The Bronx High School of Science
Valerie Reidy, Principal
Department of Biology
J. Donahue, IA Asst. Principal
Name
Date
ACTIVITY 16: Investigating the properties of enzymes (Reminder: Bring goggles for this lab) (full lab write-up)
Introduction:
It is the presence of molecules called enzymes that give cells the ability to carry out the chemical reactions
necessary for life.
Enzymes have the following characteristics:
1) They are organic catalysts. A catalyst is a chemical agent (inorganic or organic) that changes the rate
of a reaction. In the case of enzymes, they usually speed up reactions.
2) They participate in the reaction, but remain unchanged in the end. (Common to all catalysts)
3) They are protein in chemical composition.
4) All formal names of enzymes end with ase.
Many cells produce hydrogen peroxide (H2O2) as a by-product of their metabolism. H202 is toxic. Cells that
produce H2O2 contain enzymes called catalases and peroxidases that mediate the reaction shown below:
catalase
2H2O2 ⎯⎯→ 2H2O + O2
NOTE: Catalase is the name of an enzyme. Remember that enzymes are also catalysts (definition above)
Question: How does the presence of these enzymes enable the cells to survive? (Answer on a separate sheet of
paper)
Materials:
8- 9 cm test tubes
test tube holder
2 test tube racks
3-15 cm test tubes
1 razor blade
splints
matches
20 cm piece of dialysis membrane
waxed paper for liver and potato
1 small funnel
hot plate
microscopes
plastic pipette
forceps
marking pencil
1 250 ml beaker
dropper bottle H2O
small bottle of dilute HC1
small bottle of dilute NaOH
thermometer
2- 500 beakers for water bath
filter paper
1 small Erlenmeyer flask
Opposite each sink: slides, coverslips, lens paper, potato juice, potato pieces, liver juice, liver pieces,
stock H202, methylene blue, MnO2 (manganese dioxide), NaCl (salt), ice cubes.
Procedure: Procedural Hints:
Divide the work to be done according to your teacher's instructions. Each student's laboratory report should contain
all problems investigated, and a section for Materials and Methods, Results, and Discussion.
Problems to be investigated: (include answers to these questions in your lab write-up)
Using the materials provided design experiments to answer the following questions. Prepare a hypothesis for
Activity 16: Revised September 2003
Page 1 of 4
each experiment in the space provided, and write in the discussion section whether or not your hypothesis was
valid.
1) How does the catalytic ability of inorganic and organic substances compare in the breakdown of
hydrogen peroxide?
a) In this investigation be sure to demonstrate that the material is only a catalyst, and not a reactant.
b) Use one or two potato and liver chunks.
c) How do extreme temperatures affect the ability of inorganic and organic catalysts to mediate their
reactions?
2) How does high, neutral, and low pH affect the catalytic action of enzymes?
3) How does the size of the enzyme molecule compare to the size of its substrate molecule?
a) In preparing your enzyme molecules, you must remove them from their cells. Should you use the
potato/liver chunks or potato/liver juice?
b) Be sure to demonstrate that these enzymes can work outside the living cell (in vitro).
Problem: How do extreme temperatures affect the ability of inorganic and organic catalysts to mediate their
reactions?
Materials and Methods: A sample of the organic catalyst(s) and the inorganic catalyst(s) were obtained. Each
sample was subjected to either an ice water bath or a boiling water bath. After this treatment, the samples were
returned to room temperature and tested to see whether they retained their catalytic properties. This was
accomplished by (describe experiments in your own words).
Results: It was found that when inorganic catalysts are heated (describe your results). When they are chilled,
(describe your results). When the organic catalysts from (potato or liver) are heated (describe your results). When
chilled (describe your results). Use the Data Table below. Results may also be presented in graph form.
Remember to label them Table or Figure 1 etc. and refer to the table or figure number in your results and
discussion sections.
Discussion: From these results, it is possible to make the following generalizations regarding the effect of
temperature on inorganic and organic catalysts. Discuss the meaning of your results here.
In some tests, the results were not as expected. This may be due to the following sources of error in the
experimental procedure (list possible sources of error).
Based on these findings, it is easy to see that high fever during illness may cause death because (put in
your own words).
Such temperature effects also explain the use of refrigeration to preserve food since (put in your own
words).
The following tables are for recording data. Give each table an appropriate title.
Scoring system:
0 = no activity
1 = light activity
2 = moderate activity
3 = heavy activity
Activity 16: Revised September 2003
Page 2 of 4
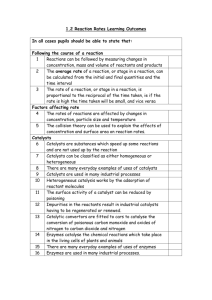
Table 1
Substance
1
Trial #
2
3
Conclusions
Potato
Liver
Manganese
dioxide (MnO2)
NaCl
Table 2
Substance
Ice water
Treatment
Room temp. Boiling water
Conclusions
Potato
Liver
Manganese
dioxide (MnO2)
Table 3
Low pH
(acid)
Prior treatment
Neutral pH
(distilled H20)
High pH
(base)
Substance
Conclusions
Potato
Liver
Table 4
Substance
Reaction occurs
Inside membrane Outside membrane
Conclusions
Potato juice
Liver juice
Activity 16: Revised September 2003
Page 3 of 4
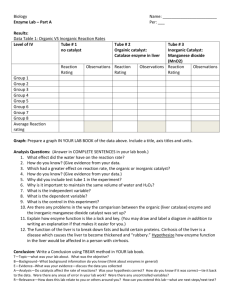
Table 5: Class Data
Group #
Effect of pH on enzyme activity
Low pH
Potato Liver
Potato/Liver/Manganese dioxide
(MnO2) values
Ice Water bath Room temp Boiling water
High pH
Neutral pH
Bath
L
M
L M P L M P
Potato Liver Potato Liver P
1
2
3
4
5
6
7
8
9
Average
Discussion Questions:
Observations drawn from your investigations should enable you to answer the questions below, in addition to
developing a coherent view of what enzymes are, what they do, and how they work. These are the ideas that should
be emphasized in your Discussion.
1) What generalization can be made regarding the kinds of cells which have catalase, based on your laboratory,
investigation?
2) Why do cells require comparatively small quantities of enzymes (as compared to substrate concentration) to
carry out most metabolic functions?
3) Based on your observations of the effect of extreme temperatures on inorganic and organic catalysts, construct
a graph that compares the effect of temperatures on the catalytic action of organic catalysts (enzymes) and
inorganic catalysts. Use one set of axes, labeled with the appropriate independent and dependent variables. Use
a solid line and a broken line to make the distinction between the two lines on the graph. Alternately, you can
use a different color for each line on the graph.
Application of Concept:
1) Why is an excessively high body temperature often life threatening?
2) Why are refrigeration and pickling successful methods of food preservation? HINT: Food spoilage is the result
of the metabolic activity of bacteria.
WRITE UP YOUR LABORATORY REPORT USING THE RESEARCH PAPER FORMAT
SAMPLE LABORATORY REPORT ON ONE INVESTIGATION
Activity 16: Revised September 2003
Page 4 of 4
 0
0