Orders - Gbhn.ca
advertisement

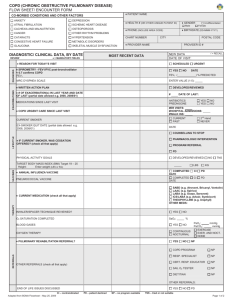
GREY BRUCE HEALTH NETWORK ADDRESSOGRAPH Page 1 of 4 SITE: _____________________ Allergies: □ NKA or: ________________________ Weight (kg) _______________ Processed Chronic Obstructive Pulmonary Disease (COPD) Order Set Open Box indicates optional order, activated when checked . Checked box indicates mandatory order unless crossed out. Admit to _________________: Dr. __________________ to consult or assume MRP Diagnosis: COPD: with pneumonia __________________________________ Isolation: Contact Droplet Airborne re: ____________ ARO Droplet/Contact Comorbidities: a) ____________ b) _______________ c) _______________ ________ ________ ________ ________ ________ Code Status: ________ Full Resuscitation No CPR Defibrillation only No Defibrillation No Intubation Do Not Resuscitate ________ Family Physician: Same as MRP, or __________________________________ Clinical Pathway: Cerner Order for COPD Pathway Kardex ________ ________ ________ ________ ________ ________ ________ Consults ________ ________ ________ ________ CCAC Clinical Nutrition Physiotherapy Social Work Discharge Planning Occupational Therapy Palliative Care Nurse Pharmacy Speech/Language Pathology Respiratory Therapy (to teach regarding puffer use) ________ ________ ________ Regular Diet ________ Activity as Tolerated ___________________ Reason: __________________________ Diet Regular Diet, NPO after 2400 hrs May take meds with sips if NPO NPO Energy Controlled Diet ________kcal Other Diet: ______________________________ Healthy Heart Diet Activity ________ ________ ________ ________ ________ ________ ________ ________ Ambulate within 8h and TID Vital Signs VS + O2 sats q4h x 24h, qid x24h then bid when stable VS + O2 sats q4h ________ VS + O2 sats qshift ________ ________ ________ O2 to keep O2 sats greater than 92% COPD Patient: O2 to keep O2 sats 88% - 92% 02 ________L/minute via NP 02 ________________________ VS + O2 sats q ________h Respiratory ________ ________ Patient Care ________ Direct Care: ________ Tubes/Drains: Height and Weight on admission Daily Weight Chart fluid intake and output x 24 h then reassess Foley Catheter NG tube suction NG tube straight drain Physician’s Signature __________________________Date ___________Time __________ C/1/GBHN/Med/-/COPD/MD/07-07/v1/- Copyright © 2007 Grey Bruce Health Network NOTE: this is a CONTROLLED document as are all files on this server. Any documents appearing in paper form are not controlled and should ALWAYS be checked against the server file versions (electronic version) prior to use. ________ ________ GREY BRUCE HEALTH NETWORK ADDRESSOGRAPH Page 2 of 4 SITE: _____________________ Allergies: □ NKA or: ________________________ Weight (kg) _______________ Chronic Obstructive Pulmonary Disease (COPD) Order Set Processed ________ POC: POC Capillary Glucose QID x 2 days POC Capillary Glucose daily Kardex ________ Laboratory (Order details blood and routine unless otherwise noted) On admission (if not already done in ER): ________ CBC APTT INR ________ Lytes, Creatinine, Glucose ABG CPK, Troponin Calcium, Magnesium ________ ALT, ALP, Bili AST Culture Sputum ________ Culture Blood x 2 STAT Urinalysis Culture Urine ________ Additional Labs: ________________________________________________ Day 1 (first morning post admission): ________ CBC, APTT INR ________ Lytes, Creatinine CPK, Troponin Calcium, Magnesium ________ ALT, ALP, Bili AST ________ Additional AM Labs: _____________________________________________ ________ ________ Additional Lab Tests: CBC in AM on day two of admission Lytes, Creatinine, Glucose in AM on day two of admission Culture Blood x 2 if temp greater than or equal to 385 °C ________ ________ ________ ________ ________ ________ ________ ________ ________ ________ ________ ABG if O2 sats less than 90% ________ Additional follow up labs: __________________________________________ ________ Diagnostic Tests ________ ________ ________ ________ ________ CXR PA and Lateral (next morning if after hours) ECG with chest pain and notify MD ECG on admission ________ ECG Day 1 CT Scan ____________ Re: ____________________________________ Ultrasound __________Re: ____________________________________ 2D echo Re: _______________________________ Doppler US to R/O DVT _______________________________________________________ ________ ________ ________ ________ IV Solutions ________ Bolus IV: __________________________________________________ After Bolus IV finished: NS With 20 mmol KCl per L of IV fluid ________ IV Fluid: 2/3 1/3 ________ Other ________________ With 40 mmol KCl per L of IV fluid ________ Rate ___________________ mL/h Decrease IV to TKVO when drinking well. Discontinue IV if Pt not on any IV medications. Saline Lock Physician’s Signature __________________________Date ___________Time __________ C/1/GBHN/Med/-/COPD/MD/07-07/v1/- Copyright © 2007 Grey Bruce Health Network NOTE: this is a CONTROLLED document as are all files on this server. Any documents appearing in paper form are not controlled and should ALWAYS be checked against the server file versions (electronic version) prior to use. ________ ________ ________ ________ GREY BRUCE HEALTH NETWORK ADDRESSOGRAPH Page 3 of 4 SITE: _____________________ Allergies: □ NKA or: ________________________ Weight (kg) _______________ Chronic Obstructive Pulmonary Disease (COPD) Order Set Processed Kardex Medications Antibiotic Therapy: **Antibiotics are indicated when exacerbations of COPD are accompanied by two of the following signs: increased dyspnea, increased sputum or increased sputum purulence** ________ **If the patient has been on antibiotic therapy in the last 3 months (regardless of clinical success), the therapy chosen should be a regimen based on a different mechanism of action.** Moxifloxacin 400 mg IV x 1 dose STAT, then Moxifloxacin 400 mg PO daily x 10 days ________ ________ **HDH Only** Levofloxacin 500 mg IV x 1 dose STAT, then Levofloxacin 500 mg PO daily x 10 days ________ ________ ________ Anticholinergic bronchodilators: ________ ________ ________ ________ Azithromycin 500 mg PO on admission, then Azithromycin 250 mg PO daily x 4 days Ipratropium 40 mcg inhaled QID OR Tiotropium 18 mcg inhaled once daily ________ ________ 5 mg inhaled Q4H 2.5 mg inhaled Q1H PRN ________ ________ Short-acting β-agonist (SABA): salBUTAMol 2.5 mg inhaled Q4H 200 mcg inhaled Q4H ________ Long-acting β-agonist (LABA): ________ Inhaled steroid: 200 mcg inhaled Q6H 5 mg inhaled Q1H PRN salMETERol 50 mcg INH BID Fluticasone 500 mcg inhaled BID ________ ________ ________ Other Bronchodilators: ___________________________________________________ ________ ________ ________ Monitor response to treatment with peak expiratory flow bid ________ Steroid Therapy: ________ ________ ________ ________ ________ methylPREDNISolone 125 mg IV Q6H X 3 days then predniSONE 50 mg PO daily X 10 days, discontinue with no taper PRN Medication: **max Acetaminophen from all sources 4000 mg/24h** Acetaminophen 650 mg PO or NG or Rectally q4h PRN Aluminum hydroxide/magnesium hydroxide oral suspension 30 mL PO q4h PRN dimenhyDRINATE 25-50 mg IV or NG or PO q4h PRN Bowel Care Clinical Protocol Adult Potassium Oral Dosing Clinical Protocol ________ Smoking Cessation: ________ Sedation: ________ Nicotine transdermal patch _____mg/day Lorazepam 1 mg PO or sublingual qhs PRN, OR Zopiclone 3.75-7.5 mg PO QHS PRN Physician’s Signature __________________________Date ___________Time __________ C/1/GBHN/Med/-/COPD/MD/07-07/v1/- Copyright © 2007 Grey Bruce Health Network NOTE: this is a CONTROLLED document as are all files on this server. Any documents appearing in paper form are not controlled and should ALWAYS be checked against the server file versions (electronic version) prior to use. ________ ________ ________ ________ ________ ________ ________ ________ ________ GREY BRUCE HEALTH NETWORK ADDRESSOGRAPH ADDRESSOGRAPH Page 4 of 4 SITE: _____________________ Allergies: □ NKA or: ________________________ Weight (kg) _______________ Processed Chronic Obstructive Pulmonary Disease (COPD) Order Set Kardex Diabetes Management Protocols ________ Hypoglycemia Clinical Protocol greater than or equal to 16 years Adult Subcutaneous Insulin Order Set ________ DVT Prophylaxis Protocol ________ ________ ________ ________ If Creatinine clearance is greater than 30 mL/min, consider (check with pharmacy to determine calculated Creatinine clearance – Height, Weight and serum creatinine are required for this calculation): Enoxaparin 40 mg subcutaneous once daily. Dalteparin 5000 units subcutaneous once daily (HDH only). If Enoxaparin/Dalteparin ordered, then CBC, APTT, INR, Creatinine prior to initiating therapy if not already ordered CBC day 1, 3, 7, and 14 to monitor platelet count CBC weekly for patients on therapy greater than 14 days If Creatinine clearance is less than 30 mL/min OR serum Creatinine greater than 150mmol/L, consider: Heparin 5000 units subcutaneous q12h. If Heparin ordered, then CBC, APTT, INR, Creatinine prior to initiating therapy if not already ordered CBC, day 1, 3, 7, and 14 to monitor platelet count CBC weekly for patients on therapy greater than 14 days Anti-emboli Stockings: Knee high Thigh High Reassess DVT prophylaxis therapy when patient is ambulating and on day of discharge __________________________________________________________ __________________________________________________________ __________________________________________________________ __________________________________________________________ __________________________________________________________ __________________________________________________________ __________________________________________________________ __________________________________________________________ Physician’s Signature __________________________Date ___________Time __________ C/1/GBHN/Med/-/COPD/MD/07-07/v1/- Copyright © 2007 Grey Bruce Health Network NOTE: this is a CONTROLLED document as are all files on this server. Any documents appearing in paper form are not controlled and should ALWAYS be checked against the server file versions (electronic version) prior to use. ________ ________ ________ ________