelines for the Australian Methadone Program being established in 1985. There are currently over 41,000 people in pharmacotherapy programs for opioid dependency, from illicit and, increasingly, also therapeutic origin. This number is rising steadily. However, the Australian Methadone Program 1 imposes significant costs on all participants: prescribing medical practitioners, dispensing pharmacists and the participants themselves. Under the current program, participants are usually required to attend a dispensing centre (usually a pharmacy or a medical clinic) once a day where they are assessed by the dispensing clinician (usually the pharmacist or medical practitioner). The dispensing clinician is required to assess the patient prior to them receiving their medication to ensure they do not display signs of sedation (e.g. from other drug use) or withdrawal. They are also required to dispense the medication and supervise the patient taking it. Depending on the work load at the dispensing centre, it may take several hours for the patient to receive their medication. Patient costs particularly relate to daily attendance at the dispensing centre such as: travel, time spent waiting for medication and issues relating to contact with other drug users also waiting to receive medication at the dispensing centre. Some patients who meet stability criteria are allowed to receive takeaway medication doses from the pharmacy to be taken at a later time away from the dispensing centre. The takeaway policy differs significantly between States and Territories with the most liberal policy in Tasmania where 3 takeaway doses a week may be authorised by the prescribing medical practitioner. However, the takeaway dose system still imposes significant costs on the prescribing medical practitioner/dispensing pharmacist particularly in relation to investigating and instigating requests for changes to the takeaway dose prescription. In addition, diversion of medication to the black market and misuse of medication are significant problems. The Remote Dosing Program has taken into consideration the issues with the existing pharmacotherapy programs for opioid dependence, including the methadone maintenance program, and proposes a new addition to the program using innovative new technology. It is believed the pre-dose assessment in the new program will be more rigorous than the current system and the dose dispenser more secure. This will result in both cost savings to 1 Other forms of pharmacotherapy such as treatment with buprenorphine also follow the guidelines of the Australian Methodone Program. © STACK & JAMES 2010 Proposal for Remote Dosing Program 4 doctors and pharmacists and a safer, more secure drug replacement program that facilitates faster rehabilitation of drug dependent patients. Regardless of the costs and issues with pharmacotherapy programs, they have been found to be a cost-effective and safe way of managing opioid dependence. There is substantial research indicating an extension of the program is warranted particularly if the issues regarding diversion of takeaway doses can be addressed. Over a period of approximately 20 years Dr Clive Stack has accumulated considerable clinical experience through work in the community including treating patients in both hospital and private practice in Tasmania. During this time, he has identified many shortcomings in the current programs to manage and rehabilitate drug dependent patients. He has subsequently developed the Remote Dosing Program to address these problems. There are three major elements to the Remote Dosing Program. The first is a remote objective, reproducible and cheap pre-dose assessment of the patient to determine their level of stability. The second is a secure dosing machine, continuously remotely monitored to reduce the harm usually associated with home dosing. The third is remote dispensing of medication and supervision of the patient by an assessor to ensure the medication is taken correctly at the same time as dispensed. The program is aimed at people already stable on existing pharmacotherapy programs for opioid dependence, and introduces new technology to remotely assess patients and dispense medication. The technology relies on internet web cameras to allow the dosing clinician to contact and observe the patient as well as assess the patient using a simple reaction test and pupillometry (which measures changes in the size and reactivity of the patient’s pupil to light). If the patient passes the assessment, the medication is dispensed through a dosing machine which can only be unlocked via an internet connection with the dosing clinician. It is anticipated the Remote Dosing Program will not only result in cost savings to prescribing medical practitioners and dispensing clinicians but will contribute also to a safer, more secure pharmacotherapy program for opioid dependence. In addition, the Remote Dosing Program has significant advantages for patients particular the potential to provide split dosing throughout a day and to eliminating the need for daily visits to a dispensing centre to receive medication. This may facilitate a more productive contribution to the community. For example, the Remote Dosing Program will accommodate the patient’s medication needs allowing them to return to employment or study. In addition, the program can be extended to © STACK & JAMES 2010 Proposal for Remote Dosing Program 5 communities that are currently unable to access pharmacotherapy programs such as remote or rural communities. It should be noted that the development of the sedation assessment tools that can be undertaken remotely has many other possible uses of great community significance. For example, a simple, quick, objective and reliable tool for assessing sedation has obvious uses in policing and in industries where undetected sedation has safety implications such as flying planes, driving or operating machinery. 1.1 METHADONE TREATMENT IN AUSTRALIA 1.1.1 HISTORY OF METHADONE TREATMENT IN AUSTRALIA Methadone is a synthetic opioid that was first developed in Germany in 1937 for the relief of pain. It was first used to treat heroin dependency in the USA in 1964 and introduced into Australia for the same purpose in 1969. Methadone maintenance was endorsed as an appropriate and useful method for treating heroin dependency at the launch of the National Campaign Against Drug Abuse in 1985. In the 1980s concern about the spread of HIV/AIDS amongst injecting drug users (and from them to the rest of the community) increased. Concurrently, evidence emerged of the role of methadone in reducing the spread of HIV/AIDS amongst those who use illegal opioids. From this time, there has been a steady and substantial increase in the number of people receiving methadone treatment in Australia. In addition, there has been an increasing reliance on the private sector (medical practices and dispensing pharmacies) for the provision of methadone services. Currently, State, Territory and the Commonwealth Governments all contribute to the funding of methadone treatment services. However, direct service provision is a State/Territory responsibility. 1.1.2 THE AUSTRALIAN METHADONE PROGRAM National guidelines for methadone treatment were first endorsed by the Australian Health Ministers' Conference in 1985 and subsequently reviewed in 1987 and 1991. The guidelines were further reviewed in 1997. These guidelines are the framework for policies and procedures for methadone treatment throughout Australia. © STACK & JAMES 2010 Proposal for Remote Dosing Program 6 The objectives of methadone treatment, as identified in the National Guidelines, are: to reduce harmful opioid and other drug use to improve the health of clients to help reduce the spread of blood-borne communicable diseases associated with injecting opioid use to reduce deaths associated with opioid use to reduce crime associated with opioid use to facilitate and improve social functioning. From this, it should be noted that the goal of methadone treatment in Australia is to reduce the health, social and economic harm, associated with unsanctioned opioid use, to individuals and the community. While abstinence from all opioid use is seen as a desirable outcome of the methadone program, it is not one of the objectives. The national treatment protocols enable the majority of opioid dependent people to be maintained safely on methadone. These protocols include the following 6 steps. 1. Suitability for Methadone Treatment Admission into a methadone treatment program is usually voluntary. Methadone treatment is suitable for people who are opioid dependent as indicated by: neuroadaption as evidenced by tolerance and withdrawal on cessation or sudden reduction in the use of opioids the use of opioids or other drugs to avoid withdrawal continued desire to use opioids regardless of persistent or recur rent problems associated with their use narrowed repertoire of behaviours associated with or fixated on opioid use repeated unsuccessful attempts to reduce or stop opioid use priority of opioid use over other life activities. 2. Assessment for Methadone Treatment A person is only admitted into the methadone program after an approved medical practitioner has assessed them as being appropriate for treatment. The assessment examines the client's needs, determines their suitability for treatment and the setting in which this is best to take place (e.g. a dispensing pharmacy or specialist dispensing setting) and establishes a treatment plan. It includes the collection of relevant medical, social and personal details including drug use history, risk taking behaviours associated with drug use, observation of clinical symptoms related to drug use including intoxication or © STACK & JAMES 2010 Proposal for Remote Dosing Program 7 withdrawal and examination for the purposes of detecting evidence of drug use. In addition, the person's identity must be confirmed. Following the assessment, the person is eligible for entry into the program. However, entry into a methadone program is predicated on this being the best treatment for the patient’s condition and social circumstances. If this is agreed, entry into the program occurs as quickly as possible. Research indicates that successful commencement and continuation of treatment is improved by prompt access to programs. 3. Induction into Treatment The medical practitioner who conducts the assessment is responsible for providing each client with information about the purpose, methods, demands, risks and inconveniences of methadone treatment. Information is also provided on: the policies and procedures of the treatment program addictive qualities, side effects and drug interactions of methadone hazards and problems associated with the use of methadone (e.g. risk of excessive sedation in early stages of the program) information about any other health risks identified such as management of hepatitis or HIV/AIDS alternatives to methadone treatment. 4. Dosing Usually, methadone is dispensed as a syrup. Physeptone tablets are dispensed in exceptional circumstances instead of syrup. The medical practitioner who undertakes the initial assessment prescribes the dosage of methadone and prescribes the way in which it will be delivered to the patient (e.g. all supervised doses). The aim of treatment is to arrive at an effective maintenance dose where drug elimination and the rate of drug administration are balanced, using safe dose increments. The dose of methadone is increased slowly until the effective maintenance dose is identified unless the medical practitioner identifies signs of toxicity or intoxication. The initial dose of methadone is based on the person's history of opioid use including quantity, frequency, and route of administration. A person maybe considered for a supplementary dose if they re-present with withdrawal symptoms at least 4 hours after the initial dose on the first day of treatment. However, cases for supplementary treatment need to be assessed by a medical practitioner. Generally, the initial dose of methadone is 20 -30mg but it is often gradually increased to 60mg (or more). It has been found that people receiving 60mg (or more) are less likely to use other opioids and remain in the treatment program than people receiving a lower dose of methadone. © STACK & JAMES 2010 Proposal for Remote Dosing Program 8 Once the maintenance dose of methadone has been established, i t can only be changed by a prescribing medical practitioner. Each time a prescription for methadone is provided or renewed, the prescribing medical practitioner must assess the patient's progress. Supervised dosing is an essential component of the methadone program. Usually, methadone is taken each day at a location approved by the State of Territory jurisdiction under direct supervision. Generally, the location is an approved dispensing pharmacy under the direct supervision of the pharmacist but a medical practice may also be approved with supervision provided by the practice nurse or general practitioner. If a patient misses a methadone dose, it may be an indicator of instability. This instability could take the form of using other drugs to alleviate their discomfort. This action could result in a memory disturbance or significant sedation so patient does not get to the dispensing pharmacy for their usual dose. Alternatively, there may be social factors preventing the patient from getting to the dispensing pharmacy (e.g. car breakdown). Therefore, it is necessary to assess the person for stability and intoxication before dosing recommences. This is usually done by the dispensing pharmacist. However, if the patient does not take their methadone for three consecutive days, they cannot recommence the program until assessed by a medical practitioner. This assessment is necessary as the patient’s tolerance to methadone may be reduced and their stability needs to be reassessed to ensure the best dosing arrangements for their wellbeing. 5. Monitoring of Drug Use (Other than Methadone) Concurrent use of other drugs by people in the methadone program is common. These drugs may include heroin, amphetamines, alcohol or prescribed medication. It is necessary to attempt to detect and monitor the use of other drugs as the use of other drugs may lead to toxicity or indicate that the patient is at risk of diverting their methadone dose for payment. Such actions can put other people at risk (i.e. by making prescription medication available on the black market). Detection and monitoring of other drug use is done at the time of supplying methadone by the dispensing clinician (through observing the person) as well as by the prescribing medical practitioner (through case history, physical examination, blood and urine tests etc). 6. Takeaway Methadone Doses Where a patient meets certain criteria, the prescribing medical practitioner may authorise either a single or regular dose of methadone to be taken and consumed away from the dispensing location (takeaway doses). Takeaway © STACK & JAMES 2010 Proposal for Remote Dosing Program 9 doses are an intervention to assist in rehabilitation and normalisation of the patient’s lifestyle (e.g. so they can attend work or further education). There are significant risks associated with takeaway doses including: methadone diversion and involvement in drug trafficking injection of the dose with the risk of overdose, toxicity (methadone syrup contains sorbitol which may be toxic if injected), bacterial infection or spread of blood-borne viruses injection of the dose which may result in increased tolerance to the medication accidental overdose/death of the person (if they take the doses too close together) or other people (if accidentally taken by other people such as children). Therefore, to be eligible to receive takeaway doses, the patient must meet stability criteria including: the patient has been in continuous methadone treatment for 2 months where all doses of methadone have been consumed under supervision takeaway doses should be the same dose normally dispensed the patient is assessed as stable in that they: o are not engaged in continuing, hazardous use of opioids, benzodiazepines, alcohol and/or psychostimulants o have regular, reliable contact with the clinic and pharmacy and comply with the requirements of the methadone program o have functioning social behaviours the patient has not shown any of the following contraindicators for takeaway doses: o o diversion of methadone doses recent injection of methadone o serious psychiatric illness the prescribing medical practitioner has assessed the patient, their past performance and progress in methadone treatment and their reason for requesting takeaway doses. Patients who meet the stability criteria are considered “stable”. Those who do not meet one or more of the criteria are considered “unstable”. It should be noted there are often significant difficulties in distinguishing stable patients from unstable patients and an objective, easy to administer and cheap test that can be applied in every situation is not always available. Each takeaway dose is supplied in a separate, child-resistant container. If a patient reports a takeaway dose has been lost, stolen or damaged it will only be replaced at the prescribing medical practitioner's discretion. If the prescribing medical practitioner has reason to believe the dose has been © STACK & JAMES 2010 Proposal for Remote Dosing Program 10 already consumed or sold, they may choose to require the patient to return to supervised dosing. A patient who is approved for takeaway doses will gradually have the number of takeaway doses they may receive increased in response to their level of stability. In addition, the prescribing medical practitioner will consider whether takeaway doses assist the achievement of a stable lifestyle particularly in relation to whether the takeaway doses will support the patient’s normalisation of their social functioning. Initially they may get one takeaway dose per week to be taken on a particular day. As patient demonstrates their ability to manage their medication, their takeaway privileges can be increased until they have full privileges: 3 takeaway doses per week to be taken on any day. (On the days they do not have a takeaway dose they must attend a pharmacy or dispensing clinic and received a supervised dose). Patients may request takeaway doses if travelling overseas. Generally, takeaway doses will only be provided for the shortest possible time f or the patient to reach their destination. Physeptone (methadone) tablets rather than syrup is provided in some states. The following guidelines apply to takeaway doses for overseas travel: if a methadone program is available at the destination, transfer t o that program is preferable to takeaway doses the destination country must allow entry to people in possession of prescribed methadone medication the State/Territory jurisdiction must approve the provision of the takeaway doses giving consideration to public and personal safety issues. © STACK & JAMES 2010 Proposal for Remote Dosing Program 11 2. PROBLEM IDENTIFICATION 2.1 STATISTICAL ANALYSIS OF DRUG DEPENDENCE Opioid dependence includes people dependent on prescribed opioids, such as morphine, as well as illicit drugs such as heroin. It is estimated that 40 % of people who suffer chronic pain are dependent on opioid medication. In Australia, people with opioid dependence have been treated using opioid pharmacotherapy. The number of pharmacotherapy patients has been steadily increasing since 2006 but an increase of approximately 2,500 patients in 2008 was more than in any of the previous three years. At 30 June 2008 there were 41,347 patients in Australia receiving some form of pharmacotherapy treatment for opioid dependence. In Australia, the pharmacotherapies for opioid dependency currently used include: Methadone Hydrochloride – This drug has been used as a pharmacotherapy since 1969. It is a synthetic opioid agonist primar ily used in maintenance therapy. Methadone is taken orally usually on a daily basis. Buprenorphine – This drug has been used as a pharmacotherapy since the 1980s. It is a partial opioid agonist with high receptor affinity and has actions similar to full agonist drugs in that it can prevent withdrawal effects. The major benefit of using buprenorphine is that an increase in the dose of buprenorphine does not correspondingly increase the side effects such as respiratory suppression or sedation. This is significantly different to methadone where the side effects continue to increase with increases in dosage levels. This often allows buprenorphine to be dosed every second or third day rather than daily as the body can be safely loaded with the drug so its duration of effect can be extended. Buprenorphine is taken sublingually. Buprenorphine/Naloxone –The combination of two the drugs, buprenorphine/naloxone is also taken sublingually. The naloxone is not absorbed if taken this way and therefore has little impact on the users when taken as prescribed. However, if the combination is illicitly injected, the naloxone will attenuate the effects of the buprenorphine and other opioids. Therefore, the addition of naloxone to buprenorphine will help to discourage the injection of the drug combination and so reduce the harm related to such injection. © STACK & JAMES 2010 12 Proposal for Remote Dosing Program The number of people receiving pharmacotherapy treatment for opioid dependence has only shown a slight increase over the last 3 years, as indicated by the figures below. This compares with an increase of over 50% in patients accepted into treatment between 1998 and 2004. Table 1 – Number of People Receiving Pharmacotherapy Treatment for Opioid Dependence in Australia 2005-2008 YEAR NSW VIC QLD WA SA TAS ACT NT AUSTRALIA 2005 16,460 10,753 4,470 2,883 2,857 588 764 183 38,937 2006 16,355 10,736 4,637 2,888 2,823 602 790 134 38,965 2007 16,348 11,051 4,309 2,822 2,834 600 765 114 38,843 2008 17,168 11,821 4,899 2,908 3,052 588 786 125 41,347 When the mode of pharmacotherapy treatment is disaggregated, it is found that methadone was the most common form of pharmacotheraphy treatment in 2008. Over 60% of patients in most States/Territories are prescribed methadone alone with methadone being the pharmacotherapy treatment most likely to be prescribed in TAS, NSW and the ACT. Figure 1 – Comparison of Pharmacotherapy Treatment for Opioid Dependence in Australia: 2008 In addition, VIC, QLD and WA indicated that Buprenorphine/Naloxone was administered more often than Bupronorphrine alone. (NSW did not record any buprenorphine/naloxone as these figures were included in the buprenorphine only figures.) © STACK & JAMES 2010 Proposal for Remote Dosing Program 13 Figure 2 – Comparison of Pharmacotherapy Treatment for Opioid Dependence by State/Territory: 2008 The place where a patient receives treatment (dosing point sites) is also significant. In 2008, pharmacies dispensed treatment to nearly 70% of patients. In addition, nearly 87% of patients receiving buprenorphine/naloxone in 2008 received their treatment from a pharmacy compared with only 66% of people receiving methadone treatment. (In Figure 3, “Other” includes correctional settings.) Figure 3 – Dispensing Location for Pharmacotherapy Treatment for Opioid Dependence in Australia: 2008 Each jurisdiction has a registration process which authorises a medical practitioner to prescribe pharmacotherapy drugs. In 2008, there were 1,393 practitioners nationally authorised to prescribe pharmacotherapy drugs. However, it is significant to note that over 65% of patients received treatment from a private practitioner (i.e. working in private practice rather than a government funded setting such as a hospital or clinic). © STACK & JAMES 2010 Proposal for Remote Dosing Program 14 The geographical coverage of the dispensing centre is also critical. In the USA it was found that 85% of heroin addicts did not have access to legal methadone. While the exact figures in Australia are unknown, it would be reasonable to assume that there are insufficient authorised medical practitioners to fully cover all Australian communities with remote or rural areas most likely not to have access to pharmacotherapy programs for opioid dependence. 2.2 ECONOMIC ISSUES Opiate dependence imposes a significant economic burden on society in terms of costs related to treatment and prevention services, other health costs, work absenteeism of addicts, productivity losses from premature death of addicts and social welfare expenditure. The National Policy on Methadone Treatment identifies the following issues associated with illegal opioid use which contribute to health, social and economic costs to the individual and community: significant risk of dying from overdose quantifiable costs relating to illicit drug use in Australia were estimated at $1,700M in 1992 while the tangible social costs of drug use in Australia in 1998-99 were estimated to be $18.3B (or approximately 5.5% of the gross domestic product) law enforcement costs relating to illegal drugs were estimated at $450M in 1992 infectious diseases such as HIV/AIDS and hepatitis B and C are readily transmitted through shared injecting equipment. The estimated lifetime treatment costs to Australian public funds for treatment care, social security etc of HIV/AIDS was estimated as $250,000 in 2006 while the lifetime costs per person infected with hepatitis C was approximately $46,000 per person in 2005. 50-70% of injecting drug users are infected with hepatitis C which will place demands on the health system with approximately 20% of infected people progressing to liver cirrhosis within 20 years and up to 10% developing liver cancer within another 5-10 years. In addition, once a person becomes dependent on opioids, particularly heroin, the cost of maintaining the habit contributes to an escalating involvement in income-generating crime, such as trafficking drugs, property crime or prostitution, to support the habit. In 1998-99, it was estimated that the total social costs of alcohol, tobacco and illicit drugs was $34,440M of which nearly 20% was due to illicit drugs. Economic evaluation of opioid dependence interventions is complex as post treatment outcomes affect both the drug dependent person and society as a © STACK & JAMES 2010 Proposal for Remote Dosing Program 15 whole. In addition, economic evaluation of certain outcomes such as the duration of medical problems is difficult. Despite this, there is a growing number of studies which show the benefit of maintenance programs e xceed the costs. In particular, studies between 2001 and 2004 showed for each additional healthcare dollar spend on a methadone maintenance program, $15 of crime-related expenditure was saved. In addition, a 2002 study found the benefits of dependence intervention for participants after being included in the program for one year included a 83% reduction in criminal activity, an 11% increase in employment earnings and a 6% reduction in health services utilisation. Significant costs are associated with the treatment of opiod dependence. A study of the methadone program in 2001 found significant differences in the costs of the program depending on whether it was delivered as an outpatient service or residential treatment with the cost of the labour involved in delivering the treatment responsible for up to 88% of the cost. While the labour costs associated with delivery of a methadone program is significant, other costs include testing to ensure the dosage and frequency of treatment is appropriate. These tests include urine and blood test. Other tests such as pupillometry, which involves measurement of the diameter of the pupil, are being investigated for use in treatment programs. The costs as well as the advantages and disadvantages of these tests are shown in Table 2 (on Page 16). Studies of methadone maintenance programs have indicated that their expansion is cost-effective and decreasing barriers to access generates significant health benefits for the community. In addition, a 2003 study in Sydney, Adelaide and Brisbane found that treatment with methadone was less expensive and more effective than treatment with buprenorphine. Despite these findings, expansion of the methadone maintenance program is dependent on governments funding treatment places and the availability of prescribing medical practitioners and dispensing pharmacists. As stated in a 1999 study, funding shortages require more efficient and less expensive ways of delivering treatment. © STACK & JAMES 2010 Proposal for Remote Dosing Program 16 Table 2 – Comparison of Drug Tests TEST COST PER TEST TIME/ ADVANATAGES RESOURCES TO ADMINISTER DISADVANTAGES Urine Approximately $30 plus the cost of labour to administer and analyse the test Approximately 5 minutes Tests for other drugs of dependence May detect drugs that have been in a the body for up to a week Fast Immediate results so can act to modify treatment protocol if necessary Diuretics and other drugs can mask Needs to be done in a medical clinic Too expensive to do every day Blood Approximately $30 plus the cost of labour to administer and analyse the test Approximately 3 days Tests for other drugs of dependence Not easily confused by other drugs Unreliable – only tells what is present in the body at time of test Needs to be done in a medical clinic Too expensive to do every day Not able to get immediate results Pupillometry (as proposed in this project) Only the cost of labour to administer and analyse the test 5 minutes Uncomplicated Accurate Immediate results so can act to modify treatment if necessary New protocols are not easily confused by other drugs Maybe able to tell if multiple drugs of dependency are being used Can be remotely administered Still being researched and developed Initial start up cost – over $200 Goggles not yet commercially available © STACK & JAMES 2010 Proposal for Remote Dosing Program 17 2.3 COST TO PATIENT OF METHADONE MAINTENANCE THERAPY Patients who undertake methadone maintenance therapy may experience a number of expenses including 2: The cost of daily transport to and from the dispensing pharmacy or medical centre (either cost of petrol, maintenance of car, taxi or bus fare) Lost productivity for employed patients who have to leave work to receive their medication The stress of keeping the details of their methadone maintenance therapy away from employers, family and friends so they do not lose their jobs or experience prejudice Personal safety issues related to being in contact with other drug users at the dispensing pharmacy or medical centre as some of these people may intimidate or threaten other patients to gain access to their doses Personal safety issues related to loss of resolve towards stability when exposed to low stability patients, or drug dealers who have ready access to other drugs of dependence Personal safety issues relating to loss of privacy due to other, less stable, patients attending the same pharmacy or dispensing centre who may discover personal information (e.g. place of residence, access to takeaway doses) Loss of personal dignity when having to queue at the pharmacy (often for extended periods such as hours), in view of other customers (which can lead to involuntary disclosure of their participation in the program), as people on the methadone program are often the last to be served Lost family time spent obtaining their daily medication (e.g travel time, dosing time) Difficulty travelling (for both work and on holidays) due to perceived or real restrictions in arranging supervised dosing (e.g. dispensing centres may not be available in remote areas). 2.4 SUPERVISION OF METHADONE TREATMENT Generally, people who are part of a methadone program (and other opiod pharmacotherapy program) are required to take their medication in front of a pharmacist or a medical practitioner. The exceptions to this rule are people 2 Most of the expenses of methadone maintenance therapy also apply to people on other forms of opioid pharmacotherapy programs. © STACK & JAMES 2010 Proposal for Remote Dosing Program 18 who are considered stable and are therefore approved to take medic ation home (known as takeaways). There is substantial evidence from a range of studies which indicates diversion and misuse (e.g. taking medication early, overdosing, injecting) is an integral risk in prescribing opioids (including methadone) either for management of dependence or management of pain. Much of the diverted opioids find their way to the black market where it is sold to be ingested orally or intravenously. In these circumstances the dosage and frequency of methadone taken by the person becomes uncontrolled. Diversion of prescribed opioids is associated with many adverse consequences including fatal overdose, an increased incidence of addiction, and the compromise of the public acceptance of treatment programs. As the methadone program expanded rapidly in the 1990s, there was an increase in methadone-related deaths. Approximately 75% of these deaths occurred as a consequence of diversion of methadone to people not in the methadone program. A study in Sydney in 1995 found an active black market in methadone with 88% of the drug sourced from diverted takeaway doses. A further study in 2002 found diversion of the drug to the black market increased proportionally with unsupervised treatment while a 2005 study found that the restrictiveness of takeaway policies, the market availability of heroin, and the dose dilution of takeaway doses, all also impact on the amount of methadone diverted. Making treatment accessible, affordable and attractive to potential patients needs to be balanced against the need to minimise diversion and maximise safety of the patient and the community. While supervision of treatment has been clearly shown to decrease diversion of medication, people in regular employment or studying may find daily attendance at the dispensing pharmacy disrupts the lifestyle stability they are trying to achieve. Although there is significant therapeutic value in the methadone program, supervising the program effectively imposes a significant cost on both the prescribing medical practitioner and the dispensing pharmacist. These costs include the time taken to consider all the issues surrounding requests for early, extra, replacement and takeaway doses and to take any necessary action (e.g. changing prescriptions). In particular, it is necessary to differentiate between stable and unstable patients. Stable patients will generally have a valid reason for requesting early, extra, replacement or takeaways doses (e.g. travelling for work). The prescribing medical practitioner and dispensing pharmacist are often aware that the consequences of denying the request may destabilise the patient (e.g. may lose job as cannot travel and then become depressed and unstable). In comparison, a request which is found to be invalid may be an early indication of instability requiring prompt attention to prevent rapid escalation of their instability. Therefore, validating requests and following up on potentially unstable patients, although time consuming, is an essential part of the patient’s rehabilitation and the continued safe provision of the methadone program. © STACK & JAMES 2010 Proposal for Remote Dosing Program 19 3. PROPOSED REMOTE DOSING PROGRAM 3.1 INTRODUCTION The proposal for the Remote Dosing Program is the accumulation of approximately 20 years community and clinical experience both in hospital and private medical practice. During this time, opioid dependency due to the use of prescription and illicit drugs was observed to be a significant problem within the community. Shortcomings in the current programs to rehabilitate drug dependent patients were also identified. The Remote Dosing Program was developed by Dr Clive Stack to address a number of the problems and issues involved in the treatment and rehabilitation of drug dependent patients. The program is aimed at patients who are stable on the existing opioid pharmacotherapy programs and introduces new technology to facilitate remote location dosing. This has the potential to enhance rehabilitation through increased social integration. As in existing opioid pharmacotherapy programs such as the methadone maintenance program, the interaction between the prescribing medical practitioner, dispensing pharmacist and patient is still pivotal. However, daily contact and assessment of the patient will be undertaken by a new Central Dosing Service using internet technology to assess the patient and dispense the medication. This will allow the patient to access medication at locations remote to a dispensing pharmacy or medical centre. Prototypes of the new technology for assessment of the patient and dispensing medication have been developed through collaboration between Dr Clive Stack and Dr Tim Gale of the University of Tasmania. These prototypes have been tested in preliminary trials that indicate further investigation is warranted. 3.2 AIMS OF PROGRAM The aims of the Remote Dosing Program are to: allow the provision of multiple doses over a day to assist in the tailoring of the program to match the individual's need (e.g. pain management where a number of smaller doses are required throughout the day) to assist and support reduction and termination of treatment by providing multiple, smaller doses throughout the day (as when the dose is reduced, a single daily dose is often insufficient to prevent withdrawal symptoms) © STACK & JAMES 2010 Proposal for Remote Dosing Program 20 reduce the potential harm of takeaway doses such as diversion, overdosing, other people taking dose (e.g. children) increase the flexibility of dosing to aid in the integration back into normal society (e.g. to make it easier for participants to undertake employment or study) allow investigation of other replacement programs utilising shorteracting medications help increase safer treatment options for other programs using other drugs of dependence (dexamphetamines, benzodiazapines) decrease the amount of drugs of dependence finding their way onto the black market from licit sources (i.e. diversion) increase the availability of the opioid replacement program to the community by removing the emotional, financial and time cost burden, created by the provision of takeaway doses, on the prescribing medical practitioner and dispensing pharmacist reduce the potential for instability caused by taking the takeaway dose inappropriately (e.g. intravenously or excessive amounts) which may result in the patient not having enough medication for each day reduce the health burden of increasing daily dose which is a natural consequence of the patient presenting to the prescribing medical practitioner as though the dosage is not maintaining them (e.g. with physical or emotional symptoms of withdrawal) requesting and seeming to need their dose increased hasten recovery from drug dependence through the ability to more effectively manage an individual's program due to the early discovery and treatment of instability. The process consists of 3 steps: Meeting the criteria for consideration of entry into the Remote Dosing Program and referral to the Remote Dosing Program Initial assessment by the Remote Dosing Program Daily assessments and medication dispensing. 3.3 PROCESS FOR REMOTE DOSING PROGRAM 3.3.1 CRITERIA FOR INVOLVEMENT IN REMOTE DOSING PROGRAM The patient will be initially assessed for entry into the Remote Dosing Program by the clinical staff from the Central Dosing Service in conjunction with the prescribing medical practitioner and dispensing pharmacist. A patien t will only be considered for the Remote Dosing Program if they are eligible for takeaway doses and display objective signs of a stable lifestyle within the preceding year. These include: © STACK & JAMES 2010 Proposal for Remote Dosing Program 21 Assessed as stable by prescribing medical practitioner Assessed as stable by their regular dispensing pharmacist Regular attendance to prescribing medical practitioner for medication and review No unexplained and/or unsubstantiated missed or lost doses of medication No unexplained and/or unsubstantiated requests for earl y or extra take away doses No request for dose to cover an unexplained or unsubstantiated specific activity (e.g a patient would need to produce receipt to show they have gone to funeral to get take away dose for that activity) No dosage escalation No escalation of other drugs of dependence No other illicit drug use including no needle puncture marks Regular job attendance or demonstrated ability to successfully undertake study (e.g. at TAFE or university) Financial stability Regular or stable housing (for security reasons associated with storage of medication in the Remote Dosing Program, the patient cannot be in a caravan) No unavoidable association with unstable drug users. 3.3.2 INITIAL ASSESSMENT The patient will initially be assessed for entry into the Remote Dosing Program by clinical staff in the Central Dosing Service in conjunction with the prescribing medical practitioner. The prescribing medical practitioner will be largely responsible for identifying patients who meet the criteria for involvement in the Remote Dosing Program as outline above. The Central Dosing Service ensures a consistent standard for entry into the Remote Dosing Program is maintained firstly by verifying the patient meets the entry criteria and then by further assessing the patient. The Central Dosing Service assessment consists of the following: Confirming patient’s identity and recording identification pictures and demographic data Full urine screens – These provide an overall screen for drugs of dependence and will show if the patient is using drugs of dependence other than opioids A simple reaction test – The person is comfortably seated facing a computer monitor holding a mouse in one hand. When the computer monitor screen changes from grey to green, the person clicks the left button on the mouse as quickly as possible. This is done a number of © STACK & JAMES 2010 Proposal for Remote Dosing Program 22 times to acquaint the person with the test as they will initially decrease their response time but after they have done the test a number of times, their reaction time will stabilise. Pupillometry – Five aspects of pupil physiology (the size and reactivity of the pupil) change with the methadone level in opioid dependent people. Unlike a number of other physiological effects caused by opioids, these changes do not adapt as the person continues to take opioids. Pupillometry can record these 5 changes which makes it a reliable test regardless of the duration of drug use. These aspects of pupil physiology can be recorded by a high resolution camera in response to changing light levels (i.e. a pupillogram).The pupillometry test is conducted with the patient wearing a pair of goggles fitted with computer controlled light emitting diodes (LED) and an infrared camera linked to a USB webcam. An infrared LED is used to illuminate the pupil for the camera while a white LED is used to create the pupil reaction. The USB webcam records the pupil reaction to the changes in the light which is then used to measure the five changing aspects of pupil physiology. Image 1 – Prototype of Pupillometry Goggles The information from these assessments forms the baseline data for each patient. Information gathered during the daily assessments will be compared to this baseline data before medication is dispensed through the dosing machine. A person who has an eye disease or who is totally blind may not be able to participate in the Remote Dosing Program as they may not be able to be monitored using pupillometry. In addition, people with poor upper limb dexterity such as quadriplegics may not be able to participate in the Program © STACK & JAMES 2010 Proposal for Remote Dosing Program 23 as they may not be able to manipulate the equipment or undertake the simple reaction test. During the assessment process, the patient's medical practitioner will also be contacted to advise of the patient's current medication regime including the dosage and frequency of taking medication. Details of the medication regime will be held on file at the Central Dosing Service and medication will only be authorised in accordance with this regime. The patient's medical p ractitioner will also be connected to the Central Dosing Service and can record any variation in the medication regime determined as a result of any contact with the patient directly onto the Central Dosing Service files. Only the patient's prescribing medical practitioner can make changes to medication dosages and frequency as the Central Dosing Service will neither prescribe medication nor change medication regimes. In addition, only the patient's medical practitioner can read the patient's file held by the Central Dosing Service. The Central Dosing Service will use the same encrypted secure internet technology and protocols currently used between doctors when transferring a patient’s file to ensure the patient's confidentiality and privacy are maintained. After the initial assessment has been undertaken and the patient is accepted into the Remote Dosing Program, arrangements are made with the pharmacy that usually dispenses the patient's medication for their inclusion in the program. This includes inducting the pharmacist into the program, training the pharmacist in how to load medication into the dosing machine, providing the pharmacist with codes to allow access to the Central Dosing Service , and providing the pharmacist with the hardware and software to participate in the program. 3.3.3 DAILY ASSESSMENTS AND MEDICATION DISPENSING The remotely monitored medication system will be set up to dispense tablet or liquid forms of medication after assessment of the patient by a clinician at the Central Dosing Service. While the initial assessment and prescription of a medication regime must be conducted by the patient’s medical practitioner, the clinician who assesses the patient before medication is dispensed each day does not need to be a medical practitioner as they are comparing specific behaviours displayed by the patient at the time of dosing to the baseline data previously recorded. The dosing machine is linked to a computer, which has communication and data acquisition software loaded, cameras to monitor the dispensing of the medication, and a pupillometry device for patient assessment. The computer has a high speed secure internet connection which links the patient with the Central Dosing Service. © STACK & JAMES 2010 Proposal for Remote Dosing Program 24 The patient can only access the dosing program through a password which is authenticated each time. The clinician at the Central Dosing Service also confirms the patient's identity each time the patient accesses the program by comparing their video image with images taken of the patient during their initial assessment and stored in their file. Figure 4 – Computer Connections Between Patient and Central Dosing Service Web cameras Pupillometry goggles Patient’s computer Communication and data acquisition software Remote dispenser Secure Internet connection Central Dosing Service Computer Web cameras Communication and data acquisition software Establishment of connection with the Central Dosing Service, including visual and audio communication, is required as part of the daily assessment process. Each dosing machine has its own code. When a connection is established with the Central Dosing Service over the secure internet, it will read the code on the patient's dosing machine and confirm the medication contained is correct according to information provided by the dispensing pharmacist when the machine was loaded. In addition, the location of the dosing machine will be confirmed by GPS. Images of the client, the doses within the dosing machine plus audio information will be sent to the Central Dosing Service both before and after the medication is dispensed. The pre-medication assessment consists of: © STACK & JAMES 2010 Proposal for Remote Dosing Program 25 Observation of facial expressions/of the patient – the clinician notes a number of aspects including: blink rate, closing eyes (as if falling asleep), nodding, losing context in sentence (forgetting what they’re saying.) A simple reaction test – the patient is requested to click the left side of their computer mouse when a square changes colour on their computer screen. The test is done five times and the results averaged. Pupillometry – the patient will be required to put on a set of pupillometry goggles and the pupillometry assessment, as previously described, will be undertaken. Upon receiving this data, the clinician at the Central Dosing Service can compare it to baseline information obtained during the initial assessment. This comparison is necessary to ensure over-medication of the patient is not occurring and the patient is stable enough to be receiving remote dosing. If the patient looks sedated or any of their assessment does not conform to the baseline information, the assessment will be failed. When this occurs, the patient will need to be immediately reviewed by their prescribing me dical practitioner. A medical practitioner at the Central Dosing Service may do the review if the patient cannot get in to see their prescribing medical practitioner. The patient will not receive their dose of medication until this review occurs. The Central Dosing Service may arrange transport to either the medical practitioner or the Central Dosing Service office if it is required. As part of the review, the patient will immediately have to undergo an assessment including breathalyser test, urine/drug screen, pupillometry and reaction tests to determine if they have been overdosed, taken other drugs or substances which have interfered with their behaviour or reactions or have another medical problem which must be addressed. If the patient is found to be unstable, they may be excluded from the Remote Dosing Program and may have to return to daily visits to the dispensing pharmacy to get their medication. After a period of time (e.g. 6 months but the exact length of time will be determined by the nature of the instability and the danger to the patient or community as a result of their instability) the patient may apply to be again included in the Remote Dosing Program. If the patient passes the pre-medication assessment and the doses contained in the dosing machine match the contents indicated on file, the Central Dosing Service initiates dispensing of the medication. The details of the medication including the type, dose, date and time of dispensing are recorded. When the dose is authorised, the lock on the machine is automatically disengaged. The patient must follow predetermined protocols for where they are to stand and how they are to hold the dosing machine to ensure the cameras can fully record the medication being taken. When the medication is dispensed, the © STACK & JAMES 2010 Proposal for Remote Dosing Program 26 cameras must have direct vision of it leaving the machine and being taken by the patient. The patient must then follow a set of protocols allowing the cameras clear vision of their mouth to show the medication has been taken correctly (e.g. if it’s been swallowed, placed under the tongue) Every protocol must be followed every time the patient gets medication from the dosing machine. If any protocol is not followed (e.g. the patient turns their head away from the cameras at the time of swallowing the medication, which could indicate the patient is spitting it out), the patient will be assessed with possible immediate exclusion from the Remote Dosing Program by the Central Dosing Service clinician. If this occurs, the patient will have to return to daily visits to the dispensing pharmacy to get their medication. After a period of time (e.g. 6 months, but the exact time period will be determined by the nature of the breach) the patient may apply to be again included in the program. It will take approximately 3 minutes for the pre-medication assessment to be undertaken and the medication dispensed. Another 5-15 minutes will be needed for medication dissolution depending on the form of medication dispensed (eg buprenorphine takes up to 15 minutes to dissolve under the tongue). If a patient does not contact the Central Dosing Service for a scheduled medication dose (e.g. does not contact the Service for 24 hours) they will be immediately investigated. If it is found there is no reasonable explanation for the patient failing to make contact (e.g. the patient has taken other illicit drugs) they will be excluded from the Remote Dosing Program and will have to return to daily visits to the dispensing pharmacy to get their medication. After a period of time (e.g. 6 months, but the exactly length of time will be determined by the nature of the instability and the danger to the patient or community as a result of their instability) the patient may apply to be again included in the Remote Dosing Program. Figure 5 – The Dosing Process Patient log-in Client side Staff Server side Authentication Server Connection & Authentication Connection Established Webcam on Audio On Visual, Pupillometry and Reaction Time Assessment Appropriate Medication Delivery Data Stored © STACK & JAMES 2010 Proposal for Remote Dosing Program 27 The audio and video communication plus the software required for monitoring of the patient require 3.5G wireless technology to be available to both the patient and the Central Dosing Service. 3.4 THE DOSING MACHINE The dosing machine, which contains the medication, will be a metal tube which holds tablets or a liquid form of medication. Depending on the patient's prescription, there may be multiple doses of medication in a single dosing machine or the patient may need to have multiple dosing machines each containing a single dose. Each dosing machine has a unique computerised serial number. The dispensing pharmacist assigns one or more specific dosing machines to each patient by its serial number. A record of the patient and the serial number on machines they are allocated is made by the dispensing pharmacist at the time the machine is filled and forwarded to the Central Dosing Service over the secure internet. The patient must keep the dosing machine in an accessible place which has mobile phone coverage and cannot move the dosing machine through areas which do not have mobile phone coverage except with prior approval from the Central Dosing Service. The dosing machine is connected to the Central Dosing Service via the patient's computer. When connected, it will send its serial number which will then be confirmed by the Central Dosing Service as being allocated to a machine assigned to the patient. The dose contained in the machine will be checked through an internal camera against the records from the dispensing pharmacy to ensure they are the same. The Central Dosing Service can unlock the dosing machine via the internet connection to allow the medication to be received. When this occurs, a retractable tube will ex tend from the dispenser and the patient places their mouth over the tube to receive their medication. The time of ejection of the medication is controlled by the patient using a push-button mounted on the dosing machine. When medication in the dosing machine is insufficient for the next dose, the patient will get a message telling them it needs to be refilled. © STACK & JAMES 2010 Proposal for Remote Dosing Program 28 Image 2 – Prototype of the Dosing Machine It is not possible for the patient to unlock the dosing machine without breakin g into it. Each dosing machine will have internal GPS acquisition technology, light and pressure sensors which will be activated if the machine is moved, opened or breached by an unautherised person. The dosing machine will have a mobile phone facility which will send a SMS if movement or a break-in occurs. It also has its own software that records video/audio data as soon as the machine is moved or the light or pressure sensors are activated. This is to assist in identification of the circumstances and people who steal or break into the machine. This data will be sent to the Central Dosing Service if the machine is in mobile phone range or sent as soon as the machine comes back into mobile phone range. There will be an alarm on the Dosing Machine but it will not be audible. The alarm will alert the Central Dosing Service which will immediately respond to it or any other signal of unautherised movement of the dosing machine, breach or break-in. This is necessary to reduce morbidity and mortality that might result from any unauthorised use of the medication. If it is found that the patient is responsible or involved in the theft or break-in, they will be excluded from the Remote Dosing Program and will have to return to daily visits to the dispensing pharmacy to get their medication. After a period of time (e.g. at least 12 months) the patient may reapply to be included in the program. When a patient is approved for inclusion in the Remote Dosing Program, they will be provided with a safe in which to keep their dosing machine. The safe, which will be set into a concrete block, will be delivered to the patient’s home address by the Central Dosing Service. The safe must be kept outside the patient’s house so there is no risk to the patient or their family if somebody decides to steal or break into it. In addition, the safe must be in an area which can be accessed by the Central Dosing Service in the case event of an emergency (e.g. the dosing machine alarm is activated). © STACK & JAMES 2010 Proposal for Remote Dosing Program 29 3.5 DUTIES OF PARTICIPATING PHARMACISTS The duties of the dispensing pharmacists in the Remote Dosing Program will be different to their duties under the current opioid pharmacotherapy such as the methadone maintenance program. While they will still be required to dispense medication, the method in which they do this will be significantly different. In addition, they will not be required to dose the patient. The pharmacist will dispense medication via the dosing machine provided to them by the Central Dosing Service. To dispense the medication th ey must access the machine by plugging it into the computer terminal that connects them to the Central Dosing Service. Once the Central Dosing Service has confirmed the dosing machine is fully functional through a remote diagnostic check of the machine they will authorise its loading. This will allow the dosing machine to be opened without activating the alarm systems. The pharmacist will then load the machine with the patient’s medication in accordance with the prescription provided by the prescribing medical practitioner. At the time this is done, the Central Dosing Service will be advised (via the internet connection) of the dosage and type of medication being provided to the patient. The dosing machine being loaded is also identified by serial number. At times, the Central Dosing Service may require the dispensing pharmacist to replace a dosing machine (e.g. if a machine requires repair or to be updated). In addition, the dispensing pharmacist may be requested to keep the dosing machine rather than refill it and refer the patient back to their prescribing medical practitioner. This may be necessary if the pre- or postassessments undertaken by the Central Dosing Service indicate the patient’s medication level needs to be reviewed. Finally, if the patient has breached a condition of the Remote Dosing Program (i.e. turned away from the web cam camera while taking a dose of medication), the dispensing pharmacist may keep the dosing machine and refer the patient to the Central Dosing Service who will explain both why they are being excluded from the program and the duration of their exclusion. Patients who are excluded from the Remote Dosing Program will still be able to access their medication by going to the dispensing pharmacy each day. Pharmacists who participate in the Remote Dosing Program will be provided with the training and software to undertake their responsibilities under the program. 3.6 PATIENT AGREEMENT © STACK & JAMES 2010 Proposal for Remote Dosing Program 30 Before the patient is included in the Remote Dosing Program, they must enter into an agreement to abide by the conditions of the Remote Dosing Program and acknowledge repercussions may apply where conditions are breached. This agreement will be in writing and patients may seek advice from other people (e.g. a legal representative) before entering into the agreement. The conditions in the agreement with the patient will include: Not to break into, tamper with or in any other way breach the dosing machine To immediately notify the Central Dosing Service if they become aware the dosing machine is breached, tampered with or stolen Except when in use, to keep the dosing machine in the safe provided Not to give access to the dosing machine safe, including not providing anybody else with the combination code or leaving details of the combination code where somebody else may find it To immediately notify the Central Dosing Service if the safe is breached or stolen If the dosing machine alarm is activated at the Central Dosing Service indicating that the machine has been breached or tampered with, to provide the staff of the Central Dosing Service access to their home to ensure the safety of all household members (i.e. that nobody has taken an overdose) To follow the set protocols for dosing when using the dosing machine (e.g. face the web cam) To give permission for the Central Dosing Service staff to view data on their dosing event and analyse the data To accept any consequences for a breach of the conditions which may be applied (e.g. exclusion from the Remote Dosing Program for a period of time) To notify the Central Dosing Service if they wish to move the Dosing Machine from the area of the safe such as when taking an overnight trip away or when returning the machine to the pharmacy to be reloaded. © STACK & JAMES 2010 Proposal for Remote Dosing Program 31 3.7 COMPUTER NETWORK Figure 6 – The Computer Network Prescribing Medical Practitioner Dispensing Pharmacy Central Dosing Service Patient’s Dosing Machine Patient 3.8 ADVANTAGES OF REMOTE DOSING PROGRAM The advantages of the Remote Dosing Program include: Increasing the rigour of pre-dose assessments which in this program are more onerous than current pre-dose assessments conducted by dispensing pharmacies. The Remote Dosing pre-dose assessments are objective, based on physiological reactions which cannot be controlled by the patient. They are more likely to objectively detect signs of intoxication, overdosing or the possibility of other drugs consumption. Conversely, they may identify signs of withdrawal. This makes the system safer for the patient and also for the public. © STACK & JAMES 2010 Proposal for Remote Dosing Program 32 Decreasing the risk of unstable patients receiving takeaway dose s of medication as the patient will only have access to medication through an automatic dispenser controlled via the computer network if the pre medication assessment is passed. The patient cannot manipulate or intimidate the assessor into providing medication as they do not hav e direct or physical access to the assessor. Rigorous assessment every time medication is dispensed allows a clear record of the patient to be maintained and quicker detection of instability. Where instability is suspected, appropriate measures can be instigated to resolve problem before it escalates into a situation that is harder to address. Potentially increases access to drug substitution programs (e.g. the methadone maintenance program) as it reduces the workload on prescribing medical practitioners and dispensing pharmacists through a reduced workload related to responding to requests from patients for take away doses. Facilitates implementation of opioid reduction programs as smaller doses can be provided more frequently to deal with the problem of doses being too small to last for the whole 24 hours which affects compliance with the reduction process. Allows for the use of short acting/short half-life medications in drug replacement programs such as amphetamines or short half -life opioids. Currently, these cannot be used because of a lack of available of programs or inability to suitably supervise multi-dosing throughout a day. Eliminates the possibility of the patient taking more than one dose at a time. This in turn decreases the risk of overdosing or death. May create an environment of greater stability by preventing a “bouncing” phenomenon where the patient takes too much medication then has insufficient medication in their next dose to stop withdrawal so requests or takes a higher does when medication becomes available again Eliminates the possibility of other people (e.g. children) accidentally taking the medication and overdosing or dying Allows for immediate detection of interference with doses through alarm systems fitted to the dosing machine © STACK & JAMES 2010 Proposal for Remote Dosing Program 33 Reduces black market availability of prescribed medication as the dosing machine is more secure than current arrangements for takeaway doses Facilitates the expansion of opioid pharmacotherapy programs as will allow greater participation by those people currently excluded due to their location or employment situation Allows people on the program to travel because they only need to be connected to the internet service to be dosed. Current programs are restrictive as the person must be approved for takeaway doses or attend a designated dispensing pharmacy or medical practice to receive their medication. If they travel within Australia, arrangements can be made for their medication to be transferred to another pharmacy or medical practice. However, the person must make travel arrangements around attendance at the designated location. This may be particularly onerous for people who are required to travel as part of their employment Allows people to work or live in rural and remote areas (providing they have 3G coverage) to access opioid pharmacotherapy programs. Currently, the access to opioid pharmacotherapy programs for people in rural and remote areas is restricted by the number of and distance to doctors or pharmacies participating in a program and whether the patient can attend the pharmacy or medical practice on a daily basis Allows shift workers access to the opioid pharmacotherapy programs. Some shift workers are restricted in accessing opioid pharmacotherapy programs as their availability to attend dispensing pharmacies or medical practices may not coincide with the hours they are open. Reduces discrimination against people participating in the opioid pharmacotherapy programs as the likelihood of their drug treatment being detected is reduced. If these people are observed going to pharmacies to get their daily medication, it may result in discrimination both personally and professionally with a significant loss of productivity to the community Reduces discrimination against people “in the public eye” participating in the opioid pharmacotherapy programs as these people often cannot take part in the current program due to the likelihood of their drug treatment being detected © STACK & JAMES 2010 Proposal for Remote Dosing Program 34 Reduces the risks of harassment or discrimination by other people in the pharmacy (e.g. other people on opioid pharmacotherapy programs or pharmacy customers) Reduces centralisation of drug users in one area (i.e. the pharmacy where they receive their medication) which may lead to inappropriate networking (e.g. discussion of how to get more medication) or black market pressure (e.g. intimidation to divert medication) Allows people with a combination of drug dependence and pain issues to be given supervised doses of medication suitable for pain control. Many of these patients often have reduced mobility so daily travel to a dispensing pharmacy is not practicable. In addition, people with this combination of issues often require multi-dosing through the day which is not currently possible if they have reduced mobility or restricted access to a dispensing pharmacy (e.g. limited access to public transport) Allows participants to explore family or academic or work options that are not currently available due to the restrictions of the existing program imposed by pharmacy hours, time taken to travel to/from pharmacy, cost of transport, child care issues (e.g. sick children and nobody to care for them while go to pharmacy) Reduces the cost to the community of daily dosing as the patient will only need to be assessed by a medical practitioner if they fail the predosing assessment. © STACK & JAMES 2010 Proposal for Remote Dosing Program 35 3.9 DISADVANTAGES OF REMOTE DOSING PROGRAM The disadvantages of the Remote Dosing Program include: Some costs are associated with the implementation of the system including production of dosing systems (dosing machine and pupillometry goggles), implementation of software and training doctors and pharmacists in the new software Costs are associated with the implementation of the Central Dosing Service including costs of its establishment and staffing and implementation of emergency teams Many patients currently using takeaway doses will not like the intensity of assessment as it will detect instability more quickly than the current system. Some unstable patients have a vested interest in the retention of the existing takeaway dose system as the opportunity for medication abuse through diversion is increased The process must be controlled by a non-government organisation as it must not be able to be challenged on the grounds of discrimination (e.g. by people on opioid pharmacotherapy programs contacting members of parliament or ombudsmen to complain about their exclusion from the program). The proposed program will actively discriminate to exclude patients who are unstable or who have been found not to comply with the guidelines. This is necessary to ensure the safety of the program. It would be inappropriate for patients to have access to a complaint or appeal procedure if they are excluded. (It should be noted these patients will not be excluded from the opioid pharmacotherapy program – just the Remote Dosing Program. They will still be able to receive medication through dispensing pharmacies or medical practices.) However, monitoring of the program to ensure guidelines are being applied in a non-discriminatory and consistent way is appropriate and will occur. © STACK & JAMES 2010 Proposal for Remote Dosing Program 36 4. PRELIMINARY RESEARCH In 2009, two preliminary trials relating to the Remote Dosing Program were undertaken: the first to assess remote assessment technology by comparing pupillary reactions and simple reaction time to the blood methadone levels, and the second to trial the dosing machine. The dosing machine, pupillometry goggles and software for the remote assessment were developed by the Department of Engineering, University of Tasmania in consultation with Dr Clive Stack. Both trials where undertaken as a collaboration between Dr Clive Stack and Dr Tim Gale of the University of Tasmania. The trials were subject to rigorous assessment and authorisation by the Tasmania Scientific Research Advisor y Committee and the Tasmania Health and Medical Human Research Ethics Committee. The Therapeutic Goods Administration, Department of Health and Aging (Cmth) was also notified of the trials in compliance with the Clinical Trial Notification (CTN) scheme, pursuant to Schedule 4 of Regulation 7.1 of the Therapeutic Goods (Medical Devices) Regulations 2000. 4.1 TRIAL OF REMOTE ASSESSMENT TECHNOLOGIES This trial was designed to investigate whether or not the pupillometry system, including the custom designed goggles, and simple reaction time test could be used as pre-dose assessment tools in the Remote Dosing Program. If it could be shown that a person’s pupil and physical reactions changed in response to methadone levels, these measures could be used to indicate whether or not that person was unstable through already having non-prescribed opioids or other drugs of dependence in their body or if that person was, potentially, in withdrawal. The trial consisted of 9 stable patients currently undertaking the metha done maintenance program and 2 people not taking methadone or any other drugs. All 11 people were given the full initial assessment (pupillometry, reaction time test, urine drug screen). In addition, the 9 people on the methadone program had their blood taken before the trail commenced for an initial measurement of blood methadone level. The people who were already taking methadone were then given their prescribed dose of medication. Each hour after this, over a 6 hour period, blood methadone levels were measured via blood tests and at the same time pupillary reactions to various levels of light stimulation were recorded. A computer controlled pupillometer was used to take the pupillograms and © STACK & JAMES 2010 Proposal for Remote Dosing Program 37 ensured the pupillometry was undertaken in a consistent manner. Participants also undertook a simple reaction test each hour and breath alcohol tests. The 2 people who did not take methadone undertook all assessments except the blood tests. All participants in the trial also had urine drug screens performed after testing was completed at the end of the 6 hour period. The trial was undertaken in a single clinic. No dosing or assessment was carried out at a location remote to the clinic. The trial found there were at least 5 distinct aspects of pupil physiology and one aspect of the reaction time test that changed significantly with the methadone level in opioid dependent people. These characteristics were not observed in the two people who did not take methadone. The trial also found a strong correlation between blood methadone levels and the 5 aspects of pupil physiology as well as a strong correlation between blood methadone levels and the simple reaction time test. These results suggest that the pupillometry and the reaction time test are worth further investigation as assessment tools for the Remote Dosing Program. The results of this trial are in preparation for publication. 4.2 TRIAL OF DOSING MACHINE The trial was designed to test both the dosing machine and the software developed for the dosing machine. Two drug dependent patients currently on buprenorphine participated in the trial. They were placed in a room with the dosing machine linked to the controlling clinician in another room over an intranet via control and communication software build specifically for the program. This process allowed contemporaneous visual and auditory communication between the clinician and the patient. (This was necessary to monitor the patient as they took the medication and to ensure they did not divert it.) Verification of the successful delivery of the medication was demonstrated by the dosing machine delivering a dose of medication and images provided by the software (computer control of dosing machine and webcam footage). The patients filled in post-dosing questionnaires related to their perception of the functionality and usability of the dosing machine. The trial resulted in successful acceptable remote delivery of the medication directly into the patient’s mouth confirming the functionality of the system. © STACK & JAMES 2010 Proposal for Remote Dosing Program 38 5. PROPOSED RESEARCH – PILOT STUDY 5.1 AIM It is proposed to implement the dosing machine together with supporting software in a pilot study to show the equipment and assessment protocols are effective and provide a safe remote dosing system. 5.2 METHODOLOGY This pilot study will be the first remote test of the procedure, equipment and software. It will have two components: the development and production of equipment and technology followed by a clinical pilot of the equipment and technology. The production of equipment and technology will take approximately 6 months and will include: Production of dosing machine with the inclusion of security and control devices (e.g. sensing capabilities) to detect and prevent tampering Development of patient's software for dosing machine Development of Central Dosing Service software and server Development of software for interpretation of pupillograms (Neural Network Software) Production of pupillometry goggles The clinical component of the pilot study will include approximately 8 patients currently receiving takeaway doses on an opioid pharmacotherapy program and residing in the south of Tasmania. Participants will be identified and selected by the participating prescribing medical practitioner who has been monitoring their progress on the opioid pharmacotherapy program. All participants will be identified as meeting the criteria for stability and have met this criteria for at least 1 year (i.e. are very stable). It is likely they will be recruited from the patients who were involved in the preliminary research. The patients will be provided with a computer system linked to a Central Dosing Service, pupillometry goggles and dosing machine. This will allow the software for pre- and post-dosing assessment to be piloted as well as all necessary record-keeping for the Central Dosing Service. Patients will also be provided with a safe in which to keep the dosing machine when not in use. The safe will be modified by being set into a concrete block to deter theft and will be placed in the grounds of the patient’s home but © STACK & JAMES 2010 Proposal for Remote Dosing Program 39 outside the house. The Central Dosing Service will install the computer which will be used with the dosing machine in the patient's home after the area where the dosing machine is to be used has been inspected and has pas sed criteria for functionality and security. These criteria include ensuring that the area where the dosing machine is to be used is not easily accessible to the outside or to other people living in the residence, there is suitable, adequate lighting, and the webcam affords a clear view of the patient and surrounding area such that any attempt to divert medication in the dosing process will be noticed on the webcam. In addition, the Central Dosing Service may need to access to the computer located in the patient's home as well as the dosing machine and pupillometry goggles during the clinical pilot if there is a need to update or repair software or hardware. In addition to the pre- and post-dose assessments, participants will be required to participate in random urine drug screens, blood methadone level tests and physical assessments throughout the pilot study. In addition to checking for illicit drug use, these tests will allow the results of the pupillometry testing to be assessed and confirmed. The dispensing pharmacists who fill the dosing machine and the prescribing medical practitioner who supervise the participants’ medication prescription will be linked to the Central Dosing Service and will pilot the relevant software. The dispensing pharmacists who will be involved in the pilot will be recruited from the pharmacies that have already been involved in the preliminary research. Participants will be encouraged to investigate ways in which pre - and postassessments can be circumvented or dosing machines can be manipulated or breached. Participants who are successful will receive a cash bonus if they provide detailed advice as to how they achieve this and will not incur other consequences for such breaches. Patients will be monitored to ensure they do not take other drugs or non-approved medication or engage in any other harmful activity in attempts to circumvent assessments or tamper with dosing machines. Any patient who is found to have otherwise tampered with or breached a dosing machine, taken other drugs or non-approved medication, or engaged in any other harmful activity will be immediately excluded from the pilot study. The pilot study will run for approximately 1 year. The development of the infrastructure (software, dosing machines, server) will take approximately 7-8 months. The final stages of this development will overlap with implementation of the clinical phase which will take approximately 6 months. Prior to the implementation of the clinical phase, a number of single day trials will be conducted using different pharmacotherapy drugs to further test the reproducibility of the results of the assessment tools. Implementation manuals will be written during the clinical implementation. © STACK & JAMES 2010 Proposal for Remote Dosing Program 40 5.3 RESOURCES The pilot study will require the following resourcing: development of software to link participants, medical practitioner, pharmacy and Central Dosing Service production of dosing machine production of pupillometry goggles purchase and installation of computer equipment including webcams for participating patients and the Central Dosing Service provision of training for participating dispensing pharmacists and Central Dosing Service clinician. It is envisaged the following staffing will be required: two part-time research assistants who will undertake data entry, analyse results, deliver training and develop training manuals for the Central Dosing Service a part-time dosing clinician (3 hours a day, 7 days a week) who will undertake pre- and post-dosing assessment and monitor/assess the effectiveness of assessment protocols a part-time project manager (4-8 hours a week) who will oversee the budget, purchasing, resource allocation and project reporting a full-time medical practitioner/chief researcher who will be responsible for the pilot study’s implementation, monitoring and assessment of all aspects of the pilot study’s design, supervising the analysis of results and identifying trends, developing new policy and an implementation strategy for extension of the program. This person will also be available for any medical emergency such as in the case of an overdose, unauthorised movement of dosing machine and to provide urgent assessment if additional drug use is suspected plus oversee medication prescriptions and random urine/blood tests various contractors and consultants who will develop, install and maintain the software, undertake the production of pupillometry goggles, implement the computer systems, and develop and implement any necessary training. In addition, the research assistants and Central Dosing Service clinician will have responsibility for emergency call-out if the dosing machine is breached. 5.4 PROPOSED BUDGET The budget for the trial has yet to be confirmed however, it is anticipated to be in the order of $500,000. © STACK & JAMES 2010 Proposal for Remote Dosing Program 41 6. CLINICAL TRIAL Following the pilot study and any adjustment to the technology and software, a larger clinical trial will be undertaken. This trial will extend the pilot parameters to include more people (medical practitioners, pharmacies and patients), more variables (e.g. patients at different levels of stability so will increase the risk of problems occurring such as tampering with dosing machine) and a larger geographical spread. The intention of the trial is to test the procedures, equipment and software to show that the equipment and assessment protocols are effective and to provide a safe remote dosing system that is suitable for wider scale implementation with people on opioid pharmacotherapy programs. The participants in this trial will be a combination of very stable and relatively stable patients. There will be 30-40 participants in the trial each of whom must meet the criteria for inclusion in the Remote Dosing Program as well as the following criteria: Returned negative random urine drug screen for non-prescription drugs at initial screening Returned negative breathalyser test at initial screening. The trial will be run over 2 years and will have two components: development of assessment technology and implementation of the clinical trial. The development of the assessment technology will take approximately 1 year and will include: Review and improvement of design of dosing machine in light of the results of the pilot study, research and inclusion of safety and control devices (e.g. sensing capabilities) to detect tampering Review and improvement of patients’ software for dosing machine Extension and improvement of Central Dosing Service software and server Review and improvement of pupillometry goggles Development of software to analyse pupillometry and reaction time data, using modelling techniques, to facilitate automation of some of the processes. This will allow some assessment work in routine, low risk situations to be undertaken by people with less training (e.g. technicians) leaving non-routine, high risk situations to be assessed and managed by clinicians. The implementation of a larger clinical trial will follow from the improvements in the technology. The preparation for the clinical trial will include: Production of dosing machines and goggles as well as purchase of computers for patients to be involved in the project Recruitment and training of pharmacies and medical practitioners © STACK & JAMES 2010 Proposal for Remote Dosing Program 42 Recruitment and training of Central Dosing Service staff. Following these preparations, a full clinical trial will be run for approximately 1 year. This will provide sufficient time to collect data on implementation problems, investigate and implement possible solutions, and identify and implement improvements to the technology or the program. After this trial, the technology and program will be assessed to determine if it is economically feasible and socially practicable to extend the program further. © STACK & JAMES 2010 Proposal for Remote Dosing Program 43 7. APPENDIX 1 – MEDICAL TERMS 7.1 OPIOIDS Opioids are a class of compounds that are endogenous (e.g. endorphins) or naturally occurring (e.g. morphine or codeine) or synthetically produced (pethidine, methadone, and heroin). Opioids act by binding to the opioid receptors mainly in the central nervous system and gastrointestinal tract. There are many different classes of opioid receptors. Most opioids will bind to a number of different opioid receptors. Opioids can have a number different actions on receptors including fully or partially blocking or activating them. The combination of the binding strength, stimulation type of a particular opioid and the specific receptors with which it binds are responsible for many of the differences in the effects of different opioids on the human body. The human body creates endogenous opioids such as endorphins in response to a number of stimuli including pain. These bind to specific opioid receptors to decrease the perception of pain which, in turn, leads to greater pain tolerance. A feeling of euphoria may also result from the release of endorphins which can act to impress certain behaviours in the brain. Other opioids act in a similar manner when taken: binding to the naturally occurring opioid receptors to do similar things including decreasing the perception of pain and sometimes creating a feeling of euphoria. Tolerance, due to neuroadaption (i.e. the change in the body’s sensitivity to the drug through reduction in receptor number and/or sensitivity) can occur resulting in a reduced drug effect. As a result higher doses of the opioid are required to achieve the same effect. Dependency is characterised by withdrawal symptoms when opioid use is discontinued. Dependency is not the same as addiction. Addiction occurs when there is a physical and psychological dependency on the opioid. However, it should be noted that withdrawal symptoms may reinforce the addiction. 7.2 METHADONE Methadone (also known as Physeptone) is a synthetic opioid used medically as an analgesic, antitussive and maintenance anti-additive for use in patients dependent on opioids. Although chemically unlike morphine and heroin, it acts on the opioid receptors in the brain and produces many of the same effects. © STACK & JAMES 2010 Proposal for Remote Dosing Program 44 Methadone is also used in managing chronic pain. This use is due to a number of factors such as its long half life (i.e. the time it takes for the b lood level of the drug to reduce by one half). It also has a much lower cost than a number of other prescription medications. In 2004, a month supply of methadone was $120 compared to an equivalent analgesic amount of meperidine at $240 or $500 or more for hydromorphone, morphine, fentanyle or oxycodone. Methadone is useful in the treatment of opioid dependence – for both illicit and prescription opioids. Its duration of effect is longer than many other opioids and it has cross-tolerance with other opioids including heroin and morphine. This means methadone can be used to mitigate withdrawal symptoms. In addition, higher doses of methadone can block the euphoric effects of heroin, morphine or other similar drugs. As a result, properly dosed methadone patients can stop or reduce their use of these other substances. It should be noted that methadone will not always help reduce the use of non narcotic substances such as cocaine, methamphetamine or alcohol. © STACK & JAMES 2010 Proposal for Remote Dosing Program 45 8. APPENDIX 2 - LITERATURE REVIEW At 30 June 2008 there were 41,347 patients in Australia receiving pharmacotherapy treatment for opioid dependence (AIHW). The pharmacotherapies for opioid dependency currently used include: methadone, buprenorphine and buprenorphine/naloxone. When the mode of pharmacotherapy treatment is disaggregated, it is found that methadone was the most common form of pharmacotheraphy treatment in 2008 with over 60% of patients prescribed methadone alone (AIHW). There are a growing number of studies which show that the benefits of methadone maintenance programs exceed their costs. In particular, studies between 2001 and 2004 showed for each additional healthcare dollar spen t on a methadone maintenance program, $15 of crime related expenditure was saved (Godfrey, Gossop). In addition, a 2002 study found the benefits after being included in the program for one year included a 83% reduction in criminal activity, an 11% increase in employment earnings and a 6% reduction in health services utilisation (French). Studies of methadone mainten ance programs have indicated that their expansion is cost-effective and decreasing barriers to access generates significant health benefits for the community (Doran, Connock, Zaric). However, it is significant to note that while there is a need for more efficient and less expensive ways to deliver methadone maintenance programs (Ward) the benefits of the programs are dependent on the quality of the treatment (Bell). Generally, people who are part of a methadone program are required to take their medication in front of a pharmacist or a medical practitioner. The exceptions to this rule are people who are considered to be stable and are therefore approved to take medication home (known as takeaways). A study of the methadone treatment programs in the USA in 2001 found significant differences in the costs of the program depending on whether it was delivered as an outpatient service or residential treatment with the cost of the labour involved in delivering the treatment responsible for up to 88% of the cost. (Roebuck) While the labour costs associated with the delivery of a methadone maintenance program are significant, other costs include testing (such as urine and blood tests) to ensure that the dosage and frequency of treatment are appropriate. Pupillometry is being increasingly investigated as a reliable, low-cost screen for drug use. The accuracy of measurement of pupil parameters from digital images has been proven (Iskander) showing that specialised ophthalmic instruments are unnecessary. Research has shown pupillometry to be effective in verifying concomitant drug use in methadone maintained patients (Murillo). A clear correlation between pupillary changes and methadone detoxification has been shown (Grunberger) as well as the use of pupillometry © STACK & JAMES 2010 Proposal for Remote Dosing Program 46 to objectively measure the intensity of opiate withdrawal in patients (Rosse). There is a clear relationship between sedation and pupillary function which may be linked to a reduction in central sympathetic activity caused by sedating drugs (Hou). Preliminary research into the assessment of opioid dependent patients using a combination of pupillometry, reaction time and speech slurring or reaction time testing alone have also been undertaken (Patil) and warrants further investigation. Supervision of medication dosing is a significant issue in the methadone maintenance program. There is substantial evidence from a range of studies which indicates that diversion and misuse (e.g. taking medication early, overdosing, injecting) are integral risks in the prescription of opioids (including methadone) (Bell). A study in Sydney in 1995 found an active black market in methadone with 88% of the drug sourced from diverted takeaway doses (Darke). A subsequent study in 2002 found diversion of the drug to the black market increased proportionally with unsupervised treatment (Darke). An analysis of policy differences regarding takeaway doses of methadone found that States/Territories with the most restrictive policies reported the lowest incidence of methadone injecting (Ritter). However, the same study also noted that the availability of heroin and dose dilution of takeaway doses impacted on the amount of methadone diversion (Ritter). Takeaway doses of methadone could be more widely prescribed if there was an adequately secure medication dispenser that could also facilitate assessment of the patient. Current medication dispensers have poor security (i.e. there is no check that the medication is properly dispensed or taken by the patient) and lack the ability to assess the patient’s need for medication. While work has been done on remote monitoring of medication dispensation, the prototypes require the patient to load the machine themselves (Hayes, Hassuji). Preliminary research has been undertaken into the development of a dispensing machine with appropriate security protocols to be used for opioid dependent patients and utilising a remote monitoring system to assess patients (Bajracharya, Ho). © STACK & JAMES 2010 Proposal for Remote Dosing Program 47 REFERENCES AIHW (Australian Institute of Health and Welfare) (2009). National Opioid Pharmacotherapy Statistics Annual Data Collection: 2008 Report . Bulletin No. 72. Cat no AUS 115. Canberra: AIHW Bajracharya, A., Gale, T.J., Stack, C.R., Turner, P. (2008). 3.5G Based Mobile Remote Monitoring System. Conf Proc IEEE Eng Med Biol Soc., 783-786 Bell, J. (Ed.) (2007). The Role of Supervision of Dosing in Opioid Maintenance Treatment. Available at: http://www.who.int/substance_abuse/activities/supervision_dosing.pdf Bell, J., Zador, D., (2000). A Risk-Benefit Analysis of Methadone Maintenance Treatment. Drug Safety, 22:3, 179-190 Connock, M., Juarez-Garacia, A., Jowett, S., Frew, E., Liu, Z, Taylor, R.J., FrySmith, A., Day, E., Lintzeris, N., Roberts, T., Burls, A., Taylor, R.S. (2007). Methadone and Buprenorphine for the Management of Opioid Dependence: A Systematic Review and Economic Evaluation. Available at: http://www.hta.ac.uk/execsumm/summ1109.htm Darke, S., Ross, J., Hall, W (1995). The Injection of Methadone Syrup, Australia. In Bell, J. (Ed.) (2007). The Role of Supervision of Dosing in Opioid Maintenance Treatment. Available at: http://www.who.int/substance_abuse/activities/supervision_dosing.pdf Darke, S., Topp, L., Ross, J. (2002). The Injection of Methadone and Benzodiazepines Amongst Sydney Injecting Drug Users 1996-200. In Bell, J. (Ed.) (2007). The Role of Supervision of Dosing in Opioid Maintenance Treatment. Available at: http://www.who.int/substance_abuse/activities/supervision_dosing.pdf Doran, C. (2007). Economic Evaluation of Interventions for Illicit Opioid Dependency: A Review of Evidence. Available at: http://www.who.int/substance_abuse/activities/economic_evaluation_intervent ions.pdf French, M.T., Salome, H.J., Carney, M. (2002). Using the DATCAP and ASI to Estimate the Costs and Benefits of Residential Addiction Treatment in the State of Washington. Social Science and Medicine. 55(12), 2267-2282 © STACK & JAMES 2010 Proposal for Remote Dosing Program 48 Godfrey , C., Steward, D., Gossop, M. (2004). Economic Analysis of Costs and Consequences of the Treatment of Drug Misuse: 2-year Outcome Data from the National Treatment Outcome Research Study (NTORS). Addiction, 99(6), 697-707 Gossop, M., Marsden, J., Steward, D., Treacy, S. (2001). Outcomes after Methadone Maintenance Reduction Treatments: Two-Year Follow-Up Results from the National Treatment Outcome Research Study. Drug and Alcohol Dependence, 79(3), 295-302 Grunberger, J., Linzmayer, L., Fodor, G.,Presslich, O., Praitner, M., Loimer, N. (1990). Static and Dynamic Pupillometry for Determination of the Course of Gradual Detoxification of Opiate-Addicted Patients. Eur Arch Psychiatry Clin Neurosci, 240, 109-112 Hassuji, A., Miljkovic, J., Sivananthan, T., Sivasubramaniam, V. (2007). Medical Dispenser including Adverse Drug Reaction Detection. Proceedings of ENGG 3100: Design III Projects, 53-54 Hayes, T.L., Hunt, J.M., Adami, A., Kaye, J.A. (2006). An Electronic Pillbox for Continuous Monitoring of Medication Adherence. IEEE EMBS Annual International Conference, New York City: USA, 6400-6403 Ho, V., Gale, T.J., Stack, C.R. (2009). Medical Dispenser Design for Narcotic Rehabilitation Patients. IEEE EMBC Annual Conference. Minnesota: USA Hou, R.H., Scaife, J., Freeman, C., Langley, R.W., Szabadi, E., Bradshaw, C.M. (2006), Relationship Between Sedation and Pupillary Function: Comparison of Diazepam and Diphenhydramine. Br J Clin Pharmacol, 61:6, 752-760 Iskander, D.R., Collins, M.J., Mioschek, S., Trunk, M. (2004). Automatic Pupillometry from Digital Images. IEEE Transaction on Biomed Eng, 51:9, 1619-1627 Murillo, R., Crucilla, C., Schmittner, J., Hotchkiss, E., Pickworth, W.B. (2004). Pupillometry in the Detection of Concomitant Drug Use in Opioid-Maintained Patients. Methods and Findings in Exper and Clin Pharmacol, 26:4, 271-275 Patil, S.G., Gale, T.J., Stack, C.R. (2007). Design of Novel Assessment Techniques for Opioid Dependent Partients. Conf Proc IEEE Eng Med Biol Soc. 3737-40 Patil, S.G, Gale, T.J., Stack, C.R. (2009). A Test for the Assessment of Reaction Time for Narcotic Rehabilitation Patients. 13th International Conference on Biomedical Engineering, 23, 769-773 © STACK & JAMES 2010 Proposal for Remote Dosing Program 49 Ritter, A., Di Natale, R. (2005). The Relationship Between Take-Away Methadone Policies and Methadone Diversion. Drug Alcohol Rev, 244, 347-352 Roebuck, C., French, M.T., McLellan, T. (2003). DATSats: Results from 85 Studies Using the Drug Abuse Treatment Cost Analysis Program (DATCAP). J Sub Abuse Treatment, 25, 51-57 Rosse, R.R., Johri, S., Goel, M., Rahman, F., Jawor, K.A., Deutsch, S.I. (1998). Pupillometric Changes During Graduate Opiate Detoxification Correlate with Changes in Symptoms of Opioid Withdrawal as Measured by the Weak Opiate Withdrawal Scale. Clinical Neuropharm, 21:5, 312-315 Ward, J., Hall, W., Mattick, R.P. (1999). Role of Maintenance Treatment in Opioid Dependence. The Lancet, 353, 221-226 Zaric, G.S., Barnett, P.G., Brandeau, M.I. (2000). HIV Transmission and the CostEffectiveness of Methadone Maintenance. Am Jn of Public Health, 90:7, 11001111 © STACK & JAMES 2010