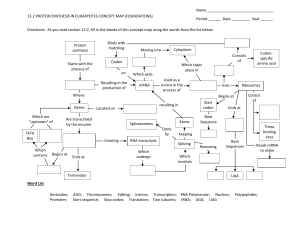
RNA Transcription and Modification

RNA Transcription and Modification
10/21/2008 6:02:00 PM
The central Dogma of Biology
DNA undergoes replication to make copies of itself. DNA also is transcribed to RNA, which in somecases is translated to protein.
Proteins affect cellular physiology so therefore DNA is the basis for the physiological state
RNA is a usually single stranded and is the complement of the template strand. The template is the antisense strand. The sequence corresponds to the coding (Sense) strand. General
Terminology
Element—something that affects transcription and is a DNA
SEQUENCE
Factor—a protein that affects transcription in any way
Cis Factor—a protein that affects the transcription of the same gene it is a product of.
Trans Factor—a protein that affects the transcription of a gene other than its own.
Promoter—DNA sequences that are recognized as initiation sites for
RNA synthesis
Terminators—RNA sequences that signal the RNA polymerase to stop synthesizing RNA
Consensus sequence—a given recognition sequence (or very much like it) that is found in all genes that are affected by a specific protein. (i.e. All proteins that work with TFIID have a TATA box because TFIID has a Tata Binding Protein-TBP. The TATA box is the consensus sequence)
PROKARYOTIC RNA TRANSCRIPTION
Does not require a primer
Does not compromise the integrity of the DNA duplex
Antisense strand is the template strand
RNA transcription is more error prone, but that is ok because an error in an mRNA just wont form the enzyme and there are more than one mRNA formed from the gene. If there were an error in
DNA replication, that defect would be lasting and be passed from generation to generation. The tolerance threshold is lower.
-RNA Polymerase.
RNA is synthesized in the 5’ 3’ direction (3’ hydroxyl of the existing chain makes nucleophilic attack on the phosphate.
Uses ribonucleotides precursors in the presence of Magnesium
4 subunits—2 (regulatory), (catalytic) and ’ (DNA binding)
There is no 3’ 5’exonuclease activity, therefore no editing
Only one RNA Polymerase for all types of RNA in prokaryotic
-Transcription Promoters—numbers in relation to initiation point (+1)
Prokaryotic o Minus 10 region (TATA Box aka Pribnow box) o Minus 35 region
Eukaryotic o Minus 25 Region (TATA Box aka Hogness box) o GC Rich (variable distance from +1 site)
Binds Sp1 o CAAT Box o A whole lot more too. There are more regulation elements in eukaryotic regulation due to complexity of organism and need for fine tuning of gene regulation
-Initiaiton—Sigma Factor
The sigma factor must be associated with the RNA polymerase core enzyme
The sigma factor allows for specific promoters to be bound lending a specificity to the core enzyme, which does not discriminate what it translates
The sigma factor recognizes the sequences to be transcribed.
After synthesis has begun, the Sigma Factor will dissociate from the core enzyme.
-Termination—two ways
Hair pin mediated o Following a GC rich sequence, the GC will form a stable hair pin structure. The GC rich sequence will be followed by A and
U sequence which has decreased binding affinity to the
Template strand. During transcription there are usually 10 nucleotide bound to DNA to keep the RNA on the template strand, but the hair pin causes most to dissociate leaving only
4-5 A and U which are not nearly as stable and will just fall off the strand.
Rho mediated o Rho hexamer binds to RNA and slides along the RNA strand by using ATP and causes dissociation.
-Lac Operon
Under glucose starved conditions, the Lac Operon will be turned on and a series of genes will be upregulated (the genes are said to be induced). -Galactosidase, permease and transacetylase. They are all regulated by the same promoter region and are transcribed on the same RNA (termed polycistronic because it codes for more than one protein). -Gal cleaves lactose into glucose and galactose and permease makes lactose able to enter the cell.
Under normal conditions, the product of the repressor region (I region) will form tetramers and bind the Operator region (O region) and inhibit the transcription of the operon.
Under glucose starved conditions, Allolactose will bind the repressor proteins, which are the product of the repressor region (I region), and disallow them to for the tetramer that binds to the operator and allow transcription of the gene.
-Inihbitors of Transcription
Rifampcin-Binds the Subunit, thereby blocking the catalytic mechanism of the subunit
Alpha-Amanitin-Toxin that indiscriminately binds to RNA Pol II and
RNA Pol III
Actinomycin D-intercalating agent that blocks both transcription (all doses) and replication (high doses)
EUKARYOTIC TRANSCRIPTION
-more complicated than prokaryotic…there are more steps and the whole thing about having a nucleus with a nuclear envelope that has to be dealt with (compartmentalization)
-RNA Polymerases
RNA Pol I—Synthesis of ribosomal RNA in the nucleolus
RNA Pol II—Synthesis of hnRNA (hnRNA mRNA) in the nucleoplasm
RNA Pol III—Sythesis of tRNA and some snRNA in the nucleoplasm
mtRNA Polymerase—Synthesis of mitochondrial RNA in the mitochondrial matrix
-Control of Gene Expression
Gene structure o Active chromatin is less condensed so that there is more access to the genetic material. o Histone H1 is missing or depleted in active chromatin o Topoisomerases are frequently found associated with active chromatin o Core histones are acetylated in activated Chromatin
Acetylation changes the charge on the histones from positive to negative, causing the DNA to dissociate from the core histone octamer. o DNA tends to be hypomethylated in active chromatin
Enzymes that methylate decrease transcription
Enzymes that demethylate increase transcription
Initation o There are more elements of the core promoter in Eukaryotic
Transcription than in Prokaryotic transcription o TFIIB Recognition Element is a DNA sequence recognized by
TFIIB that helps recruit the transcription machinery. It is part of the Pre-Initiation Complex. TFIIB Recognition Element is usually directly distal to the initiation point from the TATA Box
(-37—-32) o TATA Box binds to TATA Binding Protein (TBP). TBP is part of
TFIID. TFIID also helps recruit Translation Machinery and is part of the Pre-Initiation Complex. (-31—-26) o There is an Initiator Consensus Sequence that spans the nucleotides just flanking either side of +1. (-2—+4) o Downstream Core Promoter Element is another aspect of the core promoter that is very common in genes, but not necessarily in all genes. It is a consensus sequence that is approximately +28—+32. o Pre-Initiation Complex
1.
TFIID Binds to the TATA box by way of TBP
2.
TFIIA binds
3.
TFIIB binds
4.
TFIIF binds
5.
RNA Pol II is recruited by the TFIID/A/B/F complex
6.
TFIIE binds o Sp1 binds to the GC rich region and influences the adapter protein attached to TBP. The GC rich region that Sp1 binds with can be very far away from the gene it is influencing. o Steroid Hormones directly act on gene expression by acting directly with receptors that are transcription factors. The hormones bind with the receptor and the receptor hormone complex and act on enhancer sequences to recruit transcription machinery. o Tamoxifen directly inhibits DNA transcription by binding to its receptor and acting as a replication inhibitor. The Tamoxifen-
Receptor complex bind to the enhancer sequence and recruit transcription inhibitor factors o Enhancers and Silencers are DNA sequences that are not part of the core promoter element. They may be part of an intron or very far from the gene that it is affecting. E and S bind transcription factors and either activate or repress gene expression (activators bind to enhancers and repressors bind to silencers) o Homeotic Genes-homeobox proteins control the functional architectural plan of the embryo by regulating transcription of genes. HGs are extremely conserved from flies to humans.
These proteins have a portion that have a very basic charge that can bind DNA.
Waardenburg Syndrome is caused by a defect in homeobox protein HuP2 in humans (PAX3 in mice).
Waardenburg presents with a patch of white hair in the front of the hair line, cochlear deafness, heterochromia iridis, wide bridge of the nose due to placement of the inner canthus of each eye.
FosB deficieny mice showed less nuturing behavior than wild type. This shows that gene expression can affect behavior
Transcript Processing o 5’ Cap
7’methylguanylate attached by a 5’-5’ triphosphate linkage
Aids in translation because the cap is recognized by proteins (TFA and p220)
Stabilizes the RNA for transport to the cytoplasm for translation o Poly A Tail
After cleavage of growing mRNA strand from RNA
Polymerase II, the poly A tail is added by Poly A
Polymerase o Splicing
Eukaryotic genes come chocked full of introns (non coding regions of RNA). The introns are spliced out by forming a lariat type structure and the snRNAs are used to help this process.
Introns—Regions of the gene that are missing from the final mRNA product.
Exons—Regions of the gene that are in the final mRNA product
Some genes are not split by introns, but most are.
Histone Genes are not, some interferon genes are not, and some viral genes are not.
Splicing defect can be the root of some diseases
Thalassemia syndromes (mutation in a globin chain) can be caused by splicing defects. They can also be coded for by nonsense and frame shift mutations or stop-codon mutations
Mutations in splice start stop signals can cause mutations in the end protein product (truncated, shortened or lengthened, framshift)
RNA Translation
10/21/2008 6:02:00 PM
The Code
Three nucleotides makes up a codon. They do not overlap and are read in succession. (2 bases per codon only has room for 16 amino acids)
The Open Reading Frame (orf) is the span of codons translated without being interrupted by a stop codon (UGA UAA UAG)
The genetic code is indeed degenerate because there is more than one codon for many of the amino acids. There are 64 codons and only 20 amino acids. WTF mate!
Mammalian Mitochondrial Code is a bit different o AGA and AGG are additional stop codons o AUA codes for Me instead of Ile o UGA is a Trp instead of STOP
Yeast Mitochondrial Code is also different o TGA is a Trp instead of a STOP o CUX is Thr instead of Leu
Mutations
Deletion-(a) nucleotide(s) are removed from the code.
Insertion-(a) nucleotide(s) are inserted into the code.
Frameshift-Deletions or insertions that are not multiples of 3 will cause all the codons after the point of mutations to code for different genes.
Null Mutation-causes the gene to not produce a function
Nonsense mutation-causes a termination codon to replace an amino acid codon in the sequence
Point mutation-change in one nucleotide
Ribosomes
Two asymmetric ribonucleoproteins that form the functional machinery of translation. o Prokaryotic are the 30S and 50 S subunits o Eukaryotic are the 40S and 60 S subunits o S-value is the sedimentation coefficient (not additive)
All ribosomes of a given cell are identical
Ribosomes are reused after they are released from the mRNA
There is a peptidyl transferase site on the large subunit
There s tRNA site on the small subunit.
Multiple Ribosomes on one mRNA form Polysomes
Transfer RNA (tRNA)
3’ CCA terminus is highly conserved and is where the amino acid binds o Amino acids are attached to tRNA by way of Aminoacyl tRNA
Synthases. Each tRNA has its own.
The Anti Codon loop is where the tRNA makes direct contact with the mRNA.
tRNA has some funky bases (more than just AUGC, there are modified bases of these, but also inosine)
Charged tRNA is the term used to describe a tRNA with an amino acid covalently bound to the 3’ OH terminus. Charging requires expenditure of energy by use of ATP (two steps-actiavtion of amino acid which is then transferred to the tRNA and releases AMP.
Initiation
Prokaryotic o mRNA proceeds through the ribosome 5’ 3’. The 5’ end binds to the small subunit. It then slides down toward the 3’ end and when it encounters the Shine-Delgarno Sequence, the next AUG will be the first Methionine laid. The First
Methionine is Formyl-Methionine (fMet) o fMet is bound by a fMet receptor on neutrophils which causes chemotactic migration toward the site of infection. o Only uses three initiation factors: IF1, IF2, IF3
Eukaryotic o The small subunit slides down the mRNA and will if an AUG that is encountered by the Kozak sequence, translation will initiate.
The Kozak sequence is the optimal sequence surrounding the AUG start codon for translation to initiate. o Requires 9 transcription factors—2 parts of initiation
Activation of mRNA
P220 and 4F will bind to the 5’ methyl cap and
4A/4B will unwind any secondary structure.
Assembly of the pre-initiation complex
The small subunit is associated with 1A and 3.
The incoming Met-tRNA will have F2.
The Met-tRNA will bind to the small subunit and this is called the pre-initiation complex
The small subunit pre-initiation complex will then be able to bind the mRNA and start the search for the ideal start location. o Codon-Anticodon base pairing
mRNA is threaded through 5’ 3’ and the anticodon binds antiparallel
After the pre-initation complex has found the Kozak sequence, it will recruit the large subunit
Elongation in Eukaryotes
The growing peptidyl-tRNA will be bound in the P site. The incoming amino acyl tRNA will enter at the A site. The N of incoming amino acid will have nucleophilic attack on the carbonyl C of the growing peptidyl chain.
EF2 mediates the transfer of the peptidyl tRNA from the A site to the site with he hydrolysis of GTP.
Termination in Eukaryotes
The stop codon causes eRF-GTP to enter the A site and peptidyl transferase will cleave the bound GTP to GDP. The polypeptide chain will be released from the tRNA. The tRNA and eRF will release and then the ribosome will dissociate.
Wobble Theory
Different codons are recognized by different anticodons. The third base can sometimes be different (more than one base is tolerated by the third position)
A separate tRNA is NOT necessary for every codon because the first base in the anticodon has variable binding (the first anticodon base interacts with the third codon base)
Wobble is responsible for the degeneracy.
Wobble pairings
Anticodon Codon
C G
A
U
U
A or G (either purine)
G U or C (either pyrimidine)
I
Inhibition of translation
U C or A (not guanisine
Antibiotics
Toxins o Diptheria Toxin—Inactivates EF2 by forming ADP-ribosyl EF2.
The toxin is two subunits A and B. The toxin binds to the membrane and is then cleaved. The B subunit facilitates the entry of the A subunit into the cytoplasm. The A subunit is an enzyme that catalyzes the synthesis of ADP ribosyl EF2.
Post-Translational Modification
10/21/2008 6:02:00 PM
Membrane Anchoring to the membrane by covalent addition of fatty acid chains—Compartmentalization to the membrane
Ras Protein is covalently bonded to palmitoyl CoA (CoA is released).
Cysteine is the amino acid involved (thiol bond)
Src protein is covalently bonded to myristyl CoA (CoA is released).
The terminal amino is covalently linked to the FA by an Amide Link.
GPI anchoruses a glycophosphotidyl inositol. This anchors the protein to the lipid rafts and further compartmentalizes the lipid rafts in the membranes.
Protein Glycosylation
O-Linked—Linked to the OH of a serine residue
N-Linked—Linked to the amino group of an asparagine residue o Glucosyl transferase transfers an entire oligosaccharide from dolichol to the protein. Targets Asn-X-Thr and Asn-X-Ser. As soon as an “eligible” Asn passes through machinery in ER, it is glycosilated.
Proteolytic Processing
The inactive form of the gene is translated and is only activated when a portion is cleaved off. This is the prepropetide situation.
The prepropeptide is cleaved to form the propeptide and then cleaved again to form the active peptide.
Cleavage is preferential between two basic residues.
Is common if the protein is needed to have rapid activation. The inactive peptide can be swimming around and not do anything until the stimulus is sent to cleave the pre pro crap off.
Proteolytic processing is also useful if polycistronic mRNA is translated all as one protein. o HIV-1 gag-pol Fusion protein.
Intracellular degredation
Energy Dependant o ATP is required to ubiquitinate the undesirable protein o Short lived proteins
ATP independent o Normal degredation by proteases in the lysosomes after uptake by endocytosis of other stuff. o Long lived proteins and membrane bound proteins
Objectives
10/21/2008 6:02:00 PM