Recover Total Nucleic Acid from FFPE Samples with Ambion's
advertisement

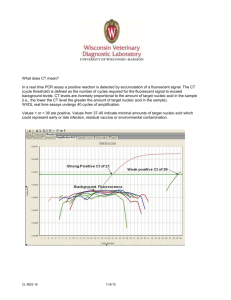
RN A I S O L AT I O N Recover Total Nucleic Acid from FFPE Samples with Ambion's RecoverAll™ Total Nucleic Acid Isolation Kit • No overnight Proteinase K digestion required–deparaffinize in the morning and perform qRT-PCR in the afternoon Deparaffinization 1. Assemble FFPE sections equivalent to < –80 µm or <–35 mg unsectioned core • Isolate total nucleic acids, including microRNAs, from FFPE tissue • Obtain typical yields of >50% that of unfixed tissue from the same sample source • Recovered nucleic acids are suitable for qRT-PCR, qPCR, and mutation screening • Up to four 20 µm sections or up to 35 mg of unsectioned core samples can be processed per reaction Many standard preservation techniques for biological tissue sample storage employ formaldehyde to maintain tissue structure and prevent putrifecation. However, this type of preservation makes it difficult to perform molecular analysis on the samples since nucleic acid is both trapped and modified by significant protein-protein and protein-nucleic acid crosslinks. Researchers at Ambion and others (Masuda 1999) have discovered that the fixation process itself does not necessarily lead to nucleic acid fragmentation. Instead, the embedding process, which requires high temperatures and vacuum for the paraffin to penetrate the tissue, quickens chemical reactions which subsequently modify RNA and DNA. These modifications lead to fragmentation over time. 20 min 3. Centrifuge for 2 min at maximum speed, and discard the xylene 4. Wash the pellet twice with 1 ml 100% ethanol and air dry Protease Digestion 1. Add Digestion Buffer and Protease 3 hr or 48 hr 2. Incubate at 50°C for 3 hr for RNA isolation and 48 hr for DNA isolation Nucleic Acid Isolation 1. Add 480 µl Isolation Additive and vortex 2. Add 1.1 ml 100% ethanol and mix 10 min 3. Pass the mixture through a Filter Cartridge 4. Wash with 700 µl of Wash 1 Deparaffinize in the Morning, Perform qRT-PCR in the Afternoon The RecoverAll™ Total Nucleic Acid Isolation procedure requires about 45 minutes of hands-on time and can easily be completed in less than 1 day when isolating RNA. FFPE samples are deparaffinized using a series of xylene and ethanol washes. Next, they are subjected to a rigorous protease digestion with an incubation time tailored for recovery of either RNA or DNA. Nucleic acids are purified using a rapid glass-filter methodology that includes an on-filter nuclease treatment and are eluted into either water or the low salt buffer provided. 2. Add 1 ml 100% xylene, mix, and incubate for 3 min at 50°C 5. Wash with 500 µl of Wash 2/3 Nuclease Digestion and Final Purification 1. Add DNase or RNase mix to each Filter Cartridge and incubate for 30 min 45 min 2. Wash with 700 µl of Wash 1 3. Wash twice with 500 µl of Wash 2/3 4. Elute nucleic acid with 2 x 30 µl Elution Solution or nuclease-free water RNAi • miRNA • RNA SAMPLE PREPARATION • RNA AMPLIFICATION • RNA RESEARCH PRODUCTS pfd_RecoverAll.indd 1 4/20/06 12:39:45 PM Maximize Yield from Archived and Freshly Fixed Tissue Ct 20 15 10 5 0 Isolating DNA from FFPE Samples Although DNA does not fragment as easily as RNA, it is more sequestered by the formaldehyde treatment, and the DNA isolation protocol therefore requires a much longer (2 day) protease digestion time to release substantial amounts of DNA. Recovered DNA can typically be used for PCR and other downstream applications. Total Amount of RNA (µg) RecoverAll Competitor B Competitor A PKC Recc1 RNAPol II FAS DDPK Untreated Nucleic Acid (Control) 15 12.5 DNase-treated Nucleic Acid (RNA) 10 7.5 RNase-treated Nucleic Acid (DNA) 5 2.5 0 14 19 24 29 34 39 44 49 Time (seconds) 54 59 64 Agilent® 2100 bioanalyzer Scan Demonstrating Presence of RNA and DNA from FFPE Mouse Liver. Nucleic acids were isolated from FFPE mouse liver (20 µm section, fixed and embedded using standard hospital protocol) using the RecoverAll™ Total Nucleic Acid Isolation Kit. Three equal amounts of sample (based on A260) were treated as follows: untreated control received no DNase or RNase treatment; RNA was isolated by DNase treatment of the nucleic acid sample; DNA was isolated by RNase treatment of the nucleic acid sample. An equal volume of each was analyzed on the Agilent 2100 bioanalyzer. 8 7 6 5 Brain 4 3 2 1 0 UBC 20 F Heart Z F Kidney Z F Z Liver F Z 30– Lung Colon Breast Kidney Bladder Yield of RNA from Archived Human FFPE Tissue Samples: RecoverAll™ vs. Two Competitor Systems 20– ORDERING INFORMATION 10– CAT# SIZE RecoverAll™ Total Nucleic Acid Isolation Kit 1975 40 purifications RELATED PRODUCTS CAT# SIZE RNAi •® miRNA • RNA SAMPLE PREPARATION RNaseZap 9780–9784 • RNA TURBO DNase™ RETROScript® GAPDH 17.5 Obtain microRNA for Expression Profiling MicroRNAs (miRNAs) are highly conserved regulatory molecules expressed in eukaryotic cells. Active, mature miRNAs are 17–24 bases long and are known to affect the translation or stability of target messenger RNAs. The nucleic acid recovered using the RecoverAll Kit includes the full component of miRNA. These miRNA can be used for miRNA expression profiling when used in combination with the flashPAGE™ Fractionator System and mirVana™ miRNA Array Technology. TBP Ct Values from Real-Time RT-PCR of Frozen and FFPE Mouse Brain. Half of each of four mouse brain samples were flash frozen in liquid nitrogen then stored at –80ºC; the other half was fixed and embedded using a standard hospital protocol. RNA was isolated from one 20 µm slice from each FFPE mouse brain using the RecoverAll™ Total Nucleic Acid Isolation Kit. RNA from the frozen controls was isolated using the mirVana™ miRNA Isolation Kit. RNA (400 ng) from each sample was used in two-step real-time RT-PCR. cDNA was synthesized using the RETROscript® Kit. Random decamer primers, one tenth of the RT reaction, and SuperTaq™ Polymerase were used for PCR. Each bar represents the mean ± standard deviation for 8 replicates. TBP=TATA Binding Protein; GAPDH=Glyceraldehyde-3-phosphate Dehydrogenase; UBC=Ubiquitin C; PKC=Protein Kinase C; Recc1=Replication Factor C; RNAPol II=RNA Polymerase II; FAS=Fatty Acid Synthase; DDPK=DNA Activated Protein Kinase. Fluorescence The ability to isolate nucleic acid that is suitable for molecular analysis from archived tissue samples provides a powerful tool in retrospective studies of diseased tissue at both the genomic and gene expression level. The recovered nucleic acids are suitable for downstream applications such as endpoint RT-PCR, PCR, qRT-PCR, and mutation screening. FFPE 25 Gene Expression from FFPE Samples 10 9 Frozen 30 The degree of RNA fragmentation that has already occurred in FFPE tissues cannot be reversed. However, the nucleic acid isolation of the RecoverAll Kit are designed to release a maximal amount of RNA fragments of all sizes, including microRNAs, in a relatively short amount of time (3 hours). let-7 miRNA from FFPE and Flash-frozen Mouse Tissue Samples. Total RNA was isolated from four different mouse tissues that had been frozen (F) or formalin-fixed and paraffin-embedded (Z). This Northern blot (1 µg RNA/sample) was incubated with an oligonucleotide probe specific for let-7 and verified the presence of intact let-7 miRNA. AMPLIFICATION • RNA RESEARCH PRODUCTS 250 ml – 4 L 2238 1,000 U Kit 1710 40 rxn flashPAGE™ Fractionator System 13100 1 each SuperTaq™ DNA Polymerase 2050 50 U RT-PCR Grade Water 9935 10 x 1.75 ml Learn more about Total Nucleic Acid Isolation Kit at www.ambion.com/prod/recoverall RecoverAll™ ORDER ONLINE ANYTIME AT WWW.AMBION.COM flashPAGE™, mirVana™, RecoverALL™, TURBO DNase™, and SuperTaq™ are trademarks of Ambion, Inc. RETROscript® and RNaseZap® are registered trademarks of Ambion, Inc. Agilent® is a registered trademark of Agilent Technologies.. Ambion, Inc. U.S. 800-888-8804 • Canada 800-445-1161 fax +1-512-651-0201 pfd_RecoverAll.indd 2 Ambion (Europe) Ltd tel +44 (0)1480-373-020 fax +44 (0)1480-373-010 Ambion K.K. (Japan) tel +81 (0)3-5638-2181 fax +81 (0)3-5638-2182 For a list of Ambion direct free phone numbers and distributors in your country, go to www.ambion.com/contact 4/20/06 12:39:50 PM