Bohr Model Practice Problems
advertisement

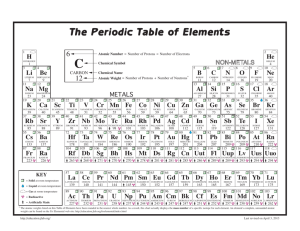
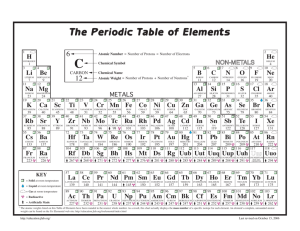
Bohr Model Practice Problems Name:______________________ Period:____ In 1943, Niels Bohr described the atom as a planetary system with electrons orbiting around the nucleus. Bohr’s model is incomplete, but it nonetheless is helpful in understanding why some atoms are very reactive and others are less so. Directions: complete each of the following models using what you know about atoms. The first is completed for you as an example. Atomic # 7__ Atomic # ____ Atomic # ____ Atomic # ____ Atomic Mass 14__ Atomic Mass_12_ Atomic Mass_40_ Atomic Mass____ Isotope Name: Nitrogen Symbol_ N_ Element:_____________ Symbol C_ Element:_____________ Element:_____________ Symbol _H_ Symbol_____ Atomic # ____ Atomic # ____ Atomic # ____ Atomic # ____ Atomic Mass_40_ Atomic Mass_11_ Atomic Mass_27_ Atomic Mass_32_ Element:_____________ Element:_____________ Symbol_____ Symbol _B_ Symbol_____ Atomic # ____ Atomic # ____ Atomic # _10_ Atomic # ____ Atomic Mass_37_ Atomic Mass_23_ Atomic Mass_20_ Atomic Mass____ Element: Sodium Element:_____________ Symbol_____ Symbol_____ Element:_____________ Symbol_____ Element:_____________ Element:_____________ Symbol_____ Element:_____________ Symbol_____ Atomic Structure practice The purpose of this assignment is to help you become more familiar with the atomic structure of some common elements by completing the chart below. For each element you have been given enough information to fill all of the blanks. Isotopic Name Isotopic Atomic # Symbol 2 Mass # # Protons Br 12 65 80 Aluminum-27 35 13 U 14 146 11 48 Ag 92 12 Krypton-84 Ca # # Valence electrons elec. 4 12 30 # neutrons 40 20 47 61 36