Ferrofluids (3)
advertisement

Ferrofluids
Overview
• Definitions
• Engineering
• Applications
Definition
• A ferrofluid is a specific type of liquid
which responds to a magnetic field.
Ferrofluids are composed of nanoscale
magnetic particles suspended in a carrier
fluid. The solid particles are generally
stabilized with an attached surfactant
layer. Ferrofluids are extremely stable
meaning that they will not cluster together
even in extremely strong magnetic fields
Ferrofluids: Magnetic Liquids
Liquid That Responds to a
Magnetic Field
=
Colloidal Suspension of
Superparamagnetic Magnetic
Material
History of Ferrofliuds
• In the 1960’s Stephen
Pappell at NASA first
developed ferrofluids as a
method for controlling
fluids in space.
• Magnets and/or magnetic
fields were used to
control this magnetic
fluid.
• Currently applications of
Ferrofluids in space have
been replaced by more
economical fluids.
Physics
• Ferromagnetismisamagneticdipolethatisfro
mthealignmentofunpairedelectronspinsinel
ementssuchasiron,cobalt,andnickel.Inthise
xperimentwewillsynthesizemagneticnanop
articlesfromironchloridesandthendispersei
ntoatetramethylammoniumhydroxidesurfac
tanttoformacolloidalsuspension
How Does A Magnetic Liquid Work?
2FeCl3 + FeCl2 + 8NH3 + 4H2O →
Fe3O4 + 8NH4Cl
Tetramethylammonium
Cation
(NH4+)
Electrostatic
Repulsion
Hydroxide
Anion
(OH-)
~ 10nm
Berger, P.; Adelman, N. B.; Beckman, K. J.; Campbell, D. J.; Ellis, A. B.; Lisensky, G. C. Journal of Chemical
Education 1999, 76, 943-8.
Chemistry
• The formation of ferrofluid involves various types
of forces that hold the components together. For
example, magnetite is held together by ionic
interactions. Ionic attractions between hydroxide
anions and tetramethylammonium cations allow
colloidal suspension of the magnetite in the
solution. Without the tetramethyl ammonium
hydroxide as a surfactant, the magnetite
nanoparticles tend to cluster together. Therefore
it is necessary to have the appropriate surfactant
to stabilize an aqueous ferrofluid
Synthesis of Magnetite Nanocrystals
FeCl3 + 3NH4OH → FeO(OH) + 3NH4Cl + H2O
FeCl2 + 2NH4OH → Fe(OH)2 + 2NH4Cl
2FeO(OH) + Fe(OH)2 → Fe3O4 + 2H2O
Processes:
1) Nucleation
2) Growth
3) Termination
+
→
+
+
+
→
• Fe(III) coordinates to 6 water molecules and
Fe(II) coordinates to 4 water molecules (not
shown) until the solid forms
• The water molecules on the periphery of the
magnetite are ultimately replaced by
tetramethylammonium hydroxide
+
+
+
+
+
+
+
+
+
+
+
+
+
+
+
Unique Properties
• Stick to Magnets
• Take on 3-Dimensional Shape of a
Magnetic Field
• Change Density in Proportion to
Magnetic Field Strength
Ferrofluid Magnetic Properties
Water-based Ferrofluid
µ0Ms = 203 Gauss
φ = 0.036 ; χ0 = 0.65, ρ=1.22 g/cc, η≈7 cp
dmin≈5.5 nm, dmax≈11.9 nm
τB=2-10 µs, τN=5 ns-20 ms
Isopar-M Ferrofluid
µ0Ms = 444 Gauss
φ = 0.079 ; χ0 = 2.18, ρ=1.18 g/cc, η≈11 cp
dmin≈7.7 nm, dmax≈13.8 nm
τB=7-20 µs, τN=100 ns-200 s
13
Langevin Equation
[
M
1
= cothα − ]
Ms
α
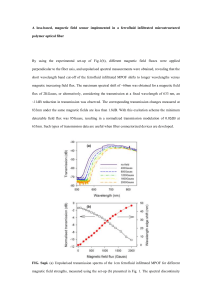
Measured magnetization (dots) for four ferrofluids containing magnetite particles (Md = 4.46x105
Ampere/meter or equivalently µoMd = 0.56 Tesla) plotted with the theoretical Langevin curve (solid line).
The data consist of Ferrotec Corporation ferrofluids: NF 1634 Isopar M at 25.4o C, 50.2o C, and 100.4o C
all with fitted particle size of 11 nm; MSG W11 water-based at 26.3o C and 50.2o C with fitted particle size
of 8 nm; NBF 1677 fluorocarbon-based at 50.2o C with fitted particle size of 13 nm; and EFH1 (positive α
only) at 27o C with fitted particle size of 11 nm. All data falls on or near the universal Langevin curve
indicating superparamagnetic behavior.
14
Applications
• Inks
• money
• Biomedical
• attach drugs to magnetic particles,
proposed artificial heart
• Damping
• speakers, graphic plotters, instrument
gauges
• Seals
• gas lasers, motors, blowers, hard drives
Berger, P.; Adelman, N. B.; Beckman, K. J.; Campbell, D. J.; Ellis, A. B.; Lisensky, G. C. Journal of Chemical
Education 1999, 76, 943-8.
Damping: Speakers
Rosensweig, R. E. Scientific American 1982, 247, 136-45.
See how a speaker works at:
http://electronics.howstuffworks.com/speaker6.htm
Damping: Rotating Shafts
Cross-sectional view of a ferrofluid
viscous inertia damper
Energy band gap apparatus
Ray, K.; Moskowitz, B.; Casciari, R. Journal of Magnetism and Magnetic Materials 1995, 149, 174-180.
Seals
Atmosphere
Vacuum
Rotating
Shaft
Magnetically
Permeable
Material
Ferrofluid
Permanent Magnet
Rosensweig, R. E. Scientific American 1982, 247, 136-45.
Ferrofluid Preparation
•
•
•
•
•
•
Step 1
Step 2
Step 3
Step 4
Step 5
Step 6
Ferrofluids Step 1
Dissolve 67.58g FeCl3.6H2O
in 250ml of 2M HCl.
Dissolve 39.76g FeCl2.4H2O
in 100ml of 2M HCl.
Modified from Berger et al, Journal of Chemical Education, 1999, 26, 7, 943-948
Ferrofluids Step 2
1M FeCl3 should be used
within one week of
preparation.
2M FeCl2 should be used
within one week of
preparation.
Ferrofluids Step 3
Combine 3ml of FeCl2
solution and 12ml of FeCl3
solution and fill a burette with
150ml of 0.7M ammonium
hydroxide solution.
Add ammonia very slowly
whilst stirring. A black
precipitate of magnetite will
form.
Ferrofluids Step 4
After addition is complete, stop
stirring and use a strong magnet
(Nd2Fe12B) to settle the black
precipitate to the bottom of the flask.
Decant off the water and add fresh
water. Rinse the precipitate and again
decant off the water. Repeat three
times to remove excess ammonia.
Ferrofluids Step 5
Transfer the viscous liquid to a
weighing boat using a little extra
water if necessary. Use a magnet
on the base of the weighing boat to
remove excess water.
Add 24ml of tetramethylammonium
hydroxide (25% solution) and stir
with a glass rod
Ferrofluids Step 6
Hold a magnet on the base of
the weighing boat and let the
solid settle to the bottom.
Decant off any excess liquid to
leave a very viscous black liquid.
The viscous liquid should form
spikes if a magnet is held
underneath the weighing boat.
You may need to adjust the
amount of water.
INGAS
• In20.5Ga67Sn12.5
• In25Ga62Sn13
• In21.5Ga68.5Sn10 – Galinstan® (Geratherm
Medical AG)
Melting: -19÷
÷10°С
Boiling: >1300 °С
Ferrofluid with metallic matrix
Mechanical Applications
• Ferrofluids are used in many ways
mechanically. They are used in
applications such as gaslasers, motors,
and blowers. In some of these applications
the ferrofluid is held in place by a strong
magnet and separate by two different
pressured chambers.They are also used
as substances for vibrational dampening in
electronic applications
Ferrohydrodynam
ic
Instabilities In
DC
Magnetic Fields
31
Labyrinthine Instability in Magnetic
Fluids
Magnetic fluid in a thin layer
with uniform magnetic field
applied tangential to thin
dimension.
Stages in magnetic fluid labyrinthine
patterns in a vertical cell, 75 mm on a side
with 1 mm gap, with magnetic field ramped
from zero to 535 Gauss. [R.E. Rosensweig,
Magnetic Fluids, Scientific American,
1982, pp. 136-145,194]
32
Rotating Magnetic Fields
Uniform
Non-uniform
bg
vθ (r )
vθ r
y
RO
Surface Current Distribution
{
K z = Re Ke
j ( Ω f t −θ )
}
x
y
bg
ωz r
RO
z
Ferrofluid
z
x
ω z (r)
Surface Current Distribution
Ferrofluid
η,ζ , η'
η,ζ ,η'
µ →∞
a. One pole pair stator
{
K z = Re Ke
µ →∞
j ( Ω f t − 2θ )
b. Two pole pair stator
Observed magnetic field distribution in the 3 phase AC stator
33
}
Ferrofluid
Drops in
Rotating
Magnetic
Fields
A Gallery of
Instabilities
Ferrohydrodynamic
Drops
34
Ferrofluid Spiral / Phase
Transformations
35
4. Dielectric Analog: Von Quincke’s Rotor
(Electrorotation)
Von Quincke’s
Rotor
(a) Von Quincke’s rotor consists of a highly insulating cylinder that is free to rotate and that is placed in
slightly conducting oil between parallel plate electrodes. As DC high voltage is raised, at a critical voltage
the cylinder spontaneously rotates in either direction; (b) The motion occurs because the insulating rotor
charges like a capacitor with positive surface charge near the positive electrode and negative surface
charge near the negative electrode. Any slight rotation of the cylinder in either direction results in an
electrical torque in the same direction as the initial displacement.
36
More on Quincke’s Rotor (Electrorotation)
Definition of Quincke Rotation: Spontaneous rotation of insulating particles (or
cylinders) suspended in a slightly conducting liquid subjected to a DC electric field
with the field strength exceeding some critical value (Jones, 1984, 1995)
Ω >0
when
ε 2 ε1
>
σ 2 σ1
where
εi
τi =
σi
is the charge relaxation time
in each region
Two Competing Forces (Torques):
The electrical torque and the fluid viscous torque exerted on the particle
37
Torques Exerted on a Micro-particle
The fluid viscous torque
The electric torque
Tv = −8πη0 R 3 Ω
Te = p × E =
Re ≪ 1
6πε1 R 3 E02 (1 − τ 1 τ 2 ) Ωτ MW
2
(1 + 2ε1 ε 2 )(1 + σ 2 2σ 1 ) (1 + Ω2τ MW
)
For a small perturbation of rotation to grow, the equation of angular motion for the
particle is re-written as (Jones, 1995):
6πε1 R 3 E02 (1 − τ 1 τ 2 )τ MW
dΩ
3
=
− 8πη0 R Ω
I
2 2
dt (1 + 2ε1 ε 2 )(1 + σ 2 2σ 1 ) (1 + Ω τ MW )
The bracket term should have a value larger than zero for the small perturbation to
grow (Jones, 1995), thus
τ 2 > τ1
38
Competition of the Viscous and Electric Torques
2
8η0σ 1
σ
Ecrit = 1 + 2
2σ 1 3ε1σ 2 (τ 2 − τ 1 )
Ωτ MW
E0
=±
−1
Ecrit
Steady
Maxwell-Wagner
Relaxation Time
τ MW
2ε1 + ε 2
=
2σ 1 + σ 2
Te @ 0.5Ecrit
E0 > Ecrit
Tvis
Te @ Ecrit
Te @ 2 Ecrit
39
5. Flow Rate Enhancement using Electrorotation
Experiments have shown that for a given pressure gradient, the Poiseuille flow rate can be
increased (Lemaire et al., 2006) by introducing micro-particle electrorotation into the fluid
flow via the application of an external direct current (DC) electric field.
E≠0
E
E=0
From Hsin-Fu Huang PhD Thesis research, supervised by M. Zahn
40
6. Continuum Analysis for Couette & Poiseuille
Flows with Internal Micro-particle Electrorotation
The Couette flow geometry
U0
γ∗
= ηeff
= i ⋅ T ⋅ iy
Stress balance τ s = ηeff
τ MW z
h
The Poiseuille flow geometry
Q = ∫ u y ( z ) dz
h
2D volume flow rate
0
41
The Continuum Governing Equations
EQS & Electro-neutrality
(Haus & Melcher, 1989)
Polarization Relaxation
Equilibrium Polarization
Continuity
∇× E ≈ 0
∇⋅D ≈ 0
DP ∂ P
1
=
+ v ⋅∇ P = ω × P −
P − Peq
Dt
∂t
τ MW
(
)
(
)
(
Peq = Peqy ( n, E0 , ε i , σ i ) iy + Peqz ( n, E0 , ε i , σ i ) iz
n
∇⋅v = 0
)
τ MW =
2ε1 + ε 2
2σ 1 + σ 2
Particle # density
Incompressible flow: treating as a single phase continuum
Linear Momentum
(Dahler & Scriven, 1961,
1963; Condiff & Dahler,
1964; Rosensweig, 1997)
Angular Momentum
(Dahler & Scriven, 1961,
1963; Condiff & Dahler,
1964; Rosensweig, 1997)
(
)
(
)
Dv
= −∇p + Pt ⋅∇ E + 2ζ∇ × ω + β ∇ ∇ ⋅ v + ηe∇ 2 v
Dt
2
No-slip boundary conditions ζ ∼ 1.5φη0 ηe = ζ + η η ' ∼ h η
η ∼ η0 (1 + 2.5φ ) Zaitsev & Shliomis, (1969);
ρ
ρI
Rosensweig, (1997)
Dω
= Pt × E + 2ζ ∇ × v − 2ω + β ' ∇ ∇ ⋅ ω + η ' ∇ 2 ω
Dt
(
)
(
)
Field conditions: free-to-spin, symmetry, stable rotation
Lobry & Lemaire, 1999; He, 2006; Lemaire et al., 2008
Spin field BCs:
ω=
β
2
∇×v
0 ≤ β ≤1
Kaloni, 1992; Lukaszewicz, 1999; Rinaldi, 2002; Rinaldi & Zahn, 2002
42
Polarization Relaxation & Equilibrium Polarization
z
r
Electric potential and field solutions to a spherical particle subjected to a uniform DC electric field
R r
rotating at an angular velocity of Ω .
Φ ( r ,θ , φ ) =
∇ Φ=0
2
( )Θ θ Ψ φ
( ) ( )
r
(
φ
Ω
K f = σ f V = σ f (Ω ix × Rir ) = −σ f ΩR(sin φ iθ + cos θ cos φ iφ )
r → ∞, E → E0 iz = E0 cos θ ir − sin θ iθ
Φ ( R − , θ , φ ) = Φ ( R + ,θ , φ )
n ⋅ J f + ρ f v + ∇Σ ⋅ K f = −
)
∂ρ f
θ
R
y
Laplace’s equation with spherical harmonics
BCs (Cebers, 1980; Melcher, 1981; Pannacci, (Jackson, 1999)
2006)
,
ε1 , σ 1
ε2 , σ2
x
n ⋅ J f = σ 1 Er ( R + , θ , φ ) − σ 2 Er ( R − ,θ , φ )
σ f = ε1 Er ( R + ,θ , φ ) − ε 2 Er ( R − , θ , φ )
∂t
E † = E0 iz
,
The proposed “rotating coffee cup model” for the retarding polarization relaxation equations with its
accompanying (quasi-static) equilibrium retarding polarization (Huang, 2010; Huang, Zahn, & Lemaire, 2010a, b):
DP ∂ P
1
P − Peq
=
+ v ⋅∇ P = ω × P −
Dt
∂t
τ MW
(
)
(
)
(
σ 2 − σ1
ε 2 − ε1
−
2ε1 + ε 2
z
3 2σ 1 + σ 2
Peq = 4πε1 R n
E0
2
1 + τ MW
Ω 2x
σ 2 − σ1
ε −ε
− 2 1
2σ 1 + σ 2 2ε1 + ε 2 E
0
2
Ω 2x
1 + τ MW
τ MW Ω x
Peqy = −4πε1 R 3 n
)
Retaining macroscopic fluid spin
Including microscopic particle rotation
Ω
± 1
Ω = τ MW
σ
Ec = 1 + 2
2σ 1
2
E0
− 1,
Ec
0,
8η0σ 1
3ε1σ 2 (τ 2 − τ 1 )
E0 ≥ Ec
ωx
E0 < Ec
E0
43
Modeling Results for the Poiseuille Flow Geometry
Schematic diagram for the Poiseuille geometry
Q = ∫ u y ( z ) dz
h
2D volume flow rate
0
44
Comparison of Poiseuille Velocity Profile Results
Zero spin viscosity Poiseuille flow velocity profiles compared with experimental results found from the
literature (Peters et al., 2010) Lemaire experimental results are from Fig. 9 of Peters et al., J. Rheol., pp.311, (2010)
σ 1 = 5.4 ×10−8 S m Ec ≈ 1.8 kV mm
η'=0
φ = 0.05
Cusp structure for zero spin viscosities
∆p
Pa
≈ 5974.6
L
m
The zero spin viscosity
solutions of our present
continuum mechanical field
equations over predicts the
value of the spin velocity
profile and has a cusp in the
mid-channel position, which is
not consistent with
experimental measurements
done by Peters et al. (2010).
However, the order of
magnitude is correct.
Huang, (2010)
Zero electric field solution of
Poiseuille parabolic profile
45
Comparison of Poiseuille Flow Rate Results
Finite spin viscosity small spin velocity Poiseuille flow rate results compared with experimental/theoretical
results found from the literature (Lemaire et al., 2006) Lemaire theory/experimental results are from Figs. 5 and 6 of
Lemaire et al., J. Electrostat., pp. 586, (2006)
HT: Huang theory (solid line)
LT: Lemaire theory (dashed line)
LE: Lemaire experiment (dotted line)
σ 1 = 4 ×10−8 S m
Ec ≈ 1.3 kV mm
φ = 0.05
φ = 0.1
Finite spin viscosity results do not
involve ad hoc fitting!
β =1
η ' ≈ h 2η ≈ h 2η0 (1 + 2.5φ )
46
Huang, (2010)
Comparison of Poiseuille Velocity Profile Results
Finite spin viscosity small spin velocity Poiseuille flow velocity profiles compared with experimental
results found from the literature (Peters et al., 2010)
Lemaire experimental results are from Fig. 9 of
σ 1 = 5.4 ×10 S m β = 1 η ' ≈ 0.012h η ≈ 2.96 ×10
−8
2
−10
N ⋅s
Best fit, within spin viscosity values calculated
by He (2006) and Elborai (2006) for ferrofluids
Ec = 1.8483 ×106 V m ≈ 1.8 kV mm
Ec = 1.8 kV mm
round and substitute to
analytic solution
Peters et al., J. Rheol., pp.311, (2010)
∆p
Pa
≈ 5974.6
L
m
φ = 0.05
Note: At this pressure gradient, MAX spin
velocity is not necessarily small. We are
kind of pushing the limit of small spin
velocities
Zero electric field solution of
Poiseuille parabolic profile
Huang Analytic Solutions V.S.
Lemaire Numeric Solutions
Agreement achieved for the all voltages
considered! (Rounding of critical electric field
strength is only within 3%)
Note: if we use particle diameter for spin viscosity,
η ' ≈ d 2η = 6.7 ×10−13 N ⋅ s
Three orders of magnitude less than best fit value.
Likely supports ER fluid parcel physical picture
Ultrasound velocity profile measurements from Prof. Lemaire’s group likely support our finite spin viscosity theory
combined with our new rotating coffee cup polarization model.
Huang, 2010
47
References
• voh.chem.ucla.edu/classes/Magnetic_fluid
s/pdf/Ferrofluids.ppt
• www.chemlabs.bris.ac.uk/outreach/resour
ces/Ferrofluids.ppt
• http://www.slideworld.com/slideshows.asp
x/Ferrofluids-ppt-426340
• http://www.slideshare.net/vponsamuel/aqu
eous-ferrofluid (method)