#=W$-x)"
advertisement

= 0.9782849
= 4j76E-O4
= 2.093E-OG
R^2adj
Rnsd
variance
Thereforeorder of reaction = l. 14
Again regressingthe abovedataputting / - 1
POLYMATHReSUIIS
Nonlinear reeression (L-M)
Model: I = k*(PCO1O.14XPH2)
v,?1Y9. . gs? g?glfg?nce
rni suess
variable
0-0075284
0.0040792
0.1
k
Precision
= -0.8194508
R^2
= -0.8194508
R^2adj
= 0.0049354
Itmsd
Variance = 1.754E-04
r4))
Therefore,k = 0.004 (gmolCHy'(gcat.min.atmr'
P5-2 Solution is in the decodingalgorithm given with the modules'
P5-3 Individualizedsolution
P5-4(a)
The kinetics of this deoxygenationof hemoglobinin blood was studiedwith the aid of a tubular reactor.
HbO2 ) IIb+Oz
-rrkCi
Ratelaw:
dY
Foo? = kCi,o(l- X )"
CIY
611 - A"dZ , where As is the tube crosssectionalarea
Mole balance:
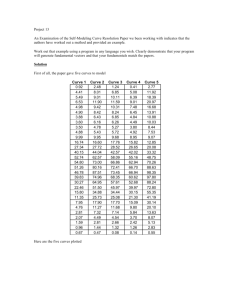
#=W$-x)"
^(#)=tna*ntn(l-x)
therefore,
where,a=ry
Position (cm)
8z (cm)
si
f _r, la: l
: l tq: l ri:f -'e:
l q---:T
l
-i
s f*J : f i , T l--t
f 5
of Hbo2 (XJ [ 0.0000 | o.otel
lConversion
W
AX
A>VAz (cmt)
fo'o:sf; f or5ilf
oo?48
0 .1 1 0
:l o.eaoz
I r.oooo
I o.eors
f*_--:foo13
oom?sTo"oo3?f
5-3
0.8900
0.0175
0.00354
Electode Position
I
617
010386| obmi8 f 9ro36s-l0103o::lmm5q
qqo:s:f 0003+s:
' 5,1
-l
LXJLz vs- z is then produced. The values
d)Udz (crrr)
A histogram plot of
of d)Vdz are evaluated using equal-arei
0.fi)395
0.0039
o-00385
0.0038
Co.mszs
€ o.oorr
$o-mo6s
$ om:o
o-(xxi55
o-0035
olxtSls
0.00340
25 5 ?.5 r0 t2.s ,, t
25 27.530 32s 3s 375 40
i tt
ij"
Ihing the values obtained above, a plot of ln(dXa/dz) vs. ln(l-Xf
irfit to the data
is produced and a line
ln(dXldz)vs. ln(l.X)
^(#)=tna+ntn(r-x)
where,a=ry
y = 1.0577x5.5477
.(#)
= -5.5477+
1.0577
rn(r- x )
I
dp(-5.5) = 4.1 x lO'3cm.
dration of blood is l50g hemoglobin per liter of blood
weight of hemoglobin = 645@
:23x10{
mol I cm3'
=45.7xlAamolesl s
F*a =
(h4
41.7xloa molesI s
=
- (\ .'.rxro-, -"'
cm)
I 4.rs-,
Z3xlAamoles I cm3xO.OI96cm2
r & law i s ,
-4-rce+
clm-.s
5-4
Ps-4(b)
First we fit a polynomial to the data.Using Polymathwe useregressionto find an expressionfor X(z)
SeePolymathprogramP5-4-b.PoI.
POLYIVIATH Results
Polvnomial Reeression Renort
lfodel: X = a0 + a1*z+ a2"[2+ a3*z^3+ a4"/4 + a5"z^5+ a6*za6
Variable
;d-a1
a2
a3
a4
a5
a5
Value
z:sLBr.-LA
-95t o-confidence
O
0
0
0
0
0
* 0.0040267
-6-14E-05
7 .7678-06
-5.08-07
1.4678-08
-1.68-10
General
Orderof polynomial= 6
Regressionincludingfree parameter
Numberof observations= 7
Statistics
R^2=
Rr2adj =
Rmsd=
Variance=
1
0
1.669E-10
1.0E+99
Next we differentiate our expressionof X(z) tofind d)vdz and knowing that
( ax\
l n l? l =l na +nl n(t-X )
\d z )
where.a=kCX'4
'
Foo
asa tunctionof ln (1- X ) give.s6 similarvaulesof slopeandintercept
Linearregression
f+l
" " \dz )
as in the finite differences.
POLYMATH Results
Linear Resression ReDort
Model: ln(dxdz)= a0 + al.ln(l-X)
Variable
eda1
Value
-5 .5 3 L 9 4 7
1 .2 8 2 4 2 7 9
95t confidence
----d:arEm-
o.3446L87
General
Regressionincludingfree parameter
Numberof observatiofls= 7
Statistics
R^2 =
R^2adi=
Rmsd=
Variancc =
n -11.28
0.9482059
0.9378471
0.0044015
1.899E-04
5-5
lna=-5.53,a=0.00396
F ooQ
p-
CXo4
-
45.7xl|amolesls
2.3xloamoles I cm3x}.Olg6cmz
.96xl}-3 cm) = 4.0s-'
Hence rate law is,
-fA=4.0cI',8
+
Am-.5
P5-5 (a)
[&uid phaseirreversible reaction:
A ) B + C;Qro=2mole/dm3
Coo-Co
=kC?
TA
n(c* :co )=,n
\r)
k +atnc o
fue time(T )min. Ca(moUdm')
15
1.5
3E
1.25
1.0
lm
gn
4.75
mm
0.5
ln(CA)
0.40546511
0.223t4355
0
-0.28768207
-0.693147r8
ln((CAo-CA/z)
-3.40tt974
-3.9252682
-4.605t702
-5.4806389
-6.6846117
By Lsing linear regression in polymath:
e Polymath program P5-5-a.pol.
In({Cao-Caoftau} vs In{Ca}
!!0LI/MATH 4esults
I"hear ReeressionReport
bd:Y=a0+al*lnQa
-1
-t-a \
h f -a o
l =mr +al nc o
\e )
ln
n
@.riable
Value
eL
C*{CS
lffifi? =
mi2adl iMGd=
2.9 99 8L 5t
95t
confidence
0. 041 1_145
=
.'
-2
t
o
y= 2.9998x-4.6081
IE
?o
(E
u
-4
0.9999443
0.9999258
0.003883
'rhiryrce =
1.256E-04
tN=slope=3
= intercept = -4.6
k = 0.01 mole-2min-I.
lrw:
-3
dc^ ^ ^- _1
= O.ylC3emolI dm3 min
-+
dt
(b) naintoualizedsolution
(c) lnaiuiaoahzedsolution
5-6
P5-8 (d) fn" rate constantwould increasewith an increasein temperature. This would result in the
pressureincreasingfaster and less time would be needto reachthe end of the reaction. The opposite is true
fr o colder temperahrres.
P5-9
Photochemicaldecayof bromine in bright sunlight:
to
t (min)
ce(ppm) 1.4
5
20
1.7
4
30
1.2
3
40
0.8
I
50
0.6
2
60
o.4
4
P5-9 (a)
Mole balance:constantV
dcn dt
r^ =-kc?
dt)
\
^(-+)=h(k)
Differentiation
T (min)
At(min)
C,c,(ppm)
ACa @pm)
/
l^^
l "o l P
Pm \
Al \mi n /
\/
+qtn(cA )
l0
2.45
|
30
20
10
r.23
t.74
50
10
0.88
60
10
o.62
o.44
-0.71
-0.51
-0.35
-o.26
-o.18
_ o .0 zl
-0.051
-0.035
-o.u26
-0.018
o.oa
rcr
tt
40
10
10
o.o.
5-ll
and
afea
dc^/dt
0.o82
0.061
H{Coldt)
-2.50r
0.896
0.554
0.030
-3.507
0.014
hC
o.M2
-3.r70
0-02r5
-2.797
o.207
-3.840
-0.r28
-4.269
-0.478
-o.82r
Usinglinearregression:s = 1.0
h k = -3.3364
t = 0.0344min-t
H-e (b)
il^
-vr^=F^
dAD
rt = 4.O344ry: = 4.0344 :m8: atCa= I pp,,,
rrun
/ min
*+ftr )[-+4 =(2s000
sotl(o.ozaa
3)(
/min/\
\
n$9
(-lt- \ =o.oru
] t'++)
" .-" !!!
rgat
/(1000*8)l
(c) t"atuio*tized solution
H5-10(a)
phase decomposition
'
A )B+ 2 C
kmine
the reaction order and specific reaction rate for the reaction
frr
dcn
-_kc tr^'
-tuerheraterawas:
dt
bacraling;
(-l-_
k(n - t) [ Co'-t
I
')
C oo"-,)
5-12
/(+s:^os)-
hr
608
gmol
* (o.ooosm3)
'
23*rc6
(too)'"'
+
. m*t ')o'ttt
*n-'
k =3.224*rcu(
\gmol )
Enalconcentration : L?E(O.Z;
"t*
= 0.107
weight fraction = 1O.77o
weightfraction= 2OVo
hitial concentrati6nof IIW = 0.2 (given)
N'
Mole balancefor fIF:
-
dt
- 6dN t
dt
d* -an(
Pv
\ v
-P
r00MW, dt
\l0O MWF)
=r.775
wherec
--j#=*(ffi)""''!0,
r ( r )l'o'=6(t'zz+
*10-' , 1o1lo'"',
)[ a . roo.,l
t rt-,
*lw" )^
\
t(t
o.77s\10.70"'ri* )=2'38e*roat
t = 331 min
P 5-14(a)
A+38 )
C+2D+E
Observationtable for differential reactor:
Temperature(K)
Conc. Of
A(moUdm3)
Conc. Of
B(moVdm3)
Conc. Of
C(moVdm3)
Rate
(moUdm3.min)
323
333
0.10
0.10
o.oo2
0.00r
0.10
0.05
0.10
o.20
0.01
o.l0
0.006
0.003
0.10
0.008
o.oo4
0.0s
o.a2
o.ol
0.01
0.01
0.02
0.01
0.01
v3
353
%3
%3
Space time for differential reactor = 2 min
v=L -'oC'
-rP
-rP
5-18
0.005
, --cP -cq'n
'P
2
t
Rate law:
fc= Af-u'')coctr
9-il-Btr\C;cB
+
Where, A is Arrhenius constant
B = activation energY/R
x is the order of reaction wrt A
Y is order of reaction wrt B
Ceis the concentrationof CzILBT
Cs is the concentrationof KI
for finding the approximatevalue of B because'at
Now using datafor tempefature 323K,333K, and 363K,
=
same. Using polymath, the rough value of B
thesetemperature,the cincen6ation of A and B are the
s500K
the initial valuesof n' m' and A ' we
while using polymath for solving the rate law apartfromguessing
So after trial and error we got B = 6500K
changethe value of B in the moJel to get the opi-u* rolirtioo.
SeePplymathprogramP5-l4-a'pol'
POLYMATH Results
Nonlinear regressio+!L$4)
Modek r = A'exp(-6500/T)-Canx'Cb^y
Ini gnress
3 - 6E+05
o .2 5
0 .2
Vari.able
A
x
v
Precision
R^ 2
R^2adj
Rmsd
Variance
=
=
=
=
Value
5.TAeEm6
0 .250855s
0.2963283
958 confidence
2 . 9 2 8 8 +0 4
0.0032606
0.0020764
0.9323139
0.8871898
3.615E-04
1.568E-06
Hence,by nonlinear regressionusing polymath
6(
A = 3.64b8+o6(moleldm3)-2't /s)
E = 6500R= 54.015KJ/mol
x=O.25
Y= 0-30
hence,
nr)go'zsgf;'s mole/dm3-min
rc = 3.64E + 06e(-s40rst
P 5-14 (b) naitioualizedsolution
P5-15(a)
5-19
ililiiiiililit:ilill:ililiilri',;i,r,,!,,,,:rrti,i,r,;.::.,.,',..,,,,
Model: rg = umax*Cc/(1+k.Cs^(-y))
Ini guess
0 .3
1 .6
L
Variable
lxnax
k
v
Precision
R^2
= 0.9999447
R^2adj
Rmsd
Variance
= 0.999917
= 0.0038269
= 1.794E-O4
Value
0 .3 2 656L4
L 6 2 .599
2 -0892232
34.273983
0.0461489
0.33C.
s/d-''h
"=;ffirt-^
P5-16
Thermal decompositionof isopropyl isocynatein a differential reactor.
Run
I
2
3
4
5
6
Rate
Concentration Temperahrre
(moVs-dm3) (moUdm3)
-(K)
700
4.9 x 10o.2
750
1.1x 10o.u
2.4xlA"
0.05
800
2.2xlO'
0.08
850
1.18x lO'
0.1
900
1.82 x 10-'
0.06
950
Ratelaw:
-rA-
/rrtzrnr)gn
Where, A is Anhenius constant
E is the activation energy
n is the order of reaction
Cais the concentation of isopropyl isocynate
See Polymath program P5-l6.pol.
POLYMATH Results
Nonlinear reeression (L-M)
Model:rA = A*exp(-El(8.314.T)).(CA)^n
Variable
A
E
n
Ini
gruess
100
1 -0 0 0
1
Value
1 .0 1 8+ 04
5 .8 0 5E + 04
1 .7 3 05416
958 confidence
327.35758
237 .32096
0.0134196
Nonlinearregressionsettings
Max#iteratiofls=&[
Precision
P.n2
R^2adj
Rmsd
= 0.6690419
= O.M84O32
= 0.0097848
5-21
= 0.0011489
variance
Hence, by nonlinear regression using polymath
A = l0l0o (mole/dm3)'2'6(t/s)
E = 580@J/mol
n= 1. 7
therefore,
- rA-r ol ooexpI
a'gJ6)c';7
\ r )^
mole/dm3.s
+
CDPs.A
Given tlre reaction P + NH'$H
--+ NH2OI{P
wbcre P is Penicillia and i-filOHPis hydroxylamine acid (&noted by subscript fIA)
I*t A * "Absorbcncy, thsn Cn^ = KA where K is some csnsratt"
C' = Cr"{l * X} {" = g for liquid phase rtaction.}
Crrrr*CoX*l{A
vno
At t =*,
A - =C=F
KA.
X =*$*
* rr
Assume raaction is irr*versible:
kCl = *CL{l * X}"
I dl'I'
dc'
-
t"
v-dr**;i*
dC"- ^
*F=%at
=-kC"r.tt-A/A-)"
+=-+$=-rc;(l-x)"
A-dr
For a batcL canstalrt volumc reactoror
(r
dA
.
--:31 .ltc;:r \'-t (e_-e)' =K{e--A}",
^\E whercK*kl:3e.1\*r
|
dt
'
LA_/
\A_/
Try integral anelysis firsl Assuroetharreactiou is zero ordef :
or
ft"o
or Jtn=A=KJ
aptotof Avs'tsbourdbelinearif
$*x
rsaction is zern order" From tbE plotbctow, it is evident thar the reaction
is nog zerc srder:
0. 6
t
0. 5
I
.
E o.+
F
€ o.g
o
a
{1,
* *-a
o-l
o
0 toe 0 3040so
* W&*38
At cs=z.lmmoresr
-" ta{$}L
c s bs =2.1 = ^{-"}.
24-r
ac 5
= -ogfr?:dn't
= -0.??5>-r:
stabic
. = t-ry4 min-r}{3r3min}
{+PL
'
\ a c sfts
irr Cs = i.l m moics,{. -rs = 0.154
t-.fflil
"rfl-"t1is
\dcs /.
y = Cto -Ct
Cro
appcarsro besnbtc-butmorc accuraccalu:uladon
nccessary
rhisconctusion
io csrablish
definilively.
-50-2'I
=.958
50
W-t2 (c\
-fs =
kcsEr
l+ KrC,+ K2C3
lf E r is reducedby 33%, -r5will alsodecreaseby 33Vo.From the original plot, we seethat if the curve-r5 is
decreasedby 33Vo,the staight line from the CSTR calculation will crossthe curve only once at
approximatelYCs = 40 mmol/L
X=0.2
W-12
W-12
(d) naiuioualizedsolution
(e) naiuiauized solution
P7-13(a)
Daraon B:rkels Ycasrat ?3.4
p^
-
iJ?
qa
no
sulfa$iiarnidc
0. 0
0_5
0.0
23.5
oC
ae
I
I
6
30 rrg
sutfunilcmidc/ml
no
adc€dto rqcCiurn sulfanilamidc
().0
17.4.
?5-5
30.R
36.4
39.5
40.0
.L
Por
*J*
aq
?0 rng
sulfanilamidc/rnl
addcdumedium
.u5 /f
:,.0
.f)425
.039I
r-0
.0303
.03245
.55
.02566
l.-<
3?.5
?{
.40
42.0
.f)?38
-o2747
.0?s3
43.0
.285
.r)?33
3.5
.0?,50
.?00
4
3
.
t
)
,0?,33
5" 0
vll$-+ wherein ihisproblem.
K E' S
rp=RITT=R,.,.,*s
.
Qot * oxygenuprakein microlirirsof
r.0
JJ.U
& per hour per mg CIfcells = rp, arld O1 is the subskate"
-.**#- - *- *{ # " * - *
Plot oi6[
I
versus
*f
*
tlJtl ln
(C(stLC(g)l
rvill havea slope
"t*
o.06
+- !$ftn
s"s
"€-
andan inierceprof iln
t'tltlrout
or}*
ofiI
0.g4
0-ol
050
s.s
rs
l/p{o}{nmrc}
05
rs
- 'ill4 = .S165 Intcrcryt = 0.019
From rhe graph,slope = '{}3CI5
I
-'. V** = 52-63*-nt-A-hr mgcclls
* 0,8684nrmllg
Ks = 0"0165Vrns,x* {S.il1$5x5?"63i
F/-13 (b)
l.&ow,wirhcompcddveinhibitisn:
E+ g cc f, " g
t + t s 'C : c t r H
E.S*rrP+5
Rare law becornss: rp *
.
v**1,
S +K *[r*]l
I
In *ris case,tireslopeis r
K* {r++ }
*f,;;*!l
^' Ir P
rc-fr *!|
V..*
ll_l o I
tSI'V*.*
Kl,
while the i*rerceptis rhesarneasin case{a}
7-31
For the caseoi uncornpetiriveinlribition:
E + 5 cc E ' 5
E.S+lc=f'€.$
g.$g+P+E
R*rc law bqccrncs:
sJs!
rp =
K|n
s{t
In ttriscasp,thc slopt is thqsarne,but thc intercept,,
#i
E + | c* E ' I'
E.I+SeI.E*5
inhibidpn:
And for thc cascof non-cornpeddve
E + S e = rE . S
l-E . $ c * E . g * I
g'gc*P+E
f,P=
V-rr
r*#
S
ts+K*l{r+fr}
r+J-
o'*=o#.*{+)
In rhiscascborhrheslopcandinrerccprchangc.Ploningthcdataot*
in mmHg u**
q[
wittr sulfanilamidspn drc :ameplor ls wasplotted the dam lor thc cascwith no sulf,asil*'rddc,ir is
sccn rharrheslapcsarudiffcrcnt, but rhc intcrccpris thc same.Thcr*fuc thc inhibition is
comParadve.
W -\3 (c) nainiaouhzedsolution
F'/-13 (d) l"aiviooutizedsolution
w-L4
For No Inhibition, *ttt*-t"O"it,oo,
Equationmodel:
.( f )
^
="0+"1[SJ
;
a0 = 0.008
aI = O.O266
For Maltose,
F,quationmodel: = r."{*)
+
a0 = 0.0098
al = 0.33
For o-dextran,
7-32