mic_class09_10.ppt [Compatibility Mode]
advertisement
![mic_class09_10.ppt [Compatibility Mode]](http://s3.studylib.net/store/data/008166544_1-00db1971666d959ed894b6f50935720d-768x994.png)
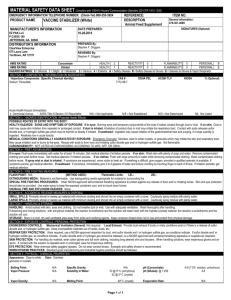
Bio 230 - Microbiology - Spring 2010 Study Guide 09 https://www8.georgetown.edu/centers/cndls/applications/posterTool/data/users/cartoon044.jpg http://www.cbu.edu/~jvarrian/122/absspex.html http://courses.cm.utexas.edu/emarcotte/ch339k/fall2005/Lecture-Ch19-3/Slide5.JPG http://cwx.prenhall.com/bookbind/pubbooks/brock/chapter13/medialib/Image2.gif http://courses.cm.utexas.edu/emarcotte/ch339k/fall2005/Lecture-Ch19-3/Slide8.JPG http://courses.cm.utexas.edu/emarcotte/ch339k/fall2005/Lecture-Ch19-3/Slide9.JPG http://cwx.prenhall.com/bookbind/pubbooks/brock/chapter13/medialib/Image2.gif TB View Spring Microbial Mats The green material from the center of the channel is are a green sulfur bacterium, Chlorobium tepidum, which lives by anoxygenic photosynthesis. It produces elemental sulfur as a waste product, and (unlike Chromatium and Thiothrix) deposits the sulfur outside its cells. Here, the water is 59°C, pH = 6.5, and highly sulfurous. http://www.lpi.usra.edu/education/EPO/yellowstone2002/workshop/tbvspring2/index.html http://www.daviddarling.info/images/Calvin_cycle.jpg http://courses.cm.utexas.edu/emarcotte/ch339k/fall2005/Lecture-Ch19-3/SlideRubisco.jpg Carboxysomes in fixed bacterial cells (top) and purified at higher mag (bottom) from Cannon et al., App. Environ. Microbiol. 67:53515361 (2001). http://www.jensenlab.caltech.edu/Projects/carboxysome.html http://www.mpi-bremen.de/Binaries/Binary135/figone.jpg http://images.vpro.nl/img.db?27852264++s(200) http://wqc-ue.eng.hokudai.ac.jp/~JOUSUI/english/Research/research%20page/ANAMMOX.png http://www.fos.su.se/~magnuss/SinNature.png Important Reactions of the Sulfur Cycle Assimilative sulfate reduction - sulfate (SO42-) is reduced to organic sulfhydryl groups (R-SH) by plants, fungi and various prokaryotes. The oxidation states of sulfur are +6 in sulfate and -2 in R-SH. Desulfuration - organic molecules containing sulfur can be desulfurated, producing hydrogen sulfide gas (H2S), oxidation state = -2. Note the similarity to deamination. http://www.biosci.ohio-state.edu/~mgonzalez/Micro521/23.html Oxidation of hydrogen sulfide produces elemental sulfur (So), oxidation state = 0. This reaction is done by the photosynthetic green and purple sulfur bacteria and some chemolithotrophs. Further oxidation of elemental sulfur by sulfur oxidizers produces sulfate. Dissimilative sulfur reduction - elemental sulfur can be reduced to hydrogen sulfide. Dissimilative sulfate reduction - sulfate reducers generate hydrogen sulfide from sulfate. http://www.biosci.ohio-state.edu/~mgonzalez/Micro521/23.html Sulfur Oxidation 1. The light-induced oxidation of hydrogen sulfide for harvesting electrons during photosynthesis by green and purple sulfur bacteria. H2S -----> So + 2 H++ 2 e2. Chemolithotrophic oxidation of hydrogen sulfide generates energy: a. Hydrogen sulfide can be oxidized to elemental sulfur: H2S + 1/2 O2 -----> So + H2O + energy b. Elemental sulfur in turn can be oxidized to sulfate: So + 1 1/2 O2 + H2O ---> SO42- + 2 H+ + energy http://www.biosci.ohio-state.edu/~mgonzalez/Micro521/23.html Chemolithotrophic Sulfur Oxidation (i) Beggiatoa - historically important because it was the first chemolithotroph identified. Gliding motility Growth on acetate Growth on O 2 is limited by the lack of catalase in Beggiatoa species. The lack of catalase is compensated for by the organisms' ability to oxidize sulfide, which detoxifies metabolically-formed hydrogen peroxide in the absence of oxygen. http://microbewiki.kenyon.edu/images/thumb/0/08/Filament.jpg/400px-Filament.jpg Chemolithotrophic Sulfur Oxidation (ii) Thiobacillus - an obligate acidophile, very tolerant of low pH; in addition to oxidizing hydrogen sulfide, this organism can extract iron from solid pyrite (FeS2) in a two-step process in which sulfur atoms are oxidized. First, the organism catalyzes the oxidation of ferrous iron, generating ferric iron Fe2++ 1/2 O 2 + 2 H+ -----> Fe3++ H2O Secondly, the ferric iron produced spontaneously reacts with pyrite FeS2 + 14 Fe3+ + 8 H2O -----> 15 Fe2+ + 2 SO42- + 16 H + A colony of Thiobacillus ferrooxidans. The reddish color is the result of iron production Some of the Reactions Undertaken by Thiobacillus http://microbewiki.kenyon.edu/index.php/Thiobacillus A marine sulfuretum is a microbial mat in which the total turnover of inorganic and organic compounds is heavily dominated by the sulfur cycle http://www.stat.rice.edu/~siefert/Links/SulfurRedTBL.html Diurnal cycle of oxygen and sulfide distribution and of microbial zonation in a marine sulfuretum. The zero line in each box indicates the interface between the sediment and the overlaying water phase. The dominant genera at each stratum are indicated in each box. http://www.stat.rice.edu/~siefert/sulfuretum.gif Model of a Marine Sulfuretum http://www.stat.rice.edu/~siefert/Links/SulfurRedTBL.html The End