to read the review
advertisement

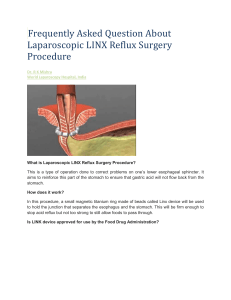
2) Esophageal sphincter device for gastroesophageal reflux disease Ganz RA, Peters JH, Horgan S, Bemelman WA, Dunst CM, Edmundovicz SA, Lipham JC, Luketich JD, Melvin WS, Oelschlager BK, Schlack-Haerer SC, Smith CD, Smith CC, Dunn D, Taiganides PA: N Engl J Med 2013;368:719-27 Aim and study design A multicenter trial was designed to assess the safety and effectiveness of a magnetic device (LINX Reflux Management System, Torax Medical, Minneapolis, USA) that augments the resistance of the lower esophageal sphincter to reflux. Between January and September 2009, 100 patients with gastroesophageal reflux disease (GERD) were enrolled in 13 centers in the United States and one in Netherlands. Eligible individuals were 18 to 75 years of age. All had at least a 6-month history of GERD, a partial response to daily proton-pump inhibitors (PPI), and an abnormal esophageal acid exposure documented by pH monitoring. Patients with large hiatal hernia, grade C or D esophagitis, body-mass index greater than 35, Barrett’s esophagus, esophageal motility disorders, dysphagia, and allergy to nickel were excluded from the study. In addition to endoscopy and pH monitoring, all patients underwent esophageal manometry and barium esophagogram. Annual screening was planned for a total 5 years. The magnetic device was implanted through a standard laparoscopic approach by academic or community surgeons who had experience with fundoplication. The primary end-points were the number of patients in whom acid exposure was normalized or was decreased by 50% or more. The secondary end-points were the number of patients with a reduction of 50% or more in the total score for quality of life, and a reduction of 50% or more in the dose of PPI. End-point analyses were performed according to the intention-totreat principle. Results The median duration of therapy with PPI was 5 years. The median percent time with pH below 4 during a median pH-monitoring period of 45 hours was 10.9% (4.8-25.4), and the median DeMeester score was 36.6 (16.3-83.8). The median quality of life score was 27 off-PPI and 11 on-PPI. The median time required for device implantation was 36 minutes. No reversions to fundoplication nor intraoperative complications occurred. All patients were discharged home within one day. The primary end-point, i.e. normalization or a 50% reduction in esophageal acid exposure, was obtained in 64% of patients (95% confidence interval 54-73). The secondary end-point, i.e. a 50% reduction in the quality of life score, was obtained in 92% of patients (95% confidence interval 56-96). Finally, 93% of patients reported a 50% or more reduction in the average daily dose of PPI. The median quality of life score was significantly decreased at one year post implantation and remained stable at 2 and 3 years (p<0.005). All individual components of the pH monitoring significantly improved after surgery (p<0.001). Dysphagia was the most frequent adverse event and was reported by 68% of the patients. Endoscopic dilatation was performed in 19 patients and was successful in 16. The percentage of patients with esophagitis significantly decreased after operation (p<0.001). Removal of the device was required in 6 patients because of persistent dysphagia (n=3), persistent reflux symptoms (n=1), persistent chest pain (n=1), and intermittent vomiting (n=1). The procedure of removal was uneventful; in 3 of the 6 patients a Nissen fundoplication was performed. At 1 and 2 years after implantation there was no evidence of device migration or erosion. At 3 years, only 2 patients reported the inability to belch or vomit. Conclusions This single-group trial confirmed that a magnetic device designed to augment the lower esophageal sphincter can be safely and effectively implanted using a standard laparoscopic approach. The device decreased esophageal acid exposure, improved reflux symptoms, and allowed cessation of proton-pump inhibitors in the majority of patients. CRITICAL APPRAISAL About 40% of patients with typical symptoms of gastroesophageal reflux disease (GERD) are refractory to pharmacologic therapy with proton-pump inhibitors (PPI) despite dose optimization (1). Laparoscopic Nissen fundoplication, the gold standard of surgical therapy, is largely underused and commonly reserved for patients with severe complications of GERD. This leaves a significant gap in the treatment continuum for GERD in the current clinical practice (2). 1) The LINX Reflux Management System was designed to provide a permanent solution to GERD by augmenting the lower esophageal sphincter barrier with a simple, reproducible, and reversible laparoscopic procedure that does not alter gastric anatomy. 2) The short-term, mid-term, and the 4-year results of a prospective feasibility clinical study evaluating 44 patients in whom the LINX was implanted at four study centers in the USA and Europe between February 2007 and October 2008 have been published (3-5). Ganz et al have now reproduced the results of this feasibility study, but in a larger cohort of patients. No significant differences were found between the studies in terms of safety and efficacy. The median DeMeester score before and after magnetic sphincter augmentation improved from 42.3 to 14.7 in the feasibility study and from 36.6 to 13.5 in the study by Ganz et al.; pH normalization rates were 80% and 58%, respectively. A reduction of at least 50% in the GERD-HRQL score compared to baseline off PPIs was achieved by 100% of patients in the feasibility study and 92% in the comparative study. Elimination of daily PPI use was reported in 80% and 87% of the patients, respectively. 3) At the University of Milan Medical School we have been involved both in the feasibility trial and in a registry of anti-reflux surgery, which is currently enrolling European patients to include treatment with either the LINX System or the Nissen fundoplication. The clinical outcomes over a six-year period in our clinical institution replicate the results from both the feasibility trial and the current trial by Ganz et al., showing that reduction in acid exposure, improvement in quality of life and elimination of PPI can be achieved in routine clinical practice (Bonavina L et al., J Am Coll Surg, in press). 4) The consistency of the results between these independent studies further supports magnetic sphincter augmentation as an important addition to the treatment options available for antireflux surgery. Further clinical experience and close patient follow-up are necessary to establish whether this innovation represents a true paradigm shift in the treatment of GERD. References 1. Katz PO, Gerson LB, Vela MF: Guidelines for the diagnosis and treatment of gastroesophageal reflux disease. Am J Gastroenterol 2013;108:308-328 2. Bonavina L, DeMeester TR, Ganz RA: LINX™ Reflux Management System: magnetic sphincter augmentation in the treatment of gastroesophageal reflux disease. Expert Rev Gastroenterol Hepatol 2012;6:667-74 3. Bonavina L, Saino GI, Bona D, Lipham J, Ganz RA, Dunn D, DeMeerster TR: Magnetic augmentation of the lower esophageal sphincter: results of a feasibility clinical trial. J Gastrointest Surg 2008; 12:2133-40 4. Bonavina L, DeMeester TR, Fockens P, Dunn D, Saino G, Bona D, Lipham J, Bemelman W, Ganz RA: Laparoscopic Sphincter Augmentation Device Eliminates Reflux Symptoms and Normalizes Esophageal Acid Exposure. One- and 2-year results of a feasibility trial. Ann Surg; 2010; 252:857-862 5. Lipham JC, DeMeester TR, Ganz RA, Bonavina L, Saino G, Dunn DH, Fockens P, Bemelman W: The LINX® reflux management system: confirmed safety and efficacy now at 4 years. Surg Endosc. 2012;26:2944-9 Critical appraisal by Luigi Bonavina, MD, Professor of Surgery, Chief of the Division of General Surgery, Department of Biomedical Sciences for Health, University of Milan Medical School, IRCCS Policlinico San Donato. Via Morandi 30, 20097 San Donato Milanese (Milano), Italy Tel. +39 02 52774621 / Fax +39 02 52774395 email luigi.bonavina@unimi.it