Calibration
advertisement

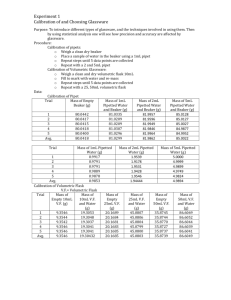
Chemistry 163 Harold CALIBRATION OF VOLUMETRIC GLASSWARE Chemistry 163 Laboratory South Puget Sound Community College Purpose The purpose of this exercise is to accurately calibrate your 25 mL volumetric pipet and buret so that in subsequent experiments, the volumes delivered will be known with greater accuracy than that provided by the glassware manufacturers markings. The calibrations will be accomplished by measuring the mass of water delivered by each of the pieces of glassware and converting those masses to volumes based on the density of water at the delivery temperature. Procedures: Volumetric Pipet If after the first trial with water the pipet retains droplets of water below the meniscus then clean the pipet with micro-clean cleaning solution. A large graduated cylinder will be provided in one of the hoods for this purpose. Rinse well with dH2O. The pipet should drain leaving no droplets in the pipet. If it does not, try soaking the pipet in microclean solution while you calibrate the buret. If it still does not drain properly speak to your instructor. When clean, fill the pipet with dH2O at room temperature, such that the meniscus is precisely at the calibration mark. Touch the tip of the pipet to the inside of a previously tared 50 mL Erlenmeyer flask and allow the pipet to drain into the flask. Allow ~15 seconds to elapse after the pipet appears to be empty. DO NOT shake the last bit out of the tip. ALWAYS use the same technique whenever using the pipet. Measure the mass of water delivered in triplicate. Calculate the volume of water at room temperature using interpolated density values determined from CRC published values of water density. The range among the results should not exceed 0.03 mL The average value is the volume value you should use whenever using your pipet. As always standard deviation and RSD should be included. 50 mL Buret Clean the buret with a buret brush and rinse well. Fill to very near the zero mark (at or below). Record an initial reading. Drain 10 mL into a tared 50 mL Erlenmeyer flask. Record the end level, measure and record the mass of water delivered. Repeat this process two more times for the 0 - 10 mL portion of the buret. Perform the same procedure in triplicate for 0 - 20 mL, 0 - 30 mL, 0 - 40 mL and 0 50 mL (DO NOT GO BELOW). Thus, each volume has been measured 3 times and an average variation from the true should graphed. Calculate the volume delivered for each of the trials at each range. Determine the variation from expected volume for each range. Create a correction graph. Vertical axis will be the mL correction. Negative values (below zero) show volumes actually delivered are less than buret values, positive show they are more. The horizontal axis will be the buret volumes 0, 10, 20, 30, 40, 50 mL. When you connect the points you have a calibration curve that can be used for any volume delivered between 0 and 50 mL. A sample graph is shown below. +.08 Buret Correction Table – add to measured values for true volume +.06 +.04 +.02 -.02 10 20 30 40 50 -.04 -.06 -.08 page 1 6/20/2007