LAB: The Metallurgy of Hairpins and Paperclips Background: Metals
advertisement

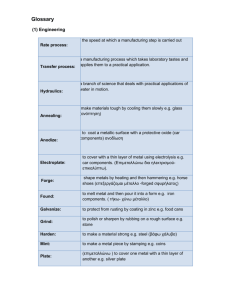
LAB: The Metallurgy of Hairpins and Paperclips Background: Metals are a crystalline substance. The crystal structure of metals affects their physical properties such as malleability and hardness. It is possible to alter these properties by altering the way that the crystal structure is formed. If you cool the metal down slowly, the crystal structure will be formed in a much more orderly fashion than if the metal is cooled down quickly, which causes a large number of imperfections (defects) in the crystal structure. It is the way that the crystal structure is formed that determines whether the metal is hard and brittle or soft and malleable. The composition of metals also affects its properties. Pure metals tend to be softer and more malleable than alloys. Alloys are solutions of different metals or, in the case of steel, metals and the element carbon. Alloys tend to be harder than pure metals and all metals used for structural purposes are actually alloys rather than pure metals in order to have the necessary hardness and strength. In this experiment we will study how the rate of cooling metals affects the hardness and malleability of the metal and attempt to use these properties to evaluate the composition of metal. We will also observe how the bending of a sample of metal will change its malleability. Objective: To study the affect of cooling rates on the properties of metal. Materials: Bunsen burner, 2 hairpins, 1 paper clip, crucible tongs, beaker, evaporating dish, Precautions: We will be working with red hot metals. Use tongs to handle metal that has been heated. Pay attention to people around you when you are heating metal. Put any heated pieces of metal in the evaporating dish or in the water beaker. Do not place heated metal pieces on the lab table. Goggles, aprons and appropriate footwear must be worn at all times in the lab area. Wash your hands thoroughly after completing the lab. Pre Lab Questions: 1. What safety material must we wear in this lab? 2. Why are metals that are used for building materials and tools alloys rather than pure? 3. Why is the rate that metals are cooled important when you are making metal structures and tools? 4. Explain how you are to safely pickup, hold and store the metals once heated. Procedure: 1. Obtain 2 hairpins and a paper clip. Compare the bending properties of the hairpin with that of the paper clip. Record your observations in your Lab Report. 2. Heat the bend of one of the hairpins in the flame until it is red hot. When the hairpin has been heated for at least 30 seconds, take it out of the flame and put it in the evaporating dish to cool slowly. 3. Heat the other hairpin like you did the first one. After it has been heated for 30 seconds, plunge it into a beaker filled with water. 4. Heat the paper clip like the hairpins. (Heat the bent part.) When it is red hot, plunge it in to the water to cool it quickly. 5. Attempt to straighten out both hairpins. Record your observations in your Lab Report, making note of the different properties of the hairpin that cooled quickly compared with the hairpin that was cooled slowly. 6. Now attempt to straighten the paper clip. Record your observations in your Lab Report. 7. Dispose of the hairpins in the landfill waste container. Return your paper clips that are still useable Post Lab Questions: Metallurgy of Hairpins and Paperclips Discuss and answer the following questions at the end of Your Lab Report. Each person must turn in their own Lab Report with these answers. Give complete sentence explanations. 1. Use your understanding of crystal formation to explain why cooling a metal slowly makes it soft and malleable and why cooling a metal quickly makes it brittle. 2. If you are making a sharp tool such as a knife would you want to cool the metal quickly or slowly in order to have it hold a sharp edge? Explain your answer based on the observations that you made for this experiment. 3. The hairpin that you started out with had much different properties than either hairpin had after the experiment. How do you think it was heated and cooled to give it the desirable properties for use as a hairpin? 4. Steel is an alloy of iron and varying amounts of carbon. The carbon atoms lie between the iron atoms in the submicroscopic structure of the steel. The hardness is directly related to the amount of carbon in the steel. High carbon steel is hard and brittle because the carbon atoms provide a greater number of defects in the crystal structure. Low carbon steel is softer and more malleable than high carbon steel because it has fewer defects, making the arrangement of the iron atoms more uniform. Based on your experiment, do you think that a paper clip is made of high or low carbon steel? 5. Is the hairpin made of low carbon or high carbon steel? Explain completely and use experimental evidence to support your conclusions. 6. Several years ago, a gasoline tanker truck caught fire on Interstate 5 in Lynnwood, WA. The fire was so hot and intense that it heated the steel bridge girders. Use what you have learned about heat treatment of steel to explain why the bridge girders needed to be replaced. http://www.komonews.com/news/archive/Tanker-Explodes-On-I-5-InLynnwood.html?tab=video&c=y Propose another experiment that you could do to further study the effects of cooling rate or bending on the properties of metal. Writing Assignment LAB REPORT: (50 pts Total Grade) HEADINGS for each section are important. The Lab Report must be done on separate lined-paper and Must Be Done Prior to Entering Lab. All that are in Bold must be ready before lab begins. Title of Experiment (2 pts) (Don’t Forget Your Name goes atop the Report, as well.) Experiment Background (5 pts) Summarize Metals and Alloys. Objective of Lab Experiment (3 pts) Do Not just copy the objective. What is this lab about? List of Materials Needed for Experiment (5 pts) Bulleted Items go great here. Safety Precautions to Note (5 pts) As always, put this in your words. Pre-Lab Questions (8 pts) (Write COMPLETE SENTENCE answers to the pre-lab questions on Lab Report. Include the subject of the question in your answer.) Experiment Procedures (7 pts) (Do Not simply copy. Put this in your words. What will I be doing for each step. Written in Detail because you will not take this paper into lab…only Lab Report, which you will be Reporting on goes with you to lab area. Experiment Observations Report (5 pts) This area only needs to be Labeled before beginning, so you can Record Observations while doing the experiment. Post Lab Questions (10 pts) Answered in Complete Sentences, after the experiment is complete. Include the subject of the question in your answer, or write the questions with your complete sentence answers.) Modified from Chemistry Lab - Metallurgy of Hairpins http://s221951463.onlinehome.us/school/chemistry/unit5/hairpin_lab.pdf