1 - Hopkinton School District
advertisement

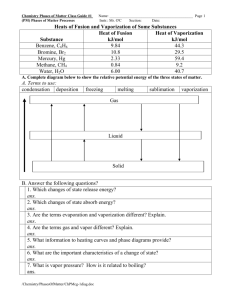
1. a. If you were told to weigh out 0.02 moles of sodium sulfate, how many grams would you place on the balance? b. How many formula units (think molecules) of sodium sulfate would you have? c. How many oxygen atoms would you have? 2. One form of Carvone, C10H14O, gives caraway seeds their characteristic smell, the other type is responsible for the smell of spearmint oil. Compute the mass percent of each element in carvone. 3. An unknown compound contains only carbon, hydrogen, and nitrogen. It was determined to have 38.67% carbon and 16.22% hydrogen. What is its empirical formula? 4. Caffeine, a stimulant found in coffee, tea, and chocolate, contains 49.48% carbon, 5.15% hydrogen, 28.87% nitrogen and 16.49% oxygen by mass and has a molar mass of 194.2 g/mol. Determine the molecular formula of caffeine. 5. Determine the empirical and molecular formula of a compound that gives the following analysis (in mass percents): 71.65% Cl 24.27% C 4.07% H The molar mass is known to be 98.96 g/mol 6a. A white powder is analyzed and found to contain 43.64% phosphorus and 56.36% oxygen by mass. The compound has a molar mass of 283.88 g/mol. What are the compound’s empirical and molecular formulas? b. Write and balance the combustion equation that will form this product. 7. Write and balance the combustion equation of ethanol, C2H5OH 8. Solid lithium hydroxide is used in space vehicles to remove exhaled carbon dioxide from the living environment by forming solid lithium carbonate and liquid water. Write and balance this chemical equation. 9. What volume will 12.0 x 1024 molecules of oxygen gas occupy at STP? 10. What is the mass of 10.0 liters of argon gas at STP? 11. Write the chemical equation showing the combustion of glucose, C6H12O6(s) 12. Balance the following: a. Al(OH)3(s) + HCl(aq) = AlCl3(aq) + H2O(l) b. Write the word equation that describes the above reaction c. Fe(s) + O2 (g) = Fe2O3(s) d. Write the word equation that describes the above reaction e. NaHCO3(s) = Na2CO3(s) + CO2(g) + H2O(g) 13. Write the NET Ionic equations for the following reactions: a. Hg2SO4(aq) + CaCl2(aq) = b. (NH4)2CO3(aq) + BaCl2(aq) = c. KI(aq) + NaNO3(aq) = d. AlCl3(aq) + Ca(OH)2(aq) = Ans 2: C 79.96%, H 9.394% O 10.65% Ans 3: CH5N Ans 4: C8H10N4O2 Ans 5: Emp: ClCH2, Molecular: Cl2C2H4 Ans 6: Emp: P2O5, Molecular: P4O10 Ans 8: 2LiOH(s) + CO2(g) = Li2CO3(s) + H2O(l) Ans 9: 446 L Ans 10: 17.8 g Ar Ans 12c: coefficients4, 3, 2 Hint 12e: balance oxygen last Hint 13a: two ppts formed 13b: BaCO3 ppt 13c: NR 13d: Al(OH)3 ppt