Chemistry Review Worksheet: Atomic Structure & Periodic Table
advertisement

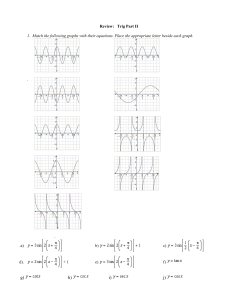
Name: ________________________ Class: ___________________ Date: __________ Chapter 4 & 5 Review 1. Unlike Democritus, Aristotle did not believe that matter was composed of tiny, indivisible ____________________. 2. The results of Rutherford’s gold foil experiment demonstrated that the ____________________ occupies a very small amount of the total space inside an atom. 3. Protons and ____________________ are found in the nucleus of an atom. 4. Neutrons and ____________________ have almost the same mass. 5. If element Q has 11 protons, its atomic ____________________ is 11. 6. The nuclei of isotopes contain different numbers of ____________________. 7. The _________________________ of an isotope is the sum of the number of protons and neutrons in its nucleus. 8. In Bohr’s model of the atom, ____________________ move in fixed orbits around the nucleus. 9. When an atom gains or loses energy, some of its ____________________ may move between energy levels. 10. The moving blades of an airplane propeller provide an analogy for the electron ____________________ model. 11. When all the electrons in an atom are in orbitals with the lowest possible energy, the atom is in its ____________________ state. 12. An atom in which an electron has moved to a higher energy level is in a(an) ____________________ state. 13. The pattern of repeating properties of elements revealed in the periodic table is known as the _________________________. 14. Phosphorus is one block to the left of sulfur in the periodic table. The atomic number of sulfur is 16. The atomic number of phosphorus is ____________________. 15. The atomic mass unit (amu) is defined as one-twelfth the mass of a(an) ____________________-12 atom. 16. Elements can be classified as metals, nonmetals, and ____________________. 17. Metals that grow dull when exposed to air are more ____________________ than metals that remain shiny. 18. The elements potassium (K), calcium (Ca), and scandium (Sc) appear from left to right in Period 4 of the periodic table. Among these elements, the most reactive is ____________________. 19. From left to right across a period in the periodic table, elements become less ____________________ and more ___________________ in their properties. 20. Element 3, lithium, has one valence electron, and element 4, beryllium, has two valence electrons. Element 5, boron, has ____________________ valence electrons. 21. Hydrogen does not have the typical properties of a metal. However, hydrogen is located above Group 1A because it has one ______________________________. 22. In general, a(an) ____________________ metal will be more reactive than an alkaline earth metal in the same period. 1 23. The two most reactive groups of elements in the periodic table are the alkali metals and the ____________________. 24. Reactive elements, such as alkali metals and halogens, are found in nature only as ____________________. 25. Mendeleev organized elements in his periodic table in order of increasing ____________________. 26. Mendeleev’s periodic table was useful because it enabled scientists to predict properties of unknown ____________________. 27. Although they are called ____________________ lights, they can contain any noble gas. 28. Which of the three subatomic particles—proton, electron, or neutron—has the least mass? 29. If an atom has 32 protons and 38 neutrons, what is its mass number? 30. If an atom of an element has a mass number of 31 and 16 neutrons in its nucleus, what is the atomic number of the element? 31. If an atom of germanium has a mass number of 70 and an atomic number of 32, how many neutrons are in its nucleus? 32. In the nucleus of an atom, there are 15 protons and 16 neutrons. What is the atomic number and mass number of this isotope? 33. How does the state of atoms in a neon light change when light is emitted? 2 Figure 5-1 34. In which segment of Figure 5-1—A or B—will properties of the elements vary the most? 35. Selenium has six valence electrons, while rubidium has one valence electron. Identify each element as a metal or a nonmetal. 36. What determines an element’s chemical properties? 37. On the periodic table, there are two numbers in the block for the element krypton, Kr: 36 and 83.80. What are each of these numbers, and what do they represent? 38. Boron (B), silicon (Si), germanium (Ge), and arsenic (As) are elements that have properties that fall between those of metals and nonmetals. What common label could you apply to these elements? 39. Period 5 of the periodic table begins with element 37 and ends with element 54. Consider the following elements from row 5: strontium (element 38), cadmium (element 48), iodine (element 53). Which of these elements is most likely to be a transition metal? 3 40. Element X has five valence electrons, element Y has one valence electron, and element Z has five valence electrons. Which two of these elements are most likely to have similar properties? Explain your answer. 41. Sodium chloride is a compound of sodium and chlorine. Which of these elements is the alkali metal, and which is the halogen? 42. Most calcium atoms have an atomic number of 20 and a mass number of 40, but some calcium atoms have a mass number of 48. What word could you use to describe these two kinds of calcium atoms? 43. What did Bohr’s model of the atom do that Rutherford’s model did not? 44. What do the whole numbers on the periodic table represent? 45. The elements silicon (atomic number 14) and chlorine (atomic number 17) are both in Period 3. Which is the more reactive element? 46. Sulfur is often found in nature as an element, not combined with other elements in a compound. What does this fact tell you about the reactivity of sulfur? 47. Suppose you are looking at elements in the periodic table in this order: element 23, element 24, element 25, element 26, and so on. Are you looking across a period or down a group? Explain your answer. 48. Among the alkaline earth metals in Group 2A, which is more reactive—beryllium (element 4) or strontium (element 38)? 4 49. Explain what the colors in a fireworks display reveal about the movement of electrons in atoms. Figure 4-2 50. Are the atomic models in Figure 4-2 arranged in the order that they were developed? Explain your answer. 51. Why do you think the proton was discovered before the neutron? (Hint: Consider the properties of protons and neutrons.) Figure 5-2 52. Which of the elements shown in Figure 5-2 are in the same period? 5 53. Which element in Figure 5-2 is a transition metal? Which is a noble gas? 54. Which elements in Figure 5-2 have the same number of valence electrons? How do you know? 55. Based on what you know about elements and the periodic table, compare and contrast the elements beryllium, Be, and iodine, I, which are shown in Figure 5-2. 56. Find the block labeled ? in Figure 5-2. Predict the properties of this element. What is its atomic number? How many valence electrons does it have? Which of the elements shown in Figure 5-2 will it most resemble? Figure 5-3 57. Classify the elements in Figure 5-3 as metals, metalloids, or nonmetals. Explain your answer. 58. Explain why knowing the properties of K, Ca, Sc, and Ti would allow you to predict the properties of Rb, Sr, Y, and Zr. 6 ID: A Chapter 4 & 5 Review Answer Section COMPLETION 1. ANS: atoms PTS: 1 2. ANS: nucleus DIF: L2 OBJ: 4.1.1 PTS: 1 3. ANS: neutrons DIF: L2 OBJ: 4.1.3 PTS: 1 4. ANS: protons DIF: L1 OBJ: 4.2.1 PTS: 1 5. ANS: number DIF: L2 OBJ: 4.2.1 PTS: 1 6. ANS: neutrons DIF: L1 OBJ: 4.2.2 PTS: 1 DIF: 7. ANS: mass number L1 OBJ: 4.2.2 PTS: 1 8. ANS: electrons DIF: L2 OBJ: 4.2.2 PTS: 1 9. ANS: electrons DIF: L1 OBJ: 4.3.1 PTS: 1 10. ANS: cloud DIF: L2 OBJ: 4.3.1 PTS: 1 11. ANS: ground DIF: L1 OBJ: 4.3.2 PTS: 1 12. ANS: excited DIF: L1 OBJ: 4.3.3 PTS: 1 DIF: 13. ANS: periodic law L2 OBJ: 4.3.3 PTS: 1 14. ANS: 15 DIF: L2 OBJ: 5.2.1 DIF: L1 OBJ: 5.2.1 PTS: 1 1 ID: A 15. ANS: carbon PTS: 1 16. ANS: metalloids DIF: L1 OBJ: 5.2.2 PTS: 1 17. ANS: reactive DIF: L1 OBJ: 5.2.3 PTS: 1 18. ANS: potassium K DIF: L2 OBJ: 5.2.3 PTS: 1 DIF: L2 19. ANS: metallic, nonmetallic OBJ: 5.2.4 PTS: 1 20. ANS: three DIF: L1 OBJ: 5.2.4 PTS: 1 DIF: 21. ANS: valence electron L1 OBJ: 5.3.1 PTS: 1 22. ANS: alkali DIF: L2 OBJ: 5.3.1 PTS: 1 23. ANS: halogens DIF: L1 OBJ: 5.3.2 PTS: 1 24. ANS: compounds DIF: L2 OBJ: 5.3.2 PTS: 1 25. ANS: mass DIF: L1 OBJ: 5.3.3 PTS: 1 26. ANS: elements DIF: L1 OBJ: 5.1.1 PTS: 1 27. ANS: neon DIF: L1 OBJ: 5.1.2 DIF: L1 OBJ: 5.3.3 PTS: 1 2 ID: A SHORT ANSWER 28. ANS: electron PTS: 1 29. ANS: 70 DIF: L1 OBJ: 4.2.1 PTS: 1 30. ANS: 15 DIF: L1 OBJ: 4.2.2 PTS: 1 31. ANS: 38 DIF: L2 OBJ: 4.2.2 PTS: 1 DIF: L2 32. ANS: atomic number = 15; mass number = 31 OBJ: 4.2.2 PTS: 1 DIF: L1 OBJ: 4.2.2 33. ANS: The atoms return from an excited state to the ground state. PTS: 1 DIF: L1 OBJ: 4.3.3 34. ANS: Properties will vary the most in segment A, which is a period. PTS: 1 DIF: L2 OBJ: 5.2.4 35. ANS: Selenium is a nonmetal. Rubidium is a metal. PTS: 1 DIF: L2 36. ANS: the number of valence electrons OBJ: 5.3.1 PTS: 1 DIF: L1 OBJ: 5.2.1 37. ANS: The integer, 36, is the atomic number, or number of protons in an atom of krypton. The decimal number, 83.80, is the atomic mass, which is the weighted average of the atomic masses of krypton found in nature. PTS: 1 38. ANS: metalloids PTS: 1 DIF: L1 OBJ: 5.2.2 DIF: L1 OBJ: 5.2.3 3 ID: A 39. ANS: cadmium PTS: 1 DIF: L1 OBJ: 5.2.3 | 5.2.4 40. ANS: Elements X and Z will have similar properties because they have the same number of valence electrons. PTS: 1 DIF: L1 OBJ: 5.3.1 41. ANS: Sodium is the alkali metal, and chlorine is the halogen. PTS: 1 42. ANS: isotope DIF: L1 OBJ: 5.3.3 PTS: 1 DIF: L1 43. ANS: Bohr’s model focused on electrons. OBJ: 4.2.2 PTS: 1 DIF: L2 44. ANS: atomic number number of protons in each element OBJ: 4.3.1 PTS: 1 45. ANS: chlorine OBJ: 5.2.2 DIF: L2 PTS: 1 DIF: L2 OBJ: 5.3.2 46. ANS: Sulfur is not a highly reactive element under ordinary conditions. PTS: 1 DIF: L2 OBJ: 5.3.1 | 5.3.2 47. ANS: across a period because the atomic number is increasing by one each time PTS: 1 48. ANS: strontium PTS: 1 DIF: L1 OBJ: 5.2.1 DIF: L1 OBJ: 5.3.2 4 ID: A ESSAY 49. ANS: When fireworks explode, the heat produced by the explosions causes some electrons in atoms to move to higher energy levels. When the electrons return to lower energy levels, some of the energy is released as visible light. The colors vary because each element has a different set of energy levels. PTS: 1 DIF: L2 OBJ: 4.3.3 OTHER 50. ANS: Yes; they go from the simplest (the solid sphere in 1) to the most complex (the electron cloud diagram in panel 3). Students may specifically cite John Dalton in connection with panel 1 and Niels Bohr in connection with panel 2 to support their answer. PTS: 1 DIF: L1 OBJ: 4.1.2 | 4.3.1 | 4.3.2 51. ANS: The proton has a positive charge, but the neutron has no charge. It was easier to detect the existence of a charged particle because its path could be deflected by a charged plate. PTS: 1 52. ANS: Be, C, N, and F DIF: L2 OBJ: 4.2.1 PTS: 1 53. ANS: V; He DIF: L1 OBJ: 5.2.1 PTS: 1 DIF: L1 OBJ: 5.2.3 54. ANS: F and I; they are in the same group in the periodic table. PTS: 1 DIF: L1 OBJ: 5.3.1 55. ANS: Beryllium is a reactive metal with two valence electrons. Iodine is a highly reactive nonmetal with seven valence electrons. PTS: 1 56. ANS: 12; 2; beryllium DIF: L1 OBJ: 5.2.3 | 5.3.1 | 5.3.2 | 5.3.3 PTS: 1 DIF: L1 OBJ: 5.3.2 57. ANS: These elements are all metals. The elements in Groups 1A and 2A are the alkali metals and alkaline earth metals, respectively. The elements in Groups 3B and 4B are transition metals. PTS: 1 DIF: L2 OBJ: 5.2.3 5 ID: A 58. ANS: When elements are arranged in a periodic table in order of increasing atomic number, the properties of elements repeat from period to period so that elements in the same group have similar properties. PTS: 1 DIF: L2 OBJ: 5.2.1 6