Buildings are often constructed on sites where volatile organic compounds (VOC) are
present in the ground. This publication provides guidance on the use of plastic
membranes to reduce vapour migration into buildings. It discusses vapour transport
mechanisms that occur in membranes, factors that influence this and the test methods
available for determining the rate of vapour permeation. It then explains how to use the
results in a risk assessment. Durability and resistance to damage when plastic
membranes are exposed to VOCs is a concern and the guide considers the potential for
degradation and which test methods are suitable to assess performance. This guide
should allow readers to make a robust evaluation of the contribution to membranes to
reducing indoor inhalation risk from VOCs in the ground and to specify the most
appropriate type of membrane for any given site.
C748
9 780860 177531
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Guidance on the use of plastic
membranes as VOC vapour barriers
Who we are
Established in 1960, CIRIA is a highly regarded, industry-responsive, not for profit research and information
association, which encompasses the construction and built environment industries.
CIRIA operates across a range of market sectors and disciplines, providing a platform for collaborative projects
and dissemination by enhancing industry performance, and sharing knowledge and innovation across the built
environment.
As an authoritative provider of good practice guidance, solutions and information, CIRIA operates as a knowledgebase for disseminating and delivering a comprehensive range of business improvement services and research
products for public and private sector organisations, as well as academia.
How to get involved
CIRIA manage or actively participate in several topic-specific learning and business networks and clubs:
zzCore
membership
zzCEEQUAL
Allows your employees to assist with the development of
and access to good practice guidance, formal networks,
facilitation, conferences, workshops and training.
zzAssociate
membership
zzLACL (Local
Allows your employees to access CIRIA’s services.
Members are able to access exclusive content via the
CIRIA website.
zzCIRIA
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
CIRIA Network
(European Marine Sand and Gravel Group)
CIRIA provides secretariat support to EMSAGG, including
management of the Group’s conferences, workshops and
website and producing its newsletter.
zzLANDFoRM
A member-based community where clients and professionals
meet, develop and share knowledge about specific topics
relevant to construction and the built environment.
zzProject
Authority Contaminated Land Network)
LACL helps local authorities address responsibilities
under Part IIA of the Environmental Protection Act 1990.
zzEMSAGG
Books Club
Members can buy most CIRIA publications at half price and
can attend a range of CIRIA conferences at reduced rates.
zzThe
CIRIA co-manages this environmental award scheme,
which promotes environmental quality in civil engineering
and infrastructure projects.
funding
(Local Authority Network on Drainage
and Flood Risk Management)
A platform for sharing knowledge and expertise in flood
risk management and sustainable drainage.
zzBRMF
Project funders influence the direction of the research
and gain early access to the results.
(Brownfield Risk Management Forum)
Promoting sustainable and good practice in brownfield
projects in the UK.
Where we are
Discover how your organisation can benefit from CIRIA’s authoritative and practical guidance – contact us by:
Post
Griffin Court, 15 Long Lane, London, EC1A 9PN, UK
Telephone
+44 (0)20 7549 3300
Fax
+44 (0)20 7549 3349
Email
enquiries@ciria.org
Websitewww.ciria.org
(for details of membership, networks, events, collaborative projects and to access CIRIA publications through
the bookshop)
CIRIA C748
London, 2014
Guidance on the use of
plastic membranes as
VOC vapour barriers
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
S Wilson EPG Ltd
S Abbot TCNF Ltd, and University of Leeds
H Mallett BuroHappold Engineering
Griffin Court, 15 Long Lane, London, EC1A 9PN
Tel: 020 7549 3300
Fax: 020 7549 3349
Email: enquiries@ciria.org
Website: www.ciria.org
Summary
Buildings are often constructed on sites where volatile organic compounds (VOC) are present in the
ground. This publication provides guidance on the use of plastic membranes to reduce vapour migration
into buildings. It discusses vapour transport mechanisms that occur in membranes, factors that influence
this and the test methods available for determining the rate of vapour permeation. It then explains how
to use the results in a risk assessment. Durability and resistance to damage when plastic membranes are
exposed to VOCs is a concern and the guide considers the potential for degradation and which tests
methods are suitable to assess performance. Physical damage during and after installation can also occur
and the guide identifies the key performance properties that should be specified to minimise the risk of
defects. This guide should allow readers to make a robust evaluation of the contribution to membranes
to reducing indoor inhalation risk from VOCs in the ground and to specify the most appropriate type of
membrane for any given site.
Guidance on the use of plastic membranes as VOC vapour barriers
Wilson, S, Abbot, S, Mallett, H
CIRIA
C748
RP960
© CIRIA 2014
ISBN: 978-0-86017-753-1
British Library Cataloguing in Publication Data
A catalogue record is available for this book from the British Library
Keywords
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Contaminated land, environmental management, ground engineering, materials sustainability
Reader interest
Classification
Hazardous ground gases, membrane and other
barriers, VOC vapours
Availability Open publication
Content
Advice/guidance, case study
Status
Committee-guided
UserConsultants, contractors, local authority
regulators, house builders, clients
Published by CIRIA, Griffin Court, 15 Long Lane, EC1A 9PN, UK
This publication is designed to provide accurate and authoritative information on the subject matter covered. It is sold and/or
distributed with the understanding that neither the authors nor the publisher is thereby engaged in rendering a specific legal or any
other professional service. While every effort has been made to ensure the accuracy and completeness of the publication, no warranty
or fitness is provided or implied, and the authors and publisher shall have neither liability nor responsibility to any person or entity
with respect to any loss or damage arising from its use.
All rights reserved. No part of this publication may be reproduced or transmitted in any form or by any means, including photocopying
and recording, without the written permission of the copyright holder, application for which should be addressed to the publisher. Such
written permission must also be obtained before any part of this publication is stored in a retrieval system of any nature.
If you would like to reproduce any of the figures, text or technical information from this or any other CIRIA publication for use
in other documents or publications, please contact the Publishing Department for more details on copyright terms and charges at:
publishing@ciria.org Tel: 020 7549 3300.
Front cover courtesy CETCO
ii
CIRIA, C748
Acknowledgements
This publication is the result of additional work carried out under CIRIA Research Project 960. It has
been written by Steve Wilson, EPG Ltd, Stephen Abbott, TCNF Ltd, and Hugh Mallett, BuroHappold
Engineering, under contract to CIRIA.
Authors
Steve Wilson MSc BEng CEng CSci MICE MCIWEM FGS
Steve is a chartered engineer and environmentalist with over 25 years’ experience of geotechnical and
environmental engineering. Steve is a well-respected specialist on the assessment of gassing sites and has
written several key technical papers on this subject. He has contributed to CIRIA, British Standards and
CIEH guidance on ground gas assessment.
Steven Abbot BSc PhD Fellow RSC
Professor Steven Abbott has a PhD in chemistry from Oxford/Harvard and was a post-doc researcher in
the Nobel prize winning laboratory of Prof J-M Lehn in Strasbourg. He was a senior research manager
at ICI for 14 years, then research and technical director of MacDermid Autotype for 15 years, during
which time he became a visiting professor at the University of Leeds. He started Steven Abbott TCNF
in 2009 where he works on technical software, coating/printing, nanosystems and formulations. His
technical software includes specialised models for diffusion processes through multi-layer barriers used
in food packaging, personal protective equipment, pharmaceuticals and biomembranes.
Hugh Mallett BSc MSc CGeol SiLC
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Hugh is a technical director at BuroHappold Engineering and has a wide variety of professional
experience gained over 35 years on the investigation, assessment and remediation of land affected by
contamination. Hugh’s experience has included the writing and presentation of technical information
for specialists, non-specialist professionals, government ministers and the public.
Project steering group
Following CIRIA’s usual practice, the research project was guided by a project steering group (PSG),
which comprised:
Peter Atchison
PA Geotechnical Ltd
Geoff Card (chair)
GB Card and Partners
Karen Day
Alderburgh
Seamus Lefroy Brooks
LBH Wembley Geotechnical and Environmental (also representing AGS)
Steve McConnell
ACS Lining
Brona McLaverty
Northern Group Systems (Environmental Health)
Richard Menage
Industrial Textiles & Plastics Ltd
Barry Mitcheson
SKM
Andy Shuttleworth
SEL Environmental Ltd
Harry Spillman
ACS Lining
Richard Stevens
Prestige Air Technology Ltd
Karen Thornton
NHBC
Niklas Ullfors
Visqueen
Peter Witherington
RSK
Guidance on the use of plastic membranes as VOC vapour barriers
iii
CIRIA Project manager
Joanne Kwan
Project funders
The work was funded by:
GB Card & Partners
Principal Building Products
SEL Environmental
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Visqueen
iv
CIRIA, C748
Contents
Summary . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ii
Acknowledgements . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . iii
Glossary . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . vii
Abbreviations and acronyms . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . viii
1Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1
1.1
1.2
1.3
2
Function of membranes in minimising VOC ingress to buildings . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3
2.1
2.2
2.3
2.4
2.5
Common misconceptions about membranes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3
Vapour transport mechanisms through membranes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4
2.2.1 Factors that affect permeation rate . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4
2.2.2 Available data on permeation rates . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 6
2.2.3 Influence of polarity on permeation rate . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 7
Permeation tests . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8
2.3.1 Permeation test methods . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 8
2.3.2 ISO 15105-2:2003 . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9
Modelling masss flux . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 10
Multi-layer VOC membranes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11
3
Factors that affect the performance of membranes as barriers to VOC migration . . . . . . . . . . . . . . . . . . 13
4
Durability of plastic membranes in relation to contaminant exposure . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14
4.1
4.2
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Background . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1
Purpose of this guide . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1
Risk-based approach . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 1
Potential for chemical degradation of membranes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 14
Chemical resistance tests . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 15
4.2.1 Test methods . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 15
5
Risk assessment . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 18
6
Geomembrane types, selection and considerations . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 21
6.1
6.2
6.3
6.4
6.5
7
Types of geomembrane . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 21
Selecting a geomembrane . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 21
What to consider . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 22
Key performance properties . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 24
CE marking and third party accreditation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 29
Installation and verification . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 30
References . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 32
Statutes . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 34
Further reading . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 36
Boxes
Box 2.1
Example of Hansen Solubility Parameters . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 11
Box 5.1
Calculation of permeation through a membrane with different floor slab constructions . . . . . 19
Box 6.1
Thermal stress and settlement stress analysis . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 26
Box 6.2
Estimating required puncture resistance . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 28
Guidance on the use of plastic membranes as VOC vapour barriers
v
Figures
Figure 2.1
Illustration of membrane permeation rate to allowable concentrations inside a building . . . . . 3
Figure 2.2
Diffusion of VOCs through the free volume of a membrane . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 4
Figure 2.3
Structure of molecules affects permeation . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5
Figure 2.4
Permeation of vapours through a membrane . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 5
Figure 2.5
Schematic of permeation test cell . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 9
Figure 4.1
Oxidation within 16 weeks immersion, sulphuric acid, % . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17
Figure 4.2Migration of vapour in reinforcement scrim of aluminium foil membranes. Delamination
within 25 days exposure to diesel vapours at 50ºC . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17
Figure 6.1
Flow chart for choosing an appropriate membrane . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 22
Figure 7.1
Unacceptable installation of a VOC membrane . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 31
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Tables
Table 2.1
Examples of permeation coefficients for VOCs through membranes (vapour phase) . . . . . . . . . 6
Table 2.2
Diffusive mass flux for VOCs through membranes (vapour phase) . . . . . . . . . . . . . . . . . . . . . . . . 7
Table 4.1
Effect of exposure to VOCs on membrane properties . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 15
Table 5.1
Reliance on membrane only . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 19
Table 5.2
Membrane and floor slab . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 19
Table 5.3
Permeation through HDPE membrane if source concentration is taken into account . . . . . . . 20
Table 6.1
Key performance properties . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 25
Table 6.2
Tensile strength required to resist thermally induced forces . . . . . . . . . . . . . . . . . . . . . . . . . . . . 26
Table 6.3
Tensile strength required to resist settlement induced forces . . . . . . . . . . . . . . . . . . . . . . . . . . 27
Table 7.1
Verification and integrity testing requirements for membranes required to resist VOC ingress . . . 30
vi
CIRIA, C748
Glossary
Challenge chemicalAny chemical used to assess the durability or permeation rate of a membrane when
exposed to that chemical.
The ability of a membrane to remain serviceable when exposed to a hydrocarbon
Hydrocarbon
resistantchallenge chemical, especially VOCs (ie to continue to prevent gas or VOC ingress).
This does not give any indication of the membranes ability to prevent or reduce the
migration of VOC vapours through it. Testing to determine resistance to damage
from VOCs is discussed in Section 4.2.
Mass fluxMass of gas that passes through a membrane per unit area per unit time (Welburn
et al, 2012). This is the property that is required for use in risk assessments.
Note this should be quoted in mg/m2/h. If the manufacturer’s literature does not
quote the permeation rate in mg/m2/h ask them to provide a conversion. It is not
possible to compare the performance of different membranes without using test
data obtained using the same test method and presented in the same units. The
mass flux is calculated from permeation rate tests and is the primary transport
mechanism for VOCs diffusion.
VOCsIn this publication, VOCs are considered to comprise organic compounds that are
volatile under ‘normal’ environmental/atmospheric condition. They may be found
in the ground in the solid, liquid and dissolved phase form as well as in gaseous
phase.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
VOC barrierA membrane is considered a barrier to a specific challenge VOC if its steady state
diffusive mass flux, when tested in accordance with ISO 15105-2:2003, is less than
that required to reduce the risk associated with VOC ingress to an acceptable level.
The required level of mass flux will be site specific and depend on a number of
factors such as the concentration gradient across the membrane and the ventilation
provided in the building. For this reason a generic minimum value of mass flux
cannot be specified for VOC barriers. In this guide the term VOC barrier is used,
rather than hydrocarbon barrier (which implies the membrane will be a barrier
to more than just VOCs). The supplier of a VOC barrier should provide all the
necessary test data referred to in this guide to allow risk assessors to determine its
suitability for a given site.
VOC permeation
The rate at which a VOC challenge chemical (liquid or vapour) will pass through
ratea unit thickness of the membrane per unit area per unit time (Welburn et al, 2012).
Units are mg/m2/h. Testing to determine the permeation rate is discussed in Section
2.3. The permeation rate will be dependent on the source concentration and
concentration gradient across the membrane (Scheirs, 2009).
VOC vapours
VOCs in gaseous phase when present in an air space.
Guidance on the use of plastic membranes as VOC vapour barriers
vii
Abbreviations and acronyms
BBA
British Board of Agrément
BTEX
Benzene, toluene, ethyl benzene, xylene
CSPEChlorosulfonate-polyethylene
DPM
Damp proof membrane
EIA
Ethylene Interpolymer Alloy
EPDMEthylene-Propylene-Diene-Monomer
EVOH
Ethylene vinyl alcohol
fPP
Flexible polypropylene
HDPE
High Density Polyethylene
HSP
Hansen Solubility Parameters
LDPE
Low density polyethylene
LLDPE
Linear low density polyethylene
PCETetrachloroethene
SSPR
Steady state permeation rate
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
TCETrichloroethene
USEPA
US Environmental Protection Agency
VLDPE
Very low density polyethylene
VOC
Volatile organic compounds
viii
CIRIA, C748
1Introduction
1.1BACKGROUND
Buildings are often constructed on sites where volatile organic compounds (VOCs) such as benzene,
toluene, chlorinated solvents, ethers etc are in the ground. The usual remedial solution is to provide a
gas membrane under or over the floor slab, of the same type as used to prevent methane and carbon
dioxide migration into buildings. The membranes are often made from polymeric materials and are
assumed to break the contaminant pathway by preventing vapour transmission. It is well known that
VOCs will migrate through polymeric materials (Welburn et al, 2012, ITRC, 2007, Massey, 2003 and
Scheirs, 2009). In some cases the permeation rate of contaminants through the membrane is so great
that it may not reduce the predicted level of indoor air contamination to acceptable levels, although this
will depend on the concentration of vapour in the ground and the nature of the membrane.
A wide variety of gas membranes are available with very different properties and performance
characteristics. Limited practical guidance currently exists, which enables a robust evaluation of suitable
membranes to enable their specification for use to act as barriers to VOC migration into buildings,
however some examples include Welburn et al (2012) and ITRC (2007).
This guide does not deal with the issues of chemical attack on plastic materials used as buried, in ground
barriers, water pipes, soil reinforcement or similar.
On the basis of current research/evidence (CIEH, 2008) it is very clear that standard damp proof
membrane (DPM) material is not suitable to act as a barrier to VOC migration into buildings in any
circumstances.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
This guide provides additional information on VOC permeation through membranes and complements
the information provided by Welburn et al (2012). In preparation of the guide due regard has been given
to the wealth of experience in studying chemical permeation through plastic materials that is available in
the food packaging, protective clothing and other industries.
1.2
PURPOSE OF THIS GUIDE
The purpose of this guide is to provide a summary of the functions of a vapour membrane and the
factors that may affect the performance of the membrane in a protection system for a building on land
contaminated by VOCs. It goes on to explain how to allow for the presence of the membrane in an
indoor inhalation risk assessment. Examples of how to do this are also explained in Welburn et al (2012).
The results of the risk assessment can help determine the necessary performance characteristics of a
membrane for use in a specification.
1.3
RISK-BASED APPROACH
The guide will explain how to take a risk-based approach to the specification of membranes to prevent
VOC migration into new buildings. The approach is consistent with CLR 11 (Environment Agency,
2004), and NHBC guidance (NHBC and Environment Agency, 2008a and b) and Welburn et al (2012).
Wilson (2008) showed that in the UK the Johnson & Ettinger (J&E) model is not suitable for the most
common types of new buildings that do not have basements. It was also suggested that a modular
approach allows the properties of a vapour resistant membrane to be incorporated into the risk
assessment, provided the barrier properties of the membrane can be adequately modelled.
Guidance on the use of plastic membranes as VOC vapour barriers
1
Membranes should not be the only consideration in VOC vapour risk assessment. Assessment
and mitigation of the pathway or link between the source of the VOCs and the building requires
consideration of more than just the membrane. So, the risk model should include the membrane (if
required), floor slab construction and any underfloor ventilation. Effects in the ground that retard
vapour migration should also be considered (eg biodegradation and other processes). With appropriate
modelling it is possible to arrive at a rational, defensible protection system that gives a reasonable balance
between risks and costs (Baker et al, 2009). The use of risk modelling should be fit for purpose, the input
and output parameters should be transparent and their source/use justifiable as part of the process. The
model should be easily understood and be able to be checked by regulators without access to proprietary
software. This will provide regulators and other stakeholders with the necessary confidence in the
data and demonstrate how the final design and specification of the vapour protection system has been
derived and is suitable for use in the context of the land contamination risks on a site-specific basis.
Assessment of the permanent gases such as methane and carbon dioxide is more concerned with acute
health effects or explosions and pressure driven flow. For this reason there is a much greater emphasis
placed on redundancy within the protection system and providing multiple levels of protection.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
The major issue with VOCs is normally long-term chronic health risks and there is less need for the
same levels of redundancy used for permanent gases, although a precautionary approach should still
be adopted. The level of redundancy in the protection system will depend on how effective in ground
processes (such as biodegradation) and the floor slab construction will be in retarding vapour migration
(Welburn et al, 2012). The potential for hazardous daughter/by-products from biodegradation processes
should also be considered, eg vinyl chloride from chlorinated solvent breakdown.
2
CIRIA, C748
2 Function of membranes in
minimising VOC ingress to
buildings
2.1
COMMON MISCONCEPTIONS ABOUT MEMBRANES
At present there are some common misconceptions about vapour transmission through membranes that
are marketed as being ‘hydrocarbon resistant’. This means that the protection provided could possibly be
much less than expected. So, first ask what is meant by hydrocarbon resistant? Does this refer to vapour
permeation rate through the membrane or to resistance to degradation when exposed to hydrocarbons
and particularly VOCs? The two properties are different (see Glossary).
In contrast to how well a membrane withstands chemical attack, permeation measures the rate at which
the challenge chemical moves through a membrane at molecular level. The polymer(s) forming the
membrane will permeate contaminants depending on how similar the molecular structure is to the
challenge contaminant.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
There is a common misconception that High Density Polyethylene (HDPE) completely prevents
VOC migration through it. VOCs and methane will permeate readily through homogeneous HDPE
membranes mainly because the two are of the same species, ie both HDPE and many VOCs are
hydrocarbons. However, in contrast to methane and carbon dioxide many VOCs can migrate through
it at a rate that is quite high in comparison to the allowable concentrations of those vapours inside
buildings. This is illustrated in Figure 2.1. Note the figure is illustrative only and the actual mass flux
through the membrane will depend on the concentration gradient across the membrane.
Figure 2.1
Illustration of membrane permeation rate to allowable concentrations inside a building
Guidance on the use of plastic membranes as VOC vapour barriers
3
So, membranes used to reduce VOC migration into buildings will require very low mass flux through
them in comparison with the internal allowable concentration of the VOC being considered. Reducing
the permeation rate for VOCs by increasing the thickness is not particularly effective or practical in most
cases. To halve the permeation rate, the thickness would need to be doubled.
Extensive experimental evidence (August and Tatzky, 1984) shows that benzene, toluene, ethyl benzene
and xylenes (BTEX) can permeate rapidly through HDPE if it is exposed to high concentrations of the
chemicals (reaching equilibrium concentrations in 10 to 20 days). In some cases they may not be that
effective as barriers to these molecules. In light of this it should not be assumed that a membrane alone
will block vapour migration of all VOC contaminants into a building and a site specific assessment of any
membrane should be completed (see Chapter 6).
2.2
VAPOUR TRANSPORT MECHANISMS THROUGH
MEMBRANES
2.2.1 Factors that affect permeation rate
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Vapour permeability will vary greatly according to membrane material and the nature of the vapour.
The correct specification for a membrane to act as a barrier to VOC ingress to buildings is critical.
However the quality of the membrane is only one factor. If any membrane is poorly installed the main
way in which vapours will pass through it will be via holes/punctures, poor joints and seals and/or other
defects, rather than through the material itself. If the quality/specification of the membrane and integrity
of the installation is adequate to minimise such defects the dominant mode of vapour transport will be via
molecular diffusion through the membrane. This is in contrast to transport mechanisms for methane and
carbon dioxide in soils, where the main driving force is often (but not always) the pressure head.
The key fact about the concentration gradient is that it depends both on the amount/concentration of
contaminant in the soil or groundwater and the solubility of the contaminant in the polymer surface
exposed to the contaminant. If it is insoluble (eg water exposed to the surface of polyethylene) then
despite a high concentration of water in the soil and despite the fact that water is a very small molecule,
little water will permeate because it is
insoluble. If it is soluble (eg benzene
exposed to the surface of polyethylene)
then the concentration gradient between
one side of the membrane and the other
can be large, so permeation will be
large, and as a result the membrane will
be ineffective as a vapour barrier.
Molecular diffusion is driven by a
concentration gradient with molecules
moving from zones of higher to lower
concentration. Any given molecule
may move through free spaces within
the membrane. The speed at which it
moves depends on the amount of free
volume in the membrane structure.
Typically membranes made of rubbers
have large amounts of free volume and
small molecules from the contaminant
source, eg benzene, will readily move to
fill this free space, ie an area of lower
concentration (Figure 2.2).
Figure 2.2Diffusion of VOCs through the free volume of a membrane
4
CIRIA, C748
The regular, symmetrical structure of HDPE means it is highly crystalline with about 20 per cent that
is amorphous. The free volume is low but benzene permeation will take place through the free volume
of the amorphous areas. If one of the hydrogen atoms is substituted with a methyl group (flexible
polypropylene) or chlorine (PVC) the crystallinity is reduced and the free volume increases. Smaller
molecules (eg benzene) will move faster than larger molecules (eg paraffin). In summary, permeation
depends on:
the concentration gradient
the nature of the membrane (free volume)
thickness of the membrane
the solubility of the contaminant in the polymer (which depends on molecular structure and
polarity, see Section 2.4)
the shape/size of the molecule and whether it is streamlined (Figure 2.3).
Bulky molecule,
eg o-xylene
Streamlined molecule,
eg p-xylene
Figure 2.3
Structure of molecules affects permeation (after Scheirs, 2009)
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
The permeation of vapours through a membrane is summarised in Figure 2.4.
Figure 2.4
Permeation of vapours through a membrane
The permeation rate will also vary depending on whether the membrane is saturated with the challenge
chemical (Wilson, 2008b). Over time VOCs can partition into the membrane until it is saturated (this will
depend on the concentration of the VOC in the ground). This will cause the membrane to swell and the
permeation rate will increase once the membrane is saturated.
Guidance on the use of plastic membranes as VOC vapour barriers
5
When a membrane is exposed to a VOC the molecules first of all diffuse into the membrane. Eventually
they will reach the other side of the membrane and breakthrough occurs. Before the breakthrough
point there is essentially no permeation as the chemical is migrating into the membrane material but
has not yet passed completely through it. The permeation rate is constant at zero. After breakthrough the
permeation builds up to its steady state (ie constant equilibrium) value as the membrane becomes saturated
with the VOC. The steady state value may be achieved within days in some cases (Chin et al, 2013).
2.2.2 Available data on permeation rates
There is limited, readily available, peer reviewed data on the permeation rates through membranes
used in building construction. One set of data for two particular VOCs (benzene and toluene) has been
published in a series of papers by Islam and Rowe (2001), Sangam and Rowe (2009), McWatters and
Rowe (2009), and McWatters and Rowe (2010). This is summarised in Table 2.1.
Table 2.1
Material
Chemical
Permeation coefficient (m2/s)
Nylon VBP15 coextruded
Benzene
0.02 × 10 -10
LLDPE:polyamide:LLDPE
Toluene
0.02 × 10 -10 to 0.03 × 10 -10
EVOH coextruded
Benzene
0.009 × 10 -12
LLDPE:EVOH:LLDPE
Toluene
0.011 × 10 -12 (1)
Benzene
0.1 × 10 -10
Toluene
0.2 × 10 -10
Benzene
0.2 × 10 -10
Toluene
0.4 × 10 -10
Benzene
0.2 × 10 -10
Toluene
0.8 × 10 -10
Benzene
2.29 × 10 -12
Toluene
7.74 × 10 -12
Benzene
2.07 × 10 -12 to 2.83 × 10 -12
Toluene
4.90 × 10 -12 to 7.22 × 10 -12
LLDPE
LLDPE
PVC
HDPE
HDPE
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Examples of permeation coefficients for VOCs through membranes (vapour phase)
Thickness of
Source
material (mm)
0.38
McWatters and Rowe (2010)
0.53
McWatters and Rowe (2010)
0.53
McWatters and Rowe (2010)
0.76
McWatters and Rowe (2009)
0.76
McWatters and Rowe (2009)
2.00
Sangam and Rowe (2001)
1.50
Islam and Rowe (2001)
Note
1The result quoted for LLDPE:EVOH:LLDPE is misleading. Other tests have not been able to detect anything passing through this type of
membrane. The data point is likely to be simply the amount of benzene absorbed by the first layer of LLDPE. It is better to consider this
value as <0.009 × 10 -12.
The values in the table are permeation coefficients, not permeation rates. If the diffusive mass flux is
not quoted for a material in mg/m2/hr for a standard concentration (eg pure liquid) it is difficult to make
any reasonable comparison of membranes in a simple risk assessment. However an estimate of the likely
diffusive mass flux of a chemical through a membrane can be estimated using the permeation rates in
Table 2.1 using the following equation (Fick’s Law of Diffusion):
Diffusive mass flux of chemical [ML-2T-1] = Pg (dCf /dz)
Where:
Pg =Permeation coefficient [L2T-1] that allows for partitioning and diffusion processes at each
side and within the membrane, from Table 2.1. Values for other VOCs have not been found
in a literature search and would have to be determined from specific testing.
6
dCf=
Difference in concentration of chemical from one side of membrane to other (Figure 2.4).
dz =
thickness of membrane (Figure 2.4).
CIRIA, C748
Care should be taken when using the permeation coefficients as they only relate to the thickness of
material and concentration of the particular chemical used in the tests.
Data on diffusive mass flux through membranes may be presented in other forms from that in Table
1.1 and commonly, results from laboratory tests quote the values in terms of a mass per unit area per
unit time (eg mg/m2/h). Values for the permeation of xylene and toluene through different thicknesses
of HDPE membrane are provided in Table 2.2. The table also shows the variation that occurs due to
changes in the concentration of the challenge chemical on the dirty side of the membrane.
Table 2.2
Material
Diffusive mass flux for VOCs through membranes (vapour phase)
Diffusive mass
flux (mg/m2/h)
Thickness of
material (mm)
916
0.8
500
2.6
750
0.75
Xylene (10 mg/l)
0.71
0.76
Xylene (50 mg/l)
3.21
0.76
Xylene (100 mg/l)
13.29
0.76
Xylene (100 mg/l)
1.79
1.52
Xylene (100 mg/l)
0.58
2.54
Toluene (10 mg/l)
0.79
0.76
Toluene (50 mg/l)
4.42
0.76
Toluene (100 mg/l)
14.58
0.76
Toluene (100 mg/l)
2.92
1.52
Toluene (100 mg/l)
1.00
2.54
Chemical
HDPE
HDPE
Xylene
LDPE
HDPE
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
HDPE
Source
Schiers (2009)
Thomas and Koerner (1996)
Thomas and Koerner (1996)
Note
The tests are assumed to have used m,p and o xylenes as the isomer is not stated in the source references.
In practice, brownfield sites are contaminated by a wide spectrum of pollutants which vary from site to
site. It is therefore important that the barrier membrane can demonstrate adequate barrier performance
against each pollutant that is of concern on a particular site (a chemical may be present, but not at levels
that constitute a risk to the occupiers).
2.2.3 Influence of polarity on permeation rate
The molecular structure and polarity of the membrane is one important factor in the rate of permeation
of the challenge chemical. Polarity is an indication of a balanced or unbalanced molecular electric field
and is determined by polar and hydrogen bonding.
A standard chemical rule is that ‘like is soluble in like’. So if the membrane material and contaminant
are either both polar or both non-polar the solubility will be high and the permeation rate will be high.
If the membrane material is polar and the contaminant is non-polar (or vice versa) the contaminant will
not be soluble and permeation will be low. Water is a common example of a polar material and grease, a
non-polar material. Grease does not readily dissolve in water (although it can form an emulsion).
Guidance on the use of plastic membranes as VOC vapour barriers
7
To be an effective barrier, a membrane needs to demonstrate a sufficiently low permeability for each
contaminant of concern on a particular site, be it a VOC or other chemical. As a membrane’s barrier
performance is defined by its composition it may be suitable as a barrier for one type of challenge
chemical but not against other types. For example, polyamide is polar and so provides a good barrier
against non-polar chemicals, especially non-polar VOCs such as BTEX. HDPE is non-polar and does not
provide a good barrier against non-polar chemicals, in particular some VOCs at high concentrations.
2.3
PERMEATION TESTS
As a result of the difficulty in finding data for this research project and the multitude of units that are
used, it is recommended that manufacturers of membranes designed to be used to limit VOC vapours
into buildings should provide data on the permeation of vapour from the following range of chemicals as
a minimum. The results of tests to determine permeation rates of contaminants for a specific membrane
are essential to allow effective risk assessment. Membrane manufacturer’s literature should be required
to verify the permeation rate relevant to the contaminant of concern. The tests should be undertaken
at a range of contaminant concentrations and on samples including a typical joint (welded or taped). A
commercial laboratory has indicated that it is feasible to test the jointed materials.
The testing should provide permeation rates for the following chemicals:
benzene
toluene
ethyl benzene
m, p and o xylenes
hexane
vinyl chloride
tetrachloroethene (PCE)
trichloroethene (TCE)
naphthalene (solid at room temperature but will still produce vapour phase contamination due
to partitioning). Test methods have been developed to test naphthalene vapour migration across
membranes although they are not standard tests (Nerin et al, 2007).
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Recommended test methods for determining the permeation rate are discussed in the following subsections.
Specific sites may require testing of the membrane for other chemicals depending on the nature of the
contamination in the ground. There may be a number of VOCs that will each require separate risk
assessments. It is also important to be mindful of volatile hazardous daughter/by-products resulting
from natural attenuation/biodegradation of the main contaminants. Synergistic effects of different
contaminants also need to be considered.
Both in the UK and elsewhere, regulators and designers are increasingly being supplied with evidence
from suppliers to allow them to carry out risk assessment modelling with regard to migration of VOC
contaminants of concern through a membrane. This is providing greater reassurance to all that the
membrane is suitable for use as a barrier to VOC migration into buildings.
2.3.1 Permeation test methods
Quantifying the effectiveness of a membrane is a key component in deciding upon a membranes
suitability for a given application or type of contamination. To do this permeation data is required that
can be incorporated into risk assessments to demonstrate the effect of proposed mitigation measures.
Originally, gravimetric test methods were developed to assess the rate of permeation. Such tests are
generally rudimentary and involve sealing a membrane over a jar of the challenge chemical and
8
CIRIA, C748
measuring the weight loss over time. Gravimetric permeation test methods include ASTM E96/E96M-05,
ASTM D5886-95 and BS EN ISO 6179:2010.
The main limitation on these tests is a lack of knowledge about the vapour concentration of the chemical
on either side of the membrane. However they usually have a very high concentration of vapour on the
chemical side of the test so are worst case for most, if not all contaminated sites. They can be carried
out quickly and easily to give some indication of likely permeation rates. The results are useful for
comparing relative transmission rates of a chemical through different membranes.
There are some limitations to the BS EN ISO 6179 test. It is only applicable to materials in sheet form
and to coated fabrics having a thickness between 0.2 mm and 3.0 mm. It is restricted to measuring
transmission rates (or mass flux) greater than 0.1 g/m2/h (100 mg/m2/h), which extrapolates to a
minimum mass flux of 876 g/m2/yr. This is too high to be useful for many VOC vapour risk assessments.
2.3.2 ISO 15105-2:2003
Over time modern permeation test methods have been developed that are highly accurate and
measure steady state permeation rates using mass spectrometry. One such test is described by ISO
15105-2:2003. This specifies a method for determining the gas transmission rate of plastic materials
in the form of film, sheeting, laminate, co-extruded material or flexible plastic-coated material. The
resistance of a membrane to permeation by liquid or gaseous contaminant chemicals is defined by the
breakthrough time and steady state permeation rate (SSPR) of the chemical through the barrier at a
given concentration of the chemical.
From a regulatory position transparency, clarity and accuracy in the testing are key. It is also essential
that there is consistency in approach to ensure comparability and there should be confidence from
all parties that the outcome of the process is accurate and protective. It is recommended that the ISO
15105-2 test method is adopted as the industry standard for providing data on permeation rates through
membranes for use in contaminated land risk assessment.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
The permeation testing introduces a test gas or vapour to one side of the membrane. On the other
side a ‘sweep’ gas collects gas permeating through the membrane and takes it to a detector. The sweep
gas is normally an inert gas such as nitrogen or helium. Detection can be undertaken using a number
of instruments including mass spectrometers. The pressure on each side of the membrane is equal. A
schematic of the permeation test is shown in Figure 2.5.
Figure 2.5
Schematic of permeation test cell
The test system records the volume of any permeate as a function of time after its initial contact with
the membrane. The gas transmission rate is reported as mol/(m2s Pa) by laboratories and the results
will require conversion to units that are easily used in risk assessment at standard conditions under
a pressure of one atmosphere (mg/m2/h). This should be done by the manufacturer to provide values
for their literature. Minimum detection rates will depend on the test apparatus set up and detection
equipment used. However they are typically much lower than those achievable using gravimetric
methods. It is also useful to determine the diffusion or permeation coefficient for a membrane.
Guidance on the use of plastic membranes as VOC vapour barriers
9
Even so, it may be difficult to measure the permeation of materials with extremely good barrier
performance. The sample may take an extremely long time to reach equilibrium saturation and steady
state permeation, resulting in a large cost. Tests that run for a long time also increase the risk of
permeation to or from the external environment. Even if steady state permeation is achieved, the flux
may be so low that detectors may not be able to measure it accurately. This is a commercial decision for
manufacturers. The laboratory test certificate should state the outcome of the tests only and it is the
responsibility of a risk assessor or protection designer to determine if a given set of results are acceptable
on a particular site.
Further information on the available test methods for measuring permeation and diffusion in polymers
is provided by Duncan et al (2005).
The test report should include the following information that is required by risk assessors:
the mass flux in mg/m2/h
the thickness of the test sample
the concentration of the challenge chemical used to complete the test (on the dirty side)
results of the evaluation process undertaken by the remediation designers to assess the impact of
providing different membranes in the floor slab construction.
2.4
MODELLING MASS FLUX
Physical testing of permeation rates through a particular membrane is usually only available for a limited
number of challenge chemicals (see previous section). This may be acceptable because the risk assessment
process for land contamination usually focuses on a few limited marker compounds. For example, using
the TPH CWG Criteria Working Group carbon bands the properties of a marker chemical are used to
define the risk assessment properties for each band (eg C6 to C8).
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
If the mass flux for a wider range of chemicals is required then the values can be estimated using
modelling, or specific testing can be undertaken (although costs and timescales may not fit in with
construction programmes). Modelling is also useful for comparing the difference in performance
between materials.
One convenient approach to modelling a large variety of chemicals interacting with a barrier is that
of Hansen Solubility Parameters (HSP) (Hansen, 1999 and 2009). This has been used to model the
permeation of chemicals through a membrane developed to provide protection to buildings against
VOC vapours. Each polymer and chemical has a set of the three parameters that make up HSP: δD, δP,
δH, which respectively capture the dispersion, polar and hydrogen-bonding chemistry. As ‘like dissolves
like’ so chemicals with HSP similar to those of a polymer will be ‘like’ and therefore soluble. A simple
calculation of ‘HSP distance’ (based on the sum of the square of the differences between the three
parameters) makes it possible to estimate solubility. For membranes, even more important than solubility
are partition coefficients. These are readily estimated from the relative HSP distances. An example of
using HSP to estimate the likelihood of solubility is provided in Box 2.1.
10
CIRIA, C748
Box 2.1
Example of Hansen Solubility Parameters
The three Hansen Solubility Parameters (HSPs) are co-ordinates for a point in three dimensions. Molecules that are close
together in this three dimensional space are more likely to dissolve into each other. The procedure to determine if the
parameters of the solvent and the membrane polymer are within range to be soluble is as follows (Abbott, 2008):
1
A value called interaction radius (R0) is determined for the membrane being dissolved using experimental test data
on a limited set of solvents. This value determines the radius of the sphere in Hansen space and the x, y and z
co-ordinates of its centre are the three Hansen parameters. Once this and the other HSPs are known for a set of
chemicals the solubility of the membrane in other chemicals can be determined.
2
Calculate the interaction radius (Ra) between Hansen parameters in Hansen space using the following formula:
(Ra)2 = 4(δD2 –δD1)2 + (δP2 – δP1)2 + (δH2 – δH1)2
δD = energy density from dispersion forces between molecules
δP = energy density from dipolar intermolecular force between molecules
δH = energy density from hydrogen bonds between molecules
D2, D1 etc refer to the parameters for the solvent and membrane respectively.
For benzene
δD = 18.4 MPa0.5, δP = 0.0 MPa0.5, δH = 2.0 MPa0.5 from (Hansen, 1999)
For a certain PVC polymer (PVC 4 HR)
δD = 24.4 MPa0.5, δP = 4.9 MPa0.5, δH = 9.9 MPa0.5 and R0 = 22.7 MPa0.5 (Hansen, 2009).
So for PVC/benzene, Ra = 15.18
3
Calculate the relative energy difference, RED = Ra/Ro = 15.18/22.7 = 0.67.
4
Compare the values to the following list:
RED <1 means the molecules are alike and will dissolve/absorb.
RED = 1 means the molecules will partially dissolve/absorb.
RED >1 means the molecules will not dissolve/absorb.
In the case of PVC and benzene RED is <1 and the initial indication is that benzene will dissolve or adsorb into the PVC
and will pass through easily.
Similar calculations for LDPE (δD = 16.5 MPa0.5, δP = 4.5 MPa0.5, δH = 0.5 MPa0.5 and R0 = 6.0 MPa0.5) gives an RED of
1.0, which again indicates that benzene may pass through it but not as easily as PVC.
Other modelling approaches that are being developed to assess diffusion of chemicals through materials are described
by Duncan et al (2005). These include methods that consider the concentration gradient across a membrane.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
2.5
MULTI-LAYER VOC MEMBRANES
In general a membrane made from a single material cannot resist all chemicals (hence packing films
in the food industry are multi-layer membranes). An HDPE membrane is not necessarily a suitable
defence against BTEX permeation and it cannot be assumed to break the migration pathway (it will
depend heavily on the vapour concentration of the contaminant in the ground below the floor slab). For
each of the common membrane materials such as butyl, chlorosulfonate-polyethylene (CSPE), ethylenepropylene-diene-monomer (EPDM), flexible polypropylene (fPP) and ethylene interpolymer alloy (EIA)
there are similar circumstances where a common pollutant chemical (VOC) can easily permeate or
compromise the physical properties of the membrane (strength, permeation rates etc) if it is present at a
sufficiently high concentration.
Examples of multi-layer membranes used to prevent ground gas or vapour ingress into buildings include:
LLDPE, LDPE or HDPE with an aluminium foil core
LDPE:EVOH:LDPE
HDPE:EVOH:HDPE
HDPE:polyamide:EVOH:polyamide:HDPE (or variations on this layering)
HDPE:VLDPE (although these materials are very similar in terms of VOC permeation)
asphalt latex:HDPE.
Guidance on the use of plastic membranes as VOC vapour barriers
11
A simple example of how multi-layer barriers work in food packaging is given as follows. The same
principles can be applied to membranes that are required to prevent VOC vapour ingress into buildings.
LDPE is an excellent moisture barrier and a poor oxygen barrier, while EVOH co-polymer is an excellent
oxygen barrier but a poor moisture barrier. So a typical packaging film will be an LDPE:EVOH:LDPE
sandwich. The LDPE provides not only the moisture barrier but also protection for the rather delicate
EVOH. The EVOH provides the oxygen barrier properties only as long as it remains protected from
moisture and handling damage by the LDPE. The two layers of LDPE provide insurance against
any deterioration of general mechanical properties from one side. For example, if the inner LDPE is
weakened by a soluble contaminant, that contaminant will tend to be blocked by the EVOH and so will
not have a chance to weaken the other LDPE layer.
An HDPE:EVOH:HDPE membrane will provide a barrier against migration of benzene. This is because
benzene has exceptionally low solubility in EVOH. The same barrier will also prevent methanol
migration because although the EVOH readily transmits methanol (the HSP distance is low), the
exceptionally low solubility of methanol in HDPE provides a barrier. The barrier properties are less
effective for intermediate pollutants, which are partially soluble in both HDPE and EVOH. For example,
for methyl t-butyl ether the reduction in permeation rates is only 5 orders of magnitude, because the
ether is less insoluble in the EVOH.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
For extra robustness further polymer layers may be added with a different solubility profile from HDPE
and EVOH. This makes the chances of adequate solubility in each layer becoming smaller and the
barrier properties remain acceptable over a wide range of pollutants.
12
CIRIA, C748
3 Factors that affect the
performance of membranes as
barriers to VOC migration
Membranes in buildings could potentially be exposed to a wide variety of VOC compounds in
the ground. The exact cocktail of chemicals will vary from site to site. The main requirements for
membranes used as barriers to VOC permeation are:
The permeation rate of the challenge chemicals through the membrane should be sufficiently low. This
should take account of the vapour concentration below the membrane if data is available to do this.
The mechanical properties (strength, tear resistance etc) of the membrane should not be adversely
affected by the challenge chemicals such that its performance as a barrier is compromised.
The membrane should be able to withstand the physical stresses placed on it both in the permanent
state, but more importantly during construction.
The membrane should not suffer from biological or other forms of degradation (oxidisation, stress
cracking etc).
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Because of the very low permeation rates that are usually required to minimise the risks associated with
VOC vapours it is extremely important that the membrane is installed correctly and is not damaged
afterwards. So, where VOC membranes are required, integrity testing should be carried out unless
the designer specifies in the verification plan that it is not required and justifies this decision. An
example where integrity testing may not be required would be where the level of risk is very low and the
membrane is being installed as a precaution as part of a multi-level protection system. This is discussed
in more detail in Chapter 6.
Guidance on the use of plastic membranes as VOC vapour barriers
13
4 Durability of plastic membranes
in relation to contaminant
exposure
4.1
POTENTIAL FOR CHEMICAL DEGRADATION OF
MEMBRANES
Membranes used in gas or vapour protection systems may be subject to various forces (tearing, shearing,
tensile forces) or can potentially be exposed to aggressive chemicals (eg benzene, toluene or other
vapours). In extreme cases this can result in the loss of integrity or serviceability and undermine the
effectiveness of the protection system.
The potential for chemical attack on a membrane will depend on:
the presence of a pathway for the challenge chemical to reach the membrane
the availability of the challenge chemical, ie concentration and the rate at which it migrates towards
the membrane
degree of contact between the challenge chemical and the membrane material
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
the sensitivity of the membrane to the challenge chemical, in particular, the polarity of the
contaminant and the susceptibility to partitioning of the membrane material by the challenge
chemical (ie how easily it is absorbed).
In building applications the membrane should not be exposed directly to the source of the
contamination and should only be exposed to vapours that have migrated through the ground. The
presence of water increases partitioning of vapours into polymers, so in theory the concentration at
the membrane interface could be higher than might be expected. This could be a concern where the
membrane is in direct contact with contaminated soils, but is less of a concern where the membrane is
separated from the source of contamination. Membranes in building construction will usually be located
either above the slab or on top of the sub-base both of which will have relatively low moisture contents. So
the vapour concentration at the membrane will normally be much lower than the source concentration in
the ground.
Different membrane materials may be adversely affected by different contaminants. For example,
HDPE readily loses greater than 50 per cent of its structural strength when exposed to 100 per cent
concentrations of challenge contaminants such as BTEX. HDPE will absorb halogenated and/or
aromatic hydrocarbons (including many fuels and oils) at greater than 50 per cent concentration (Rowad
International, 2013). For one reinforced membrane the chemical compatibility chart indicates minor to
moderate effects for benzene at one per cent and 25 per cent concentration but severe effects at 100 per
cent concentration (Layfield, 2013) although the membrane material is not stated. Another membrane
showed only minor effects when exposed to one per cent toluene but minor to moderate effects at 25 per
cent concentration.
In a building application HDPE may not be exposed to high concentrations of challenge chemicals and
may not be subject to any great tensile loads (eg when laid on top of a slab). In this instance the risk of
damage due to exposure to the challenge chemicals will be a lot lower than indicated by exposure tests
at 100 per cent concentration. The potential for adverse effects should be assessed on a site specific basis,
preferably using exposure data at concentrations that reflect site conditions.
14
CIRIA, C748
The main properties that exposure to chemicals can affect are listed in Table 4.1.
Table 4.1
Effect of exposure to VOCs on membrane properties
Property
Possible effects
Thickness
The primary effect of exposure of polymeric membranes to VOCs is swelling. The VOCs are absorbed
by the membrane, which results in weight gain. Swelling can cause waves to form across a membrane,
which can exert peel forces on welds. This is not likely to be a significant issue in small areas of
membrane that are restrained by the floor construction. Potentially the most significant effect is that
swelling can increase the permeation of chemicals through the membrane (Duncan et al, 2005).
Weight
The absorption of VOCs causes an increase in weight of the membrane. In most applications the
increase in weight is not a significant issue in itself. In chemical immersion tests a mass gain of 10 per
cent or more is taken to indicate that the membrane will not be resistant to exposure to the chemical
solution (Scheirs, 2009).
The absorption of VOCs causes softening of the material with a resultant loss of tensile strength. Where
the membrane is fully supported by a suspended floor slab this is not likely to be a significant issue. Where
a membrane is below a floor slab and is required to resist tensile forces caused by settlement it needs to
be considered. In chemical immersion tests if the elongation at break decreases by less than 20 per cent
Tensile strength
(in relative terms) this indicates that there is no significant effect on the membrane. The effects of swelling
and reduction of strength and stiffness caused by VOCs are not permanent and do not generally cause
permanent degradation of membranes. The effects also only occur up to a certain point and after the
VOCs volatilise out of the membrane the original properties are recovered (Scheirs, 2009).
Stiffness
As for tensile strength softening will reduce the stiffness of the membrane.
Antioxidant or
stabiliser levels
Absorption of VOCs can cause a loss of antioxidants or stabilisers added to the membrane material. This
can result in oxidation or stress cracking of the membrane occurring in service.
4.2
CHEMICAL RESISTANCE TESTS
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Chemical resistance is different from chemical permeation and different tests are required to those
discussed in Section 2.3. Membrane providers may claim that their membranes ‘meet chemical resistance
standards’ when in fact the membrane has a high level of permeation. The fact that a membrane
complies with a standard with regard to chemical resistance does not necessarily mean that it is fit for
purpose in a particular situation as it may have a high permeation rate to the contaminant and allow
migration through the barrier. It is important to make this distinction between chemical resistance and
barrier performance since chemical resistance tests do not provide any permeation data. A membrane’s
chemical resistance (like its barrier performance) is defined by its composition.
4.2.1 Test methods
ASTM D5322-98 (2009) is a widely recognised test method for chemical resistance and it is incorporated
within the Method 9090A (EPA, 1992) and ASTM D5747/D5747M-08 (2013)e1. The European test
method procedures described in BS EN 14414:2004 and BS EN 14415:2004 are virtually identical to
ASTM D5322-98, the main difference being that the European Standards define a fixed test period of
56 days, while the ASTM Standard allows the manufacturer to determine the duration of the test period.
The test procedure involves immersing a sample of membrane in the challenge chemical at an elevated
temperature of 50°C for the test period after which it is inspected. Thickness, weight, tensile strength
and elongation are then compared with a control sample and providing variations are within 25 per cent
of the control sample test results, the membrane is considered chemical resistant.
An important limitation of these standards is the choice of challenge chemicals that are actually tested.
BS EN 14414 tests for the following:
Method A – acid: 10 per cent solution H2SO4
Method B – alkali: saturated Ca(OH)2
Guidance on the use of plastic membranes as VOC vapour barriers
15
Method C – diesel, paraffin and lubricating oil mixture
Method D – synthetic leachate comprising a mixture of 14 acids, chlorides, sulphates and a
phosphate.
BS EN 14415 tests for the following:
Method A – hot de-ionised water
Method B – alkalis test
Method C – organic solvents (methanol, isopropanol, and glycol).
BS EN 14414 test Method C is the relevant immersion test for hydrocarbons. This single immersion test
comprises a challenge solution cocktail of 35 per cent diesel fuel, 35 per cent paraffin and 30 per cent
lubricating oil. Those familiar with basic principles of chemical solvency will realise that this test for
‘hydrocarbons’ is highly optimistic in terms of VOCs. The test materials are large molecules that make
naturally poor solvents. Although VOCs such as BTEX are ‘hydrocarbons’ they are generally much
more aggressive solvents than this test mix. So extended testing using specific chemical compounds is
preferred. In light of this it is recommended that any membrane exposed to VOCs has additional tests
undertaken to confirm its durability when exposed to the following basic suite:
benzene
toluene
ethyl benzene
m, p xylene
xylene
hexane
vinyl chloride
tetrachloroethene
trichloroethene
naphthalene.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Manufacturers should provide this test information and designers and regulators should ask for it before
agreeing to the use of any membrane that is exposed to significant levels of the chemicals (eg in direct
contact with soil or groundwater contaminated with challenge chemicals).
Tests using other chemicals may also be required depending on the specific contaminants and the
exposure conditions of a membrane on a site. It would also be beneficial if manufacturers carried out
durability tests using low concentration aqueous solutions of chemicals to obtain a better understanding
of the effects of exposure to low concentrations of chemicals (which is likely to be the dominant case
where membranes are used in building construction).
HDPE passes the EN 14414 Method C chemical resistance tests, which is an accelerated life test
comprising immersion at 50°C for 56 days in the mixture of diesel, paraffin and lubricating oil described
previously. However if the membrane is exposed to contaminants more aggressive than diesel a
reduction in structural strength could occur (eg if exposed to BTEX in high concentrations). This may
be an issue, for example, where a membrane is laid below a slab and there is movement in the soil. This
will be less of a problem where membranes are placed over the top of floor slabs.
Aluminium laminate membranes are commonly used to prevent gas and vapour ingress to buildings.
There is no disputing the robustness of an intact and continuous aluminium barrier against permeation
of almost any common organic pollutant – aluminium simply does not allow any permeation. However,
the relatively thin layer is susceptible to corrosion as a result of oxidation if it is exposed due to damage
to the protective layers on either side. This can occur in either acid (Figure 4.1) or in alkaline conditions
and reduces its barrier properties to a low level. Also the aluminium layer itself is so delicate that it has to
be protected by reinforcement scrims and can tear if there is any extension, for example, due to ground
16
CIRIA, C748
movements or shear forces during installation. So, a reinforcement scrim is necessary to reduce the risk
of tearing.
Figure 4.1Oxidation within 16 weeks immersion,
sulphuric acid, % (EN14414 Method A)
Figure 4.2Migration of vapour in reinforcement scrim of
aluminium foil membranes. Delamination within
25 days exposure to diesel vapours at 50ºC
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Permeation tests on aluminium foil laminates demonstrated that migration can also occur along the
outer membrane and this cannot be sealed at joints. This can be a significant pathway where there
are multiple joints (eg in housing where the membrane is joined to a damp-proof course around the
perimeter of a room). The carrier polymers, particularly the lower density polymers for the laminate (eg
LDPE) can themselves be more prone to be compromised by BTEX at sufficiently high concentrations
allowing the aluminium to be torn or punctured. The thinner the outer membrane the greater risk there
is of this happening. The membranes can also delaminate when the chemical condenses between the
polymer layer and the foil layer, reducing the structural integrity (Figure 4.2 shows delamination and
condensation depicted by the bubbling). Therefore such membranes are less suitable for sites with highly
elevated concentrations of VOC vapours or where significant movement of the membrane is expected,
for example, due to settlement.
Guidance on the use of plastic membranes as VOC vapour barriers
17
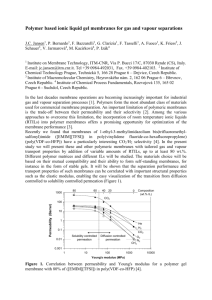
5 Risk assessment
Risk assessment for VOC vapour inhalation in buildings should be undertaken to determine if there
is a potentially unacceptable risk to the occupants of a building. The risk assessment should be site
specific using realistic parameters for the building and ground (for example, do not assume there is
a crack around the perimeter of a floor slab if the proposed construction means that one will not be
present). The J&E model assumes a perimeter crack but it can be modified to remove it. If the level of
risk is deemed to be unacceptable then the effect of introducing ventilation below the floor slab and
a membrane can be modelled in the risk assessment to determine the appropriate level of protection.
The properties of any membrane can be included in the model to help determine the necessary level of
performance and allow the specification of a suitable membrane. A method of modelling vapour ingress
that can be adapted to allow for the properties of the membrane is described by Wilson (2008), and there
is also guidance provided by Baker et al (2009) and Welburn et al (2012). It should be noted that diffusive
migration will govern VOC migration through a well-sealed membrane as the membrane effectively
prevents advective flow. Conversely advection will be the mechanism that drives flow through any defects
in the membrane.
Thick reinforced concrete floor slabs provide good resistance to vapour ingress in many cases and the
combination of slab and membrane should be incorporated into the risk assessment (taking into account
any construction joints and cracks around penetrations, whether joints are well sealed etc). Knowledge of
the slab design and the method of construction is crucial to understanding when cracks are likely to be
present that could allow vapour migration to occur. There are methods available that can help predict
crack density, eg the Danish JAGG model (Danish EPA, 2002), which is based on concrete weathering
algorithms.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
A simple example of a risk assessment for toluene is provided in Box 5.1. The example shows the
effect of providing different membranes and floor slab construction on toluene concentrations inside
the building. For a worse case assessment, it only considers the properties of the membrane and the
proportion of cracks in the concrete floor and takes no account of the rate of migration through the
ground or the concentration gradient across the membrane.
18
CIRIA, C748
Box 5.1
Calculation of permeation through a membrane with different floor slab constructions
A simple assessment of the effect of providing a membrane can be undertaken using the quoted permeation rate. In
this example two different membranes are being considered. Note the effect of attenuation of vapour migration in the
underlying soil and by the building floor slab is ignored in this example (ie it assumes the limiting factor is the maximum
diffusive flux that occurred through the membrane in tests and does not take account of the concentration of vapour
below the membrane). These other factors can have a significant effect on the risk posed by VOC vapours. It is assumed
the only barrier to vapour penetration is the membrane.
Table 5.1
Reliance on membrane only
Parameter/calculated value
Membrane 1: HDPE
Membrane 2: co-extruded membrane
with layers of HDPE, PA and EVOH
23 g/m2/h
Quoted permeation rate for toluene
(g/m2/h) in manufacturer’s literature
0.16 g/m2/h
(552 000 mg/m2/day) this figure
accounts for conversion for risk model (3763 mg/m2/day)
(24 hour period of exposure?)
Plan area of building
100 m2
100 m2
Height of ground floor
3.95 m
3.95 m
Air changes per hour in building
1 ach
1 ach
Equilibrium concentration of toluene in
occupied space using J&E equations
5823 mg/m3
for dilution in the occupied space
40 mg/m3
The allowable concentration of toluene inside a building can be taken as 5 mg/m3 (RfC used to derive TDI, see
Environment Agency, 2009b).
The analysis has been developed further by allowing for the presence of the floor slab. In this case the concrete floor slab
construction is assumed to be sufficiently thick and robust to minimise VOC permeation when compared to the rate of
permeation through the cracks. Two scenarios are considered, a perimeter crack around the edge of a reinforced concrete
floor slab (eg a ground bearing slab), and cracks across the slab at close centres (eg a precast concrete block and beam
floor slab).
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Table 5.2
Membrane and floor slab
Parameter/calculated value
Membrane 1 (same as Table 5.1)
Membrane 2 (same as Table 5.1)
Quoted permeation rate for toluene
(g/m2/h)
23 g/m2/h
0.16 g/m2/h
(552 000 mg/m /day)
(3763 mg/m2/day)
Plan area of building
100 m2
100 m2
Height of ground floor
3.95 m
3.95 m
Crack assumed around perimeter of slab
– migration pathway
1 mm
1 mm
Air changes per hour in building
1 ach
1 ach
Equilibrium concentration of toluene in
occupied space
2.9 mg/m3
0.02 mg/m3
Crack assumed across slab block and
beam floor – migration pathway
1 mm cracks at 300 mm centres
1 mm cracks at 300 mm centres
Air changes per hour in building
1 ach
1 ach
Equilibrium concentration of toluene in
occupied space using dilution equation
from J&E method
18.9 mg/m3
0.13 mg/m3
2
For a reinforced concrete ground bearing slab with an assumed 1 mm wide perimeter crack, both membranes would
reduce the estimated indoor air concentration to acceptable levels (<5 mg/m3). For a block and beam floor with lots of
cracks only membrane and floor slab would be suitable to reduce the indoor air concentration of toluene to acceptable
levels (if there is no underfloor ventilation to dilute the vapour coming out of the ground).
Guidance on the use of plastic membranes as VOC vapour barriers
19
An alternative approach would be to consider the concentration gradient of VOCs across the membrane.
This approach is slightly more detailed in terms of the modelling that is required, but uses the same
equations as the J&E method of risk assessment. It also requires soil vapour monitoring results. Using
the data from Table 2.1 and equations from Section 2.2 the influence of low soil vapour concentrations
on the mass flux rate and overall risk for Membrane 1 in Box 5.1 (HDPE) is illustrated in Table 5.3.
Membrane 1 is a 1.5 mm thick HDPE membrane with an assumed permeation coefficient of 7.22 × 10 -12
m2/s. The range of concentrations for toluene in soil vapour are indicative of values recorded on various
sites in the UK.
Table 5.3
Permeation through HDPE membrane if source concentration is taken into account
Concentration of toluene in soil vapour
0.050 mg/m3
0.100 mg/m3
100 mg/m3
Calculated mass flux through membrane, taking
account of concentration gradient across the
membrane
8.7 × 10 -7 mg/m2/h
1.7 × 10 -6 mg/m2/h
1.7 × 10 -3 mg/m2/h
Equilibrium concentration of toluene in occupied
space using dilution equation from J&E method
2.2 × 10 -7 mg/m3
4.4 × 10 -7 mg/m3
4.4 × 10 -4 mg/m3
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
This demonstrates how using data supplied by many manufacturers in isolation may lead to significant
over conservatism in the risk assessment.
20
CIRIA, C748
6 Geomembrane types, selection
and considerations
6.1
TYPES OF GEOMEMBRANE
The most common types of materials used in membranes that have been used for VOC vapour
protection and are available in UK, are:
flexible polypropylene
high density polyethylene/ethylenevinylalcohol sandwich (HDPE:EVOH:HDPE)
high density polyethylene/polyamide/ethylenevinylalcohol sandwich (HDPE:PA:EVOH:PA:HDPE)
high density polyethylene
low density polyethylene
linear low density polyethylene membrane
reinforced LDPE or HDPE with an aluminium core
spray applied asphalt-Latex membranes (bitumen/polystyrene emulsions).
Low density polyethylene DPM (low grade recycled material) has been used in the past as a membrane to
prevent the migration of VOCs. As stated in Section 1.1, standard DPM material is not usually suitable to
act as a barrier to VOC migration in buildings. It is unlikely to be installed in a manner that can provide
robust protection and data does not exist on VOC permeation rates for this type of material to allow it to
be modelled in the risk assessment. So it is not suitable and should not be used.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Membrane materials are available in a variety of thicknesses and types. Each material has different
characteristics that affect installation procedures, durability, lifespan and resistance to damage and
vapour permeability (Scheirs, 2009).
The person specifying a membrane should consider the most appropriate combination of properties
and ensure that the desired performance can be achieved and can be demonstrated as such in any given
situation (see Figure 6.1).
6.2
SELECTING A GEOMEMBRANE
The specified membrane and the reasons for its use should be described in the verification plan for
the site (see Mallett et al, 2014) for details of an appropriate verification plan). The maximum allowable
permeation rates of the critical chemicals should be determined from the risk assessment and should
be specified and used to demonstrate that there is no unacceptable risk (this should include permeation
rates at joints). A summary of the risk assessment should be reported within the verification plan.
The permeation rates quoted from manufacturer’s literature should not be used for this purpose. The
target should be site specific and allows any suitable membrane to be specified to meet the target rate
rather than limiting the choicer of membrane based on the properties from one manufacturer.
Membranes can be reinforced to improve the durability of the material and prevent over elongation.
Geosynthetic clay liners (a thin layer of dry clay powder sandwiched between two geotextiles) are not
suitable as barriers to VOC migration into buildings. This is because the liner relies on the bentonite
material becoming wet to form a barrier and this cannot be guaranteed. Even if it is pre-wetted during
installation the clay may dry out in a building application where it is not exposed to infiltrating rainfall
Guidance on the use of plastic membranes as VOC vapour barriers
21
and it will crack allowing vapour to migrate through it. Test results for water vapour transmission on
these types of barrier are not indicative of the likely rate of VOC vapour transmission (or the permanent
gases, methane and carbon dioxide).
The Environment Agency (2009a) provides a flow chart for choosing geomembranes in landfill
applications (see Figure 6.1). This flow chart has been adapted to suit the process of choosing a
membrane in a building application to prevent VOC vapour ingress.
Vapour membrane application
Step 1
Above floor slab
Below floor slab
Assess environmental framework
Required design life
Overall design framework
Step 2
Site specific considerations such as
floor slab construction
Risk assessment of site specific
properties that might affect the
performance of the membrane
including the consequences of failure
Determination of site specific minimum
allowable permeation rate
Step 3
Assess environmental stresses
Physical/mechanical
Chemical
Biological
Required design life
Required design life
Required design life
Method of jointing
Type of contamination present
Magnitude and extent of settlement
Potential attack by volatile gases
Assess macro and micro biological
exposure
Construction and installation stresses
Predicted temperatures
Predicted loadings
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Predicted temperatures
Assess environmental framework
Step 4
Figure 6.1
6.3
Select a membrane to match
the environmental setting with
a suitable factor of safety
Flow chart for choosing an appropriate membrane (adapted from Environment Agency, 2004)
WHAT TO CONSIDER
VOC membranes may not be required on all sites where there is VOC contamination in the ground. It
will depend on many factors such as the concentrations and type of contamination along with the depth
at which the contamination exists, type of soil, presence of groundwater and the nature of the building
to be constructed.
The factors identified in Figure 6.1 are discussed in more detail here and should be considered when
specifying a membrane as part of a protection system to mitigate the risks from VOC vapour migration
into buildings. They include:
22
CIRIA, C748
The required design life. Most housing has a design life of 60 years but will the contamination
remain present beyond this or will it have all naturally biodegraded by that time?
Membranes will degenerate over time due to processes such as oxidation, biological degradation,
chemical attack and degradation caused by ultra violet (UV) light. It is necessary to understand
how long a given membrane will maintain its performance over the design life. In practice,
degradation due to these factors will be minimal and is unlikely to affect the performance of a
membrane for the design life of most buildings.
As a general rule membranes used in vapour protection are not exposed to UV light after
installation. The membranes should only be exposed to UV for a short period of time during
installation and in addition membranes usually have UV stabilisers added to protect against UV
light. Where a membrane is exposed to UV light its life will be reduced.
Oxidation and biological degradation can occur, but for most membranes stabilisers are added
to the materials and this will be a very slow process. The main effect of the ageing processes
is to reduce the strength of the membrane. Where the liner is subjected to long-term stresses,
stress cracking will also lead to the development of holes. Where a membrane is above a slab it
does not usually have to carry any great load after installation, so the adverse effects associated
with these ageing processes should not be significant. Even if the membrane is below a slab and
settlement may occur it is unlikely that the loss of strength due to these degradation effects will be
significant. The ability of membranes to prevent the migration of VOCs should, in most cases be
sustainable and have a very long service life at an ambient temperature of about 20°C provided it
is not exposed to other factors that could cause loss of performance. Examples of other factors that
could cause loss of performance and reduce the service life of the membrane include temperature
increases (ie > 25°C) and/or where membranes are subject to long-term stresses or are exposed to
chemicals at a high enough concentration (eg benzene).
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
The risk of VOC migration into the building and how much reliance will be placed on the
membrane (ie the consequence of failure). The maximum permeation rate of challenge chemical(s)
through the membrane should be known (permeation data).
Will the membrane be exposed to concentrations of challenge chemicals that could adversely affect
its durability and performance in the long-term? Resistance to the challenge chemicals should be
understood via chemical resistance tests. The membranes for building protection are most likely to
be placed over the floor slab or below it on a prepared sub-base or a venting layer. The membrane
is not likely to be in direct contact with the source of any vapours and so the exposure is far less
than in landfill. The vapour concentrations should be low, which will minimise the risk of chemical
degradation of the material. (Most testing that shows damage by VOCs to membranes is based
on the use of 100 per cent concentration of the challenge chemical rather than a dilute solution.
Vapour concentrations in the ground immediately below a slab are typically at concentrations that
are less than one per cent v/v in air). It may also be necessary to consider the synergistic effects of
combinations of chemicals, for example using the HSP discussed in Section 2.4.
Is the membrane subject to tensile loads in the permanent condition? Factors that affect this will
be the extent of settlement, whether the membrane is above or below the floor slab and likely
temperature variations. The tensile strength of the sheet material and welds is required for this
assessment (allowing for loss of strength due to exposure to challenge chemicals).
Quality and robustness of installation. This can be increased by verification and integrity testing of
the materials and seams, seals etc.
Likelihood of damage to membrane during and after construction, which will help define the
puncture resistance, impact resistance and tear strength. This is very important because of the low
permeation rates through the installed membrane for VOCs.
It has also been known for rodents to gnaw through exposed membranes, although it is extremely
rare and the conditions that would allow this (an exposed membrane) are not recommended in
buildings. The membrane should be located in a position within the construction such that it
cannot be reached by rodents under most normal circumstances.
Are welded seams required? This will determine the minimum thickness of membrane.
Guidance on the use of plastic membranes as VOC vapour barriers
23
If specifying that a membrane has a British Board of Agrément (BBA) or other certificate ensures
that the requirements of the certificate are appropriate for the site and proposed use. The fact that a
membrane has a BBA certificate does not guarantee it will be suitable for a given site.
Inspections of 15 to 20 year old HDPE membranes exposed during refurbishment and extension works
at several supermarkets and other similar premises recorded no visual evidence of significant general
degradation due to UV, oxidation etc that would affect the performance as a barrier. The membranes
were still intact and performing as a gas or vapour barrier. However, it is essential that on-site QA/
QC measures are implemented and maintained in such scenarios to safeguard the performance and
integrity of the membrane. Such measures should be documented for future reference.
6.4
KEY PERFORMANCE PROPERTIES
Using the considerations from Section 6.3, the key performance properties for a membrane on any
particular site can be determined. Where a membrane has to provide resistance to VOC vapour
migration the following parameters should be specified along with appropriate values. The reasons for
choosing or omitting the properties should be justified and clearly recorded.
Unfortunately at present it has become clear from research by the authors that there is not a standard
suite of test results that are provided by suppliers of membranes for use as being fit for purpose as VOC
membranes to prevent migration of VOC contamination.
So, it is very difficult for designers to make informed judgements about the most suitable membrane
for a given application. As an example few suppliers provide details of the puncture resistance of their
membranes, which is one of the most critical properties. Without this data it is not possible to assess
claims in brochures regarding “very high puncture resistance!” Where properties are quoted there is a
range of different test methods used from ASTM to EN Standards, and from those normally used to test
geomembranes to those used on roofing membranes.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
In the absence of a defined approach, it is suggested that consultants and regulators consider the factors
and test methods in Table 6.1 when deciding on an appropriate membrane.
As a general principle where several different standards are currently referred to by various suppliers
the British and European Standards should take precedence over standards such as ASTM. For VOC
barriers in buildings the test methods proposed in Table 6.1 are based on the guidance in BS EN
13492:2013, unless alternative test methods are considered to be more relevant to the application of
barriers for limiting VOC migration into buildings.
24
CIRIA, C748
Table 6.1
Key performance properties
Properties
Test methods
Thickness
BS EN 1849-2:2009. If this test is not suitable because of the nature
of the membrane then the test method from BS EN ISO 9863-1
2005 may be used as an alternative. This method is applicable to
Index
all geosynthetics including scrim reinforced, textured and smooth
geomembranes and multi-layered/aluminium foil membranes.
Permeation rate
ISO 15105-2:2003. HSP modelling can be used to extend the suite
of suite of VOCs at
of permeation rates provided the membrane is tested for the basic
various concentrations suite of chemicals listed in Section 2.3.
Tensile strength and
elongation at yield or
1994ak (and creep
under sustained longterm loads)
ASTM D4885-01. This test is designed to provide the performance
strength of geomembranes. A 200 mm wide test strip is preferred.
There are other tests such as ASTM D882-12 that are index tests
and are less appropriate for design purposes.
Note that ISO 10319:2008 is not applicable to polymeric
geomembranes. BS EN 12311-2:2010 only uses a 50 mm wide test
strip which is not as good as a 200 mm strip for design purposes.
Index or performance test
Performance test if done
using site specific vapour
concentrations. For
standard chemical suite
and concentrations they are
index tests
Performance (required for
design assessment)
BS EN 527-3:2003 uses a narrow sample.
If creep under sustained long-term loads is a significant
consideration then specific creep testing may be required to
determine the creep rupture strength for the required design life.
Index
Puncture resistance
BS EN ISO 12236:2006
Index
Impact resistance
BS EN 12691:2006. Tests onto both hard and soft substrates
(Methods A and B).
Index
Tear resistance
ISO 34-1:2010. Method B, angle specimen without nick at a speed
of 50 mm/min
Index
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Index tests measure the physical and mechanical properties of a membrane. Performance tests measure
the actual engineering properties in conditions similar to those that are likely to be found on a site. Care
should be taken when using index tests and comparing them to design values. Appropriate factors of
safety are required and a judgement on how the test result relates to the design calculations.
Thickness
For any given material as the membrane thickness increases the puncture, impact and tear resistance will
increase. The same thickness of different materials will have different properties, ie a thin hard material
can provide the same puncture resistance, tensile strength etc as a thicker, softer material. However,
thickness is very important when considering welding. Membranes less than 1 mm thick are much
more prone to welding problems, especially burning holes in the material (Scheirs, 2009). Welding the
protective membrane layer on an aluminium foil laminate is extremely difficult to complete effectively.
For landfill liners, Scheirs (2009) reports the USEPA specify membranes that are protected should
be 0.75 mm thick and unprotected ones should be at least 1.14 mm for scrim reinforced membranes.
Conversely thinner membranes of the same material tend to be more flexible and are easier to install
especially in buildings where corners and other details are much more prevalent. Flexible membranes
can also be prefabricated into panels in the factory. So there is a trade-off between robustness and the
risk of defects due to difficulty of installation.
A similar approach to the USEPA could be used for VOC barriers taking account of the different
requirements in buildings:
minimum thickness for VOC barrier – taped joints, 0.5 mm (not including scrim thickness) –
providing the taped joints have been demonstrated to give the same permeation rates as the
membrane material (see Section 4.2). The durability of the joint should also be assessed by testing
Guidance on the use of plastic membranes as VOC vapour barriers
25
of the performance when exposed to VOCs or directly with other chemicals present on a site
because the adhesives may be affected by VOCs
minimum thickness for VOC barrier – welded joints, 1 mm unless it can be shown by testing of
welded joints that thinner membranes can be joined in a satisfactory manner (using a seam test).
Permeation rate of each relevant VOC
This will be defined based on the site specific risk assessment of VOC migration into the building.
Where taped joints are proposed the permeation testing should be carried out on a sample of jointed
membrane.
Tensile strength and elongation at yield (plus creep under sustained load)
If the membrane is exposed to tensile forces, for example if it is below a floor slab and settlement could
induce tensile stress in the membrane. Tensile stress can also occur due to thermal expansion and
contraction of the membrane and wind lift during installation as well as general stress and strain from
the installation process. Elongation at yield determines the ability of a membrane to remain serviceable
when subject to tensile stress and strains (ie how stretchy is the membrane). The tensile behaviour of
welds may also be important. An example of thermal stress and settlement stress analysis is provided
in Box 6.1. Polymeric materials such as HDPE, LDPE, PP etc creep (ie they continue to deflect under a
constant load. These effects should be considered if the membrane is subject to long-term applied loads
(eg due to settlement).
Box 6.1
Thermal stress and settlement stress analysis
Thermal stress
If the temperature changes after a membrane is laid it will either expand or contract. If it is restrained at the edges and
cannot contract this will cause tensile stress to occur in the membrane. The contraction in a membrane laid below a floor
slab can be estimated as follows (Schiers, 2009).
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Table 6.2
Tensile strength required to resist thermally induced forces
Original length of membrane, L
10
m
Coefficient of thermal expansion of membrane, C
1.9 × 10 -4
m/m°C
Change in temperature, DT
15
°C
Assume membrane is laid at 25°C and
ambient temperature below slab is 10°C
Change in length due to change in temperature
0.029
m
∆L= LC∆T
Strain due to contraction, e
2.9 × 10 -3
m/m
Young’s modulus of membrane, E (allowing for creep)
180
MPa
Tensile stress due to contraction, S
522
kPa
Factor of safety
2
Tensile strength required
1044
S = Ee
kPa
This is a worst case analysis because in practice the tensile stress would most likely dissipate via friction resistance from
the overlying concrete.
Settlement
Settlement can be an issue where a membrane is located below the floor slab and is attached to a rigid structure at
discrete points (eg ground beams or pile caps). The tension in the membrane can be assessed using the approach of
Giroud et al (1995). An example for a suspended floor slab is given in Table 6.3. It can be adapted to consider settlement
below a ground bearing slab by considering the continuation of a membrane through walls as the fixed point.
26
CIRIA, C748
Box 6.1
Table 6.3
Thermal stress and settlement stress analysis (contd)
Tensile strength required to resist settlement induced forces
Density of HDPE
940
Kg/m3
Thickness of membrane
1
mm
Width between fixed supports (eg membrane fixed to pile cap), L 6
m
p = Weight of membrane and overlying material
0.0094
kN/m2
In this case consider self
weight of membrane only
δ = interface friction between soil and membrane
10
Degrees
Assumed
Interface shear stress between soil and membrane, τ = ptand
0.0017
kN/m2
Tension at connection to rigid structure, T = τL
0.01
kN/metre
width
FOS
3
Tensile strength required
0.03
Note this should be long-term
kN/metre
strength for the design life of
width
the building
eall = Allowable strain in membrane
0.03
Assume three per cent strain
is maximum acceptable to
limit risk of stress cracking
(Smith and Orman, 1994)
Estimated total settlement of soil below membrane, s (this will
be the differential settlement between the soil and the fixed
point of the pile caps)
100
mm
eact = Actual strain in membrane = s/L
1.6
%
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
In reinforced earth design the specified strength would be based on long-term creep test results. If this data is not
available it is reasonable to assume in this scenario (low risk situations) creep will not be significant if the mobilised
tensile strength is less than 10 per cent of the yield stress determined from the wide strip tensile test. In critical
situations specific creep testing on the membrane should be carried out.
The wind uplift forces for larger areas of membrane that are left exposed for an extended period during
construction can be calculated in accordance with BS EN 1991-1-4:2005+A1:2010. This will allow a
minimum tensile strength to be specified along with the maximum area that can be left unweighted
during construction. Normally applying self-adhesive or other bonded systems is sufficient to resist the
effects of wind suction.
If settlement below a building is a significant possibility then rather than relying on the tensile strength
of the membrane it may be better to lay geogrid reinforcement below it to carry the load.
Puncture resistance
Puncture forces on membranes are most likely during construction. The most common causes of
puncture forces are from laser screeds that pass over the membrane to construct the floor slab or simply
from a person standing on top of a membrane when there is a piece of grit below it. An example of how
to estimate the puncture force on a membrane is provided in Box 6.2.
Guidance on the use of plastic membranes as VOC vapour barriers
27
Box 6.2
Estimating required puncture resistance
Example calculation (adapted from John, 1987)
Assume an even stress from the foot of a construction worker is applied to the top of a membrane that is laid over a floor
slab with a 5 mm diameter piece of sharp angular grit left below it. Assume the sole of the boot is malleable and pushes
the membrane down the side of the piece of grit.
CBR puncture resistance required (consider the forces on the membrane)
Average aggregate particle diameter of material
below membrane, d50 in the analysis
Diameter of contact area of aggregate with
membrane, dc = d50/4 for sharp aggregate
0.005
m
0.00013
m
The reaction stress is the pressure acting on the membrane – in this case from the weight of a person
Pressure on top of membrane from weight
of construction worker, P =
55
kN/m2 Human male (1.8 m tall,
medium build): 55kPa
0.0011
kN
Force = PPd502/4
7.3 × 10 -7
kN
Force = PPdc2/4
Net force acting on membrane, Fp
0.0011
kN
Fp = Fd–Fu
CBR puncture force = 0.0496 Fp/dc
0.412
kN
Assumes 50 mm dia plunger
in CBR test as in Table 6.1
Force acting down on membrane from soil, Fd
Force acting up from the grit, Fu
FOS
CBR puncture resistance required
2
824
N
Impact resistance
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Membranes should have a reasonable resistance to damage caused during or after installation before
being covered. Damage can occur from things such as dropped tools. None of the tests available for
impact testing can be related directly to impact forces on a site. So, a purely empirical approach needs to
be taken.
The test method in BS EN 12691:2006 uses a 500g puncturing tool that drops onto the sample. The
results are reported as the drop height of the tool that has not caused water leakage in four out five
specimens. It would seem reasonable to expect that if someone could drop a weight of 500g from their
hand at, for example, 750 mm height (arm down by side) it should not puncture a membrane. The
actual value specified for a site should be based on the risk of this occurring (is the membrane to have
protection boards, will it be covered before other work is carried out etc) and whether any damage is
likely to be observed and repaired (ie the level of verification and integrity testing).
Tear strength
The membrane will most likely be subject to tear forces during installation. The forces can occur when
larger areas of membrane are pulled into place or if there is wind uplift. The presence of a scrim
reinforcement such as polyester will increase the tear resistance and limits the propagation of tears in the
membrane. Tear resistance is also an indirect indicator of abrasion resistance.
Probably the most common reason for tearing of membranes in buildings is sharp objects being dragged
across the membrane after it has been placed (for example pallets, scaffolding or steel frames). The
standard tests for tear resistance are not really designed to test for this type of tearing and the tests
are actually a test of resistance to propagation of an existing tear. The most practical way of specifying
tear resistance is by observing samples of the proposed membrane in service. For a membrane to be
used to prevent vapour migration into a building, then it should have a higher resistance to tearing.
28
CIRIA, C748
For example, a membrane with tear strength greater than 350N in both machine and cross direction is
desirable in this application.
6.5
CE MARKING AND THIRD PARTY ACCREDITATION
Regulation (EU) N 305/2011 (Construction Products Regulations 2011) cover all materials used on
construction sites including the use of gas membranes in gas protection systems. It is only possible to
CE mark a product against a harmonised European Standard, and these do not exist for gas or vapour
membranes at present. Neither of the current standing CEN committees that could cover gas membranes
have developed specific testing regimes for gas barrier membranes and it seems unlikely that there will
be bespoke harmonised EN standards for some time. CE marking is a self-certified scheme that indicates
a product conforms to relevant EC Directives.
CE marking will allow checks on site to determine the manufacturing origin of the product, its batch
number and offers the opportunity to request the factory quality control data for that batch of material.
For gas membranes it will be an indication of quality control during manufacture but it will be no
guarantee that a product is right for any given application.
For example, a DPM can have a CE mark but it does not mean it is suitable as a gas resistant membrane.
Equally, a membrane can have a CE mark but it does not mean it has sufficient robustness to resist
damage from steel reinforcement placed on it without some form of additional protection. CE marking
should not be used on its own to determine if a membrane is suitable for a given application.
Currently gas membranes are being CE marked to either the DP/ basement tanking standard (BS EN
13967:2012) or to the waterproofing standard (EN 13967) on the basis that they are usually placed in the
same position as damp or waterproofing membranes and will often fulfil a duplicate role. However these
standards do not include any requirements that would demonstrate a membrane is suitable as a barrier
to VOC vapour migration.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Third party accreditation certificates (eg BBA) should also be used in an appropriate way. Simply asking
for a certified membrane does not give adequate assurance that a membrane is suitable for a particular
site. The certificate should be checked for each site to make sure the requirements in the certificate are
being followed during installation (eg many BBA certificates for membranes specify the use of protection
boards) and that the rate of VOC permeation is acceptable.
Guidance on the use of plastic membranes as VOC vapour barriers
29
7 Installation and verification
The dominant mode of fluid transport through poorly installed membranes (including vapour
migration) is via holes due to poor welds, material defects, damage etc (Scheirs, 2009, and CIEH, 2008).
This rate of permeation via this mode of transport can be much greater than diffusion through the
intact membrane. So installation and subsequent verification is vital if a membrane is to act as a barrier
to vapour ingress into buildings. Because even minor defects can significantly compromise the ability
of the membrane to maintain internal concentrations below the acceptable limits, the highest level of
installer competence and verification/integrity testing is necessary. The installation and verification
requirements that should normally be adopted are shown in Table 7.1.
Table 7.1Verification and integrity testing requirements for membranes required to resist VOC ingress (adapted from
Mallett et al, 2014)
Situation A: Public, commercial, retail, industrial, apartments
Gas regime/risk
High risk (as
defined by
Mallett, 2014)
Membrane to act
as VOC barrier
High risk (as
defined by
Mallett, 2014)
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Membrane to act
as VOC barrier
Type of slab
Non reinforced
All slabs
Reinforced
All slabs
Installer
experience
Suggested levels of verification and integrity testing
Verifier (consultant4 or third party qualified and experienced
1
installer
) to conduct a thorough verification (visual) inspection
Qualified1 and
before all concrete pours. All joints, pipe penetrations
experienced
independently air lanced to ASTM D4437-99. Installer to
installer (50
supply sign off sheets (verification evidence) to verifier for
per cent of
all pours. Consideration given to need for scope of integrity
operatives to hold
testing (eg initially on 25 to 50 per cent of pours then falling
qualification)
to 10 to 25 per cent if acceptable results obtained and no
concerns raised by visual inspections).
Verifier (consultant4 or third party qualified and experienced
installer1) to conduct a thorough verification (visual) inspection
Qualified1 and
prior to all concrete pours including vented void, subgrade etc.
experienced
All joints, pipe penetrations etc independently air lanced to
installer (50% of
ASTM D4437-99. 100 per cent leak detection considered on
operatives to hold VOC/hydrocarbon contaminated sites. Consideration given to
qualification)
need for/scope of integrity testing (eg initially on 50 to 25 per
cent of pours then falling to 25 to 10 per cent if acceptable
results obtained and no concerns raised by visual inspections).
Situation B: Low rise housing only
High risk (as
defined by
Mallett, 2014)
Membrane to act
as VOC barrier
All slabs with
min 150 mm
ventilated sub
floor void
Verifier (consultant4 or third party qualified and experienced
installer1) to conduct thorough verification (visual) inspection
Qualified1 and
of all plots, and after placement of reinforcement if no
experienced
protection provided. All joints, pipe penetrations etc air lanced
installer (all
to ASTM D4437. Consideration given to need for/scope of
operatives to hold
integrity testing (eg initially on 30 to 50 per cent of plots
qualification)
then falling to 0 to 10 per cent of plots if acceptable results
obtained and no concerns raised by visual inspections)3.
Notes
1 Relevant qualification is NVQ Level 2 (gas protection installation).
2Before the works start the contractor should produce a detailed installation plan including method statement, CQA procedures and
qualifications, on receipt of these the verification protocol could be increased or reduced.
3Consideration should be given to carrying out leak detection (ie smoke, tracer gas or dielectric testing) on the first plots. If an
unacceptable amount of holes are found during these tests then the verification consultant should discuss with the relevant personnel,
strategies to prevent this occurring, these could include changing material, improving subgrade preparation, putting up warning signs to
reduce the amount of trafficking etc.
4Verification consultant should be competent, experienced and suitably trained. A statement detailing their qualifications and relevant
experience should be included in the verification plan.
5Air lancing is the only integrity test that has an independently recognised international standard that is suitable for testing taped and
welded seams and should be used at the frequency suggested in the table.
6 Mallett et al (2014) also provides advice on the contents of the verification report that is normally submitted to the regulators.
30
CIRIA, C748
The importance of verification is shown in Figure 7.1, which is an example installation of a membrane
intended to act as a barrier to VOC migration. The installer has tried to weld the main membrane, which
is a suitable barrier to VOC migration, to a normal DPM. However, there are three issues with this:
1
The DPM would not act as a barrier to VOC migration.
2
The DPM is a different material to the membrane and different plastic materials cannot be welded
together.
3
The sealing detail around the services is poor. The services should have more space between them
to allow the membrane to be sealed to them effectively.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Figure 7.1
Unacceptable installation of a VOC membrane
Guidance on the use of plastic membranes as VOC vapour barriers
31
References
ABBOTT, S and HANSEN, C M (2008) Solubility parameters in practice – complete with software, data and
examples, first edition, Hansen-Solubility (ISBN: 978-0-95512-202-6). Go to: www.hansen-solubility.com
AUGUST H and TATZKY, R (1984) “Permeabilities of commercially available polymeric liners for
hazardous landfill leachate organic constituents”. In: Proc int conf on geomembranes, Denver, Colorado, vol
1, Industrial Fabrics Association, St Paul, Minn, pp 163–168
BAKER, K, HAYWARD, H, POTTER, L, BRADLEY, D and MACLEOD, C (2009) The VOCs handbook.
Investigating, assessing and managing risks from inhalation of VOCs at land affected by contamination, C682,
CIRIA, London (ISBN: 978-0-86017-685-5). Go to: www.ciria.org
CHIN, J, ASAVANICH, P, MOON, K W and PARK, J K (2013) “Development of reactive artificial liner
using recycled materials. 2. Chemical transport properties” Waste Management Research, vol 31, 7, SAGE
Journals, UK, pp 714–721
CIEH (2008) Local authority guide to ground gas, Chartered Institution of Environmental Health, UK
(ISBN: 978-1-90430-676-4). Go to: www.cieh.org
DANISH EPA (2002) Guidelines on remediation of contaminated sites, Environmental Guidelines, No 7,
Danish Environmental Protection Agency, Danish Ministry of the Environment, Denmark.
Go to: http://www2.mst.dk/udgiv/publications/2002/87-7972-280-6/pdf/87-7972-281-4.pdf
DUNCAN, B, URQUHART, J and ROBERTS, S (2005) Review of measurement and modelling of permeation
and diffusion in polymers, NPL Report DEPC MPR 012, National Physical Laboratory, Middlesex, UK.
Go to: http://resource.npl.co.uk/materials/polyproc/iag/october2005/depc_mpr_012.pdf
ENVIRONMENT AGENCY (2004) Model Procedures for the management of land contamination,
Contaminated Land Report 11 (CLR 11), Environment Agency, Bristol, UK (ISBN: 1-84432-295-5).
Go to: http://tinyurl.com/q6scs6x
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
ENVIRONMENT AGENCY (2009a) Using geomembranes in landfill engineering, LFE5, GEHO0409BPNHE-E, Environment Agency, Bristol. Go to: http://tinyurl.com/nnspl99
ENVIRONMENT AGENCY (2009b) Contaminants in soil: updated collation of toxicological data and intake
values for humans. Dioxins, furans and dioxin-like PCBs, Better Regulation Science Programme, Science
report: SC050021/TOX 12, Environment Agency, Bristol. Go to: http://tinyurl.com/oy26vxa
GIROUD, J P, BADU-TWENEBOAH, K and SODERMAN, K L (1995) “Theoretical analysis of
geomembrane puncture” Geosynthetics International, vol 2, 6, Industrial Fabrics Association International,
USA, pp 1019–1048
HANSEN, C M (1999) Hansen solubility parameters: A user’s handbook, CRC Press, UK (ISBN: 978-0-84937-248-3)
HANSEN, C M (2009) “Understanding absorption in polymers: Key to improving barrier properties”.
In: Proc 50th Nordic Polymer Days, 29–31 May 2013, Helsinki, Finland
ISLAM, M Z and ROWE, RK (2001) “Permeation of BTEX through unaged and aged HDPE
geomembranes” Journal of Geotechnical and Geoenvironmental Engineering, vol 135, 8, American Society of
Civil Engineers, Reston, VA, pp 1130–1140
ITRC (2007) Vapour intrusion pathway: a practical guideline, ITRC Vapor Intrusion Team, Interstate
Technology and Regulatory Council, Washington DC, USA. Go to: www.itrcweb.org/documents/vi-1.pdf
32
CIRIA, C748
JOHN, N W M (1987) Geotextiles, Blackie and Son, London (ISBN: 978-0-41201-351-5)
LAYFIELD CONSTRUCTION MATERIALS (2013) Chemical resistance tables, Layfield Construction
Materials, UK. Go to: www.layfieldgeosynthetics.com/pages/EGI/Specifications.aspx?id=5034
MALLETT, H, COX, L, WILSON, S and CORBAN, M (2014) Good practice on the testing and verification
of protection systems for buildings against hazardous ground gases, C735, CIRIA, London (ISBN: 978-0-86017739-5). Go to: www.ciria.org
MASSEY, L K (2003) Permeability properties of plastics and elastomers. A guide to packaging and barrier
materials, second edition, Plastics Design Library/William Andrew Publishing, USA (ISBN: 1-88420-797-9)
MCWATTERS, R S and ROWE, R K (2009) “Transport of volatile organic compounds through PVC and
LLDPE geomembranes from both aqueous and vapour phases” Geosynthetics International, 2009, vol 16, 6,
Institution of Civil Engineers, Thomas Telford, UK, pp 468–481
MCWATTERS, R S and ROWE, K R (2010) “Diffusive transport of VOCs through LLDPE and two
coextruded geomembranes” Journal of Geotechnical and Geoenvironmental Engineering, vol 136, 9,
American Society of Civil Engineers, Reston VA, USA, pp 1167–117
NERIN, C, CANELLAS, E, ROMERO, J and RODRIGUEZ, A (2007) “A clever strategy for permeability
studies of methyl bromide and some organic compounds through high-barrier plastic films” International
Journal of Environmental Analytical Chemistry, vol 87, 12, Taylor & Francis, UK, pp 86 –874
NHBC and ENVIRONMENT AGENCY (2008a) Guidance for the safe development of housing on land
affected by contamination, vol 1, R&D Publication 66, National House-Building Council, and Environment
Agency, UK. Go to: http://tinyurl.com/oatnase
NHBC and ENVIRONMENT AGENCY (2008b) Guidance for the safe development of housing on land
affected by contamination. Appendices and Annexes, vol 2, R&D Publication 66, National House-Building
Council, and Environment Agency, UK/. Go to: http://tinyurl.com/ocmv8qf
ROWAD INTERNATIONAL (2013) Chemical resistance for geomembranes. Technical Note, Rowad International
Geosynthetics Co Ltd, Kingdom of Saudi Arabia. Go to: www.rowadgeo.com/Chemical_Resistance.pdf
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
SANGAM, H P and ROWE, R K (2001) “Migration of dilute aqueous organic pollutants through HDPE
geomembranes” Geotextiles and Geomembranes, vol 19, 6, Elsevier BV, UK, pp 329–357
SCHEIRS, J (2009) A guide to polymeric geomembranes, John Wiley & Sons Ltd, London (ISBN: 978-047051-920-2)
SMITH, M and ORMAN, M (1994) Copper heap leaching – a case for PVC liners, Geotechnical Fabrics
Report, spring, Industrial Fabrics Association International, Minnesota, USA
THOMAS, R W and KOERNER, R M (1996) “Advances in HDPE barrier walls” Geotextiles and
Geomembranes, vol 14, 7–8, Elsevier BV, UK, pp 393–408
USEPA (1992) Method 9090A. Compatibility test for wastes and membrane liners, United States Environment
Protection Agency, Washington DC, USA. Go to: http://tinyurl.com/q7peu3y
WELBURN, P, BAKER, K, BORTHWICK, K and MACLEOD, C (2012) Remediating and mitigating risks
from volatile organic compound (VOC) vapours from land affected by contamination, C716, CIRIA, London
(ISBN: 978-0-86017-719-7). Go to: www.ciria.org
WILSON, S (2008a) “Modular approach to analysing vapour migration into buildings in the UK” Land
Contamination & Reclamation, vol 16, 3, EPP Publications, UK, pp 223–226
WILSON, S (2008b) “Brownfield vapor barriers: chemical compatibility, testing, and advances in
materials science”. In: Proc of the sixth int conf on remediation of chlorinated and recalcitrant compounds,
Monterey, CA, USA, May 2008, M Bruce (ed), Battelle Press, Columbus, OH, USA (ISBN: 1-57477-163-9)
Guidance on the use of plastic membranes as VOC vapour barriers
33
STATUTES
Standards
British
BS EN 527-3:2003 Office furniture. Work tables and desks. Methods of test for the determination of the stability
and the mechanical strength of the structure
BS EN 1849-2:2009 Flexible sheets for waterproofing. Determination of thickness and mass per unit area. Plastic
and rubber sheets
BS EN 1991-1-4:2005+A1:2010 Eurocode 1. Actions on structures. General actions. Wind actions
BS EN 12311-2:2010 Flexible sheets for waterproofing. Determination of tensile properties. Plastic and rubber
sheets for roof waterproofing
BS EN 12691:2006 Flexible sheets for waterproofing. Bitumen, plastic and rubber sheets for roof waterproofing.
Determination of resistance to impact
BS EN 13492:2013 Geosynthetic barriers. Characteristics required for use in the construction of liquid waste
disposal sites, transfer stations or secondary containment
BS EN 13967:2012 Flexible sheets for waterproofing. Plastic and rubber damp proof sheets including plastic and
rubber basement tanking sheet. Definitions and characteristics
BS EN 14414:2004 Geosynthetics. Screening test method for determining chemical resistance for landfill applications
BS EN 14415:2004 Geosynthetic barriers. Test method for determining the resistance to leaching
European
BS ISO 34-1:2010 Rubber, vulcanized or thermoplastic. Determination of tear strength. Trouser, angle and
crescent test pieces
BS EN ISO 6179:2010 Rubber, vulcanized or thermoplastic. Rubber sheets and rubber-coated fabrics.
Determination of transmission rate of volatile liquids (gravimetric technique)
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
BS EN ISO 9863-1:2005 Geosynthetics. Determination of thickness at specified pressures. Single layers
BS EN ISO 10319:2008 Geosynthetics. Wide-width tensile test
BS EN ISO 12236:2006 Geosynthetics. Static puncture test (CBR test)
BS EN ISO 15105-2:2003 Plastics. Film and sheeting. Determination of gas-transmission rate. Part 2: Equalpressure method
USA
ASTM E96/E96M–05 Standard test methods for water vapour transmission of materials
ASTM D882-12 Standard test method for tensile properties of thin plastic sheeting
ASTM D4437-99 Standard practice for determining the integrity of field seams used in joining flexible polymeric
sheet geomembranes
ASTM D4885-01 Standard test method for determining performance strength of geomembranes by the wide strip
tensile method
ASTM D5747/D5747M-08 (2013)e1 Standard practice for tests to evaluate the chemical resistance of
geomembranes to liquids
34
CIRIA, C748
ASTM D5322-98 (2009) Standard practice for immersion procedures for evaluating the chemical resistance of
geosynthetics to liquids
ASTM D5886–95 (reapproved 2011) Standard guide for selection of test methods to determine rate of fluid
permeation through geomembranes for specific applications
Regulations
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Regulation (EU) N 305/2011 of the European Parliament and of the Council of 9 March 2011 laying
down harmonised conditions for the marketing of construction products and repealing Council Directive
89/106/EEC (Construction Products Regulations 2011 and approved documents 2013)
Guidance on the use of plastic membranes as VOC vapour barriers
35
Further reading
ENVIRONMENT AGENCY (2001) Assessment and management of risks to buildings, building materials and
services from land contamination, R&D Technical Report P5-035/TR/01, Environment Agency, Bristol
(ISBN: 1-85705-484-9). Go to: http://tinyurl.com/nut7yyf
GEORGE, S C and THOMAS, S (2001) “Transport phenomena through polymeric systems” Progress in
Polymer Science, vol 26, 6, Elsevier BV, UK, pp 985–1017
HANSEN, C M (1971) “Solubility in the coatings industry” Farg och Lack, vol 17, 4, pp 69–77
PAUL, V (1994) Performance of building materials on contaminated land, BR 255, BRE Press, Building
Research Establishment, Garston, Watford (ISBN: 0-85125-624-4)
POÇAS, M F, OLIVEIRA, J C, OLIVEIRA, F A and HOGG, T (2008) “A critical survey of predictive
mathematical models for migration from packaging” Critical Reviews in Food Science and Nutrition, vol 48,
10, Taylor & Francis, UK, pp 913–28
PRIVETT, K D, MATTHEWS, S and HODGES, R A (1996) Barriers, liners and cover systems for containment
and control of land contamination, SP124, CIRIA, London (ISBN: 978-0-86017-437-0). Go to: www.ciria.org
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
UKWIR (2011) Pipe materials selection and specification for use in contaminated land – final report, 14/RG/08/6,
UK Water Industry Research, UK. Go to: www.ukwir.org
36
CIRIA, C748
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Core and Associate members
AECOM Ltd
Loughborough University
Arup Group Ltd
Ministry of Justice
Atkins Consultants Limited
Morgan Sindall (Infrastructure) Plc
Balfour Beatty Civil Engineering Ltd
Mott MacDonald Group Ltd
BAM Nuttall Ltd
Mouchel
Black & Veatch Ltd
MWH
Buro Happold Engineers Limited
Network Rail
BWB Consulting Ltd
Northumbrian Water Limited
Cardiff University
Rail Safety and Standards Board
Environment Agency
Royal HaskoningDHV
Galliford Try plc
RSK Group Ltd
Gatwick Airport Ltd
RWE Npower plc
Geotechnical Consulting Group
Sellafield Ltd
Golder Associates (Europe) Ltd
Sir Robert McAlpine Ltd
Halcrow Group Limited
SKM Enviros Consulting Ltd
Health & Safety Executive
SLR Consulting Ltd
Heathrow Airport Holdings Ltd
Temple Group Ltd
High Speed Two (HS2)
Thames Water Utilities Ltd
Highways Agency
United Utilities Plc
Homes and Communities Agency
University College London
HR Wallingford Ltd
University of Bradford
Imperial College London
University of Reading
Institution of Civil Engineers
University of Southampton
Lafarge Tarmac
WYG Group (Nottingham Office)
Laing O’Rourke
London Underground Ltd
October 2014
Buildings are often constructed on sites where volatile organic compounds (VOC) are
present in the ground. This publication provides guidance on the use of plastic
membranes to reduce vapour migration into buildings. It discusses vapour transport
mechanisms that occur in membranes, factors that influence this and the test methods
available for determining the rate of vapour permeation. It then explains how to use the
results in a risk assessment. Durability and resistance to damage when plastic
membranes are exposed to VOCs is a concern and the guide considers the potential for
degradation and which test methods are suitable to assess performance. This guide
should allow readers to make a robust evaluation of the contribution to membranes to
reducing indoor inhalation risk from VOCs in the ground and to specify the most
appropriate type of membrane for any given site.
© Copyright CIRIA 2015. No unauthorised copying or distribution permitted. For use by Triton Systems only.
Guidance on the use of plastic
membranes as VOC vapour barriers
C748
9 780860 177531