Barite solubilities and thermodynamic quantities up to 300'C and
advertisement

American Mineralogisl, Volume 62, pages 942-957, 1977
Baritesolubilities
andthermodynamic
quantities
up to 300'C and1400bars
Cue,nlrsW. BlouNr
Departmentof Geology,Idaho
State Uniuersity
Pocatello.Idaho83209
Abstract
Solubilities
of specially
synthesized
in HrO solutions
coarse-grained
bariteweredetermined
from22to 280"Cand I to 1400bars,andin 0.2and4 molalNaCl solutionsfrom 100to 250'C
and I to 500bars.Baritesolubilities
arelow, rangingbetweenl0-6 and 10-3molal.Isobaric
solubilities
aremaximumnear100oCin HzO solutions;the maximamigrateto progressively
highertemperatures
in solutionsof increasing
NaCl concentrations.
Isothermalsolubilities
increasewith rising NaCl concentrationand pressure.The effectof NaCl on increasing
solubilities
greaterwith risingtemperature.
becomes
Changingtemperatures
causedifferentresponses,
dependingon the compositionol the
solutionand thetemperature
range.Initially-saturated
dilutenaturalwaterswouldprecipitate
baritewith eitherincreasing
temperature
or decreasing
temperature,
depending
on thecircumstances.
Salinesolutionsare capableof behavingmuch like dilute solutionsbelow I molal
NaCl, but at higherNaCl concentrations
the temperature
effecton isobaricsolubilitiesis
monotonicallypositive.Barite solubilityin sufficientlysalinesolutionsin nature would
increase
with depth,due to increasing
wouldtendto
temperature
and pressure.
Precipitation
occurduringmigrationof solutionstowardthe surface.Because
the low solubilityof barite
precludeseffectivetransportof BaSOoin large quantities,precipitationof barite by the
reactionof Ba2+with sulfatederivedfrom the oxidationof 52 may be moreimportantas a
depositional
mechanism
than changes
in the temperature-pressure
environment.
Thermodynamic
equilibriumconstants(K) and the free-energy
changesfor the reaction
(AG) for the dissolutionof barite were calculatedfrom the solubilitydata.AG valuesin
kcallmoleat I bar arecloselyreproduced
X l0 1T' - 4.02952
by theequationAG: 1.15046
x 10-27+ 15.386
(Iin 'K). Enthalpies,
entropies,
andvolumesofreactionobtainedfrom the
solubilitydata arein accordwith publishedthermodynamic
data.Activity coefficients
calculatedusingbaritesolubilities
in NaCl solutionsand log K dataareaccurately
modeledby an
extendedform of the Debve-Hiickel
eouation
Introduction
This paper presents experimental determinations
of the solubility of pure, coarsely-crystalline
barite in
water and NaCl-water solutionsover a range of temperatureand pressureconditions.The solubility studies provide necessarybasic physical-chemicaldata
neededto understandthe causesofprecipitation, dissolution, or decompositionof barite in nature. They
also provide data needed for evaluating thermodyn a m i c q u a n t i t i e so f B a S O r i n a q u e o u ss o l u t i o n s .
Barite occurs in a wide variety of geologicenvironments. Most economically-importantbarite deposits
are found in sedimentary rocks as bedded barite
(Brobst, 1970). Barite also occurs in sedimentary
rocks as rosettesand nodules, as a cementing mineral, and as residual accumulations. Many occurrencesof barite are of hydrothermal origin. Sulfide
mineral deposits commonly contain barite as a
gangue mineral. Some gold deposits in the western
U.S. contain frankdicksonite(BaFr) as well as barite
(Radtke and Brown, 1974).Barite is also associated
with some carbonatites (Heinrich, 1966). The extreme diversity of barite occurrencessuggeststhat
barite is depositedunder diversegeochemicalconditlons.
Many measurementsof barite or BaSOnsolubility
have been made. In most studies,the solid utilized
was either a sample of natural barite or precipitated
942
BLOUNT.' BARITE SOLUBILITIES
BaSOo.The useof either of thesesolids can introduce
error. Natural baritesmay contain minor amounts of
other elements,such as Sr, Ca, and Pb, as substitutes
for Ba in the crystal structure.The presenceof small
amounts of impurities may affect Ba concentrations
in aqueoussolutions(Gundlach et al., 1972).Barites
with strontium showed an incongruent solubility.
Natural barites may also contain small inclusionsof
other minerals capable of dissolving and increasing
the ionic strength.The very low solubility of barite,
o f t h e o r d e r o f l 0 - 5 m o l e s B a S O +p e r k i l o g r a m o f
solvent causes similar levels of other ions to have
much greater relative effect on solution properties
than they would if concentrationswere higher. Ionic
substitution and the contamination of sampleswith
other minerals can be avoided by using precipitated
barium sulfate. However, these precipitatesmay be
poorly crystalline, have a very fine grain size, and
contain occludedbarium chloride, sodium sulfate,or
other salts,dependingupon the conditions of precipitation. Balarew (1925) pointed out that BaSOnprecipitates contain up to as much as I percent of occluded BaClr. Even the precipitates washed until
negative tests were obtained for chloride released
more Cl- on prolonged grinding. In a recentstudy of
SrSOnprecipitates,Mager and Lieser (1972) showed
that recrystallizationcaused the slow releaseof occluded NarSOo.
Particle size and crystallinity also affect measured
solubilities.Lemarchands(1928) found that particles
smaller than 1.7 microns had solubilitiesas much as
three times that of larger particles. Herovsky and
Berezicky (1929) found that freshly-precipitated
BaSOnwas about 8 times more solublethan a precipitate 30 hours old. In studies of SrSOn solubility,
Gallo ( 1935)showedthat a calcinedprecipitatehad a
lower solubility than a newly-formed precipitate.
Measurementsof celestitesolubility by Striibel (1966)
and by Blount and Dickson (1967) were significantly
lower than were obtained by Gallo on calcinedmaterial. If syntheticsolids are to be used,they must be as
free as possible of occluded ions, consist of grains
larger than 2 microns, and be highly crystalline.
Solubilities of barite and precipitated BaSOn in
water and in salt solutions made by many investigatorshave been summarizedby Striibel (1967)
and Gundlach et al. (1972). Resultsof the more consistent studies of later researchersare illustrated in
Figure l. Table I summarizes previous studies of
BaSOnand barite solubility in various kinds of salt
solutions.
A wide variety of analytical procedureswere used
943
by different investigatorsfor the solubility measurements. Templeton (1960) used emissionspectroscopy
for barium and a reduction-colorimetricprocedure
for sulfur. Radioisotope methods using precipitated
BaSOo with barium 140 and 133 as a tracer were
employed by Puchelt(1967).Stri.ibel(1967)employed
crystal weight-lossand X-ray fluorescenceto determine barium. Uchameyshviliet al. (1966)usedcrystal
weight-lossmeasurementson barite crystals.Some of
the scatter in experimental results reflectsanalytical
problems and uncertaintiesas well as significantsolubility differences.
Present study
E xp erimental materials
Samples of coarse-grainedsynthetic barite were
prepared by slow precipitation from a hot, acidic,
d i l u t e ( 0 . 0 1 m o l a l ) B a C l , s o l u t i o n sb y a d d i n g d i l u t e
NarSOa solution, following proceduresdescribedby
Blount (1971, 1974).The syntheticbarite consistedof
0 . 5 g q u a n t i t i e so f c r y s t a l sr a n g i n g f r o m 0 . 0 1 t o 0 . 1
mm thick. No amorphous or submicron mateiial was
present. Except for the growth of some crystals to
form aggregates,barite was unchanged after a year of
exposure to solutions at elevated temperaturesand
pressures.All experimentalsolutionswere made from
redistilledwater and reagent-gradechemicals.
Analytical procedures
Barium determinationson experimental solutions
were made by X-ray fluorescenceanalysis of ionexchange resin pellets after the barium had been
taken up by the resin, as describedby Blount et al.
(1971, 1973). Pre-weighedquantities of a chelating
ion-exchangeresin (Chelex 100) were equilibrated
with weighed quantities of experimentalsolutions at
a pH near 10. Strongly saline solutions were diluted
l0-fold to eliminate interferenceby sodium ion. The
resin was recoveredby filtration, dried, and pelletized
in a 0.5" diameterdie. Standardpelletswere prepared
by equilibrating solutions with known quantities of
barium with resin samples. Barium recovery was
around 99 percent from test solutions. A Phillips
model l4l0 vacuum spectrograph equipped with a
100 kV tungstentarget tube was usedto measurethe
intensity of the BaKa peak. The accuracy of the
analytical procedurewas better than *5 percent.The
Ka peak, in spite of high background, showedbetter
linearity and reproducibility for the resin sample
analysis procedure than the La line.
BLOUNT: BARITE SOLUBILITIES
+ M e l c h e r( l 9 l O )
oN
2 . O r N e u m o n( 1 9 3 3 )
A
a Srriibet il967)
o P u c h e l t( 1 9 6 7 )
r Tempelton(1960)
I
c|t
)<
-t'
A
^ r - d 6 "4-,tv
u
6 Molol
a
a
CI
t
b
5
c
.o 6
a
A
A
..-f-?
A
Molol
?
-^?ol
ffi
I Molol
)/
A..gb+s'$B-..o'bp- o
.zs w o to t
f
2roo
A^
d.oo
tooo ""-
U)
o
0 .oz
E]
L
' 'J2Nv
Hzo
-.."*
too
A
A
r9t4
A
a74
165
z--\
.ol &
o
r084
ro70
585
,A.,
\
200
300
400
500
600
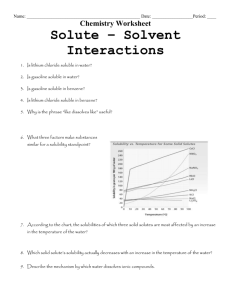
Temperoture
"C
F i g . l . R e s u l t so f p r e v i o u sd e t e r m i n a t i o n so f B a S O no r b a r i t e s o l u b i l i t y i n H r O a n d s o m e N a C I - H r O s o l u t i o n s W h e r e n e c e s s a r ys, h o r t
i n t e r p o l a t i o n so r e x t r a p o l a t i o n sw e r e u s e dt o o b t a i n v a l u e sa t t h e i n d i c a t e dN a C l c o n c e n t r a t i o n sT h e n u m b e r sn e x t t o d a t a p o i n t s a r e t h e
D r e s s u r e si n b a r s .
E xperimental equipment
Solubility measurementswere made using the hydrothermal solution equipment (HSE) describedby
Dickson et al. (1963). A 400 ml capacity collapsible
teflon sample cell, containing 0.5 g synthetic barite,
and filled with boiled, CO,-free distilled water or
NaCl-water solution, was connectedto a teflon-lined
stainlesssteel tube and installed in a large stainless
steel pressure vessel. Pressure was controlled by
pumping COr-free water into or out of the spacein
the pressurevesselsurrounding the samplecell. Heating was provided by a large muffie furnace.The HSEfurnace assembly was rocked through an angle of
l70o to provide mixing of solution and barite. Samples of this solution were taken from the sample cell
at intervals of time (2 to 12 days) by opening a valve
located at the other end of the teflon-lined tubing
connected to the sample cell. Pressurewas maintained by simultaneously pumping water into the
pressurevessel.Experimental conditions were maintained until analysesof successivesamples agreed
within the uncertainty of the analytical procedure.
Uncertaintiesin temperature,measuredwith a chromel-alumel thermocouple, were 1.5 percent. Pressures were measuredwith a calibratedprecision-type
Heisepressuregaugewith an uncertaintyof t 3 bars.
System BaSOo-HzO
Experimentaldeterminationsof barite solubility in
water are presented in Table 2 and illustrated in
Figure 2, which includes data by other workers for
comparison. Solubilities were measured from both
undersaturated("U") and supersaturated("S") conditions. Table 3 gives solubilitiesat selectedconstant
temperature and pressure conditions. The isobaric
solubility curves in Figure 2 show maxima between
100 and 150'C. Pressureand temperatureeffectson
barite solubility above l50oC are similar to solubility
94s
BLOUNT: BARITE SOLUBILITIES
T a b l e l . S u m m a r y o f e x p e r i m e n t a l c o n d i t i o n s used for p r e v i o u s s t u d i e s o f t h e
solubility of barite and precipitated BaSOnin s a l t s o l u t i o n s
Authors and
Experinental
Solid
Salt solutions
studied
Range of
Concentration
TenPerature
Fresenius Q
N H 4 C 1 ,N a c 1 ,
0 - 10 wt.%
Not Given
Hintz (1896
HNo3,HC1
"oorn
ppt. BaS04
F e C1 A 1 C 1
3,
J
MgCt,
1-100 g/liter
ppt. BaS04
Fraps (1902)
Neunan (1935)
KCl, KNO3,
0.00001to
ppt. BaS04
MgClr, Me(Nor),
0.036 \nolar
tenp.
Not given
aoon tenp.
^-o^
LaClr, La(NOr),
Templeton (1960)
NaCl
ppt. BaS04
Lieser (1965)
et al.
(1966)
25 - 950C
weight percent
Na2S04
ppt. BaS04
Uchaneyshvi1i
0.2 to 20
0 . 0 0 0 1t o
2ooc
L rnolar
N a C 1 ,K C l ,
0.1-2 moles/kg
95 - 3700C
25 e sooc
M g C 1 2 ,C a C l 2
barite
Puchelt (1967)
NaCl, KCl
0to26
ppt. BaS04
M g C 1 2 ,C a C l 2
weigllt percent
striiber (r967)
NaCl
0-2 noles/kg
z0 - 6oooc
p p t . B a S 0 OE
barite
sweptout
lined samplingtube and be subsequently
during sampling.The first few millilitersof sample
solution were discardedin an effort to reducethe
possibilityof samplecontamination.
Smootheddata correctedto regular intervalsof
were
pressure,
and NaCl concentration
temperature,
Vapor-pressure
3.
of
Figure
in
the
construction
used
data for NaCl solutionswerefrom Haas(1971).The
of BaSOnhave
presence
of millimolarconcentrations
SystemBaSOo-NaCl-H20
only trivial effectsupon solution vapor pressures.
baritesol- Baritesolubilityin NaCl solutionsis greaterthanthe
Table4 givesexperimentally-determined
ubilitiesin 0.2 and 4.0 molal (moles/kgHzO) NaCl solubilityin waterat all conditionsstudied.Increassolubility.
solutions,at temperaturesfrom 94 to 253oCand ing pressureinvariablyincreases
pressuresclose to the vapor pressureand near 500
Baritesolubilitiesof Figure3 weregraphicallyinto obtainsolubilityvalues
bars. The uncertaintyof barium analysesin the 4- terpolatedor extrapolated
and
molal solutionsis greater(t1Vo) than in wateror in at 100,150,200,and 250'C at the vaporpressure
of sodium at 500bars.Thesevalueswereusedto prepareFigure
morediluteNaCl solutions.A largeexcess
can result in less than a 100 percent recovery o-f 4, in whichlogarithmsof baritesolubilityare plotted
resin.High resultscould againsta functioninvolvingthe squareroot of NaCl
bariumby the ion-exchange
be producedby contaminationof samplesolutions concentration.The curveswere positionedon the
during sampling.Under theseexperimentalcondi- basisof an extendedDebye-Htckelequation.Solutions, BaSO4could precipitatein the coolerteflon- bilitiesplottedin thisway changealmostlinearlywith
trendsof anhydriteand celestite(Blount and Dickson, 1967,1969;Blount and Dickson,in preparagreatersolution), both of which showconsiderably
bility. Isothermal solubilitiesof all the minerals
rise.Plotsof
at increasing
rateswith pressure
increase
logarithmsof solubilitiesu.r. pressureat constant
temperature,and us. temperatureat constantpressure(above150"C),are nearlystraightlines.
BLOUNT: BARITE SOLUBILITIES
the NaCl concentration function. Figure 9 presents
25oC data of other workers plotted in the sameway.
Solubifity measurementsby previous workers are
shown for comparison in Figures 3 and 4. There is
rather good agreementbetween the solubilities obtained during the presentstudy and those obtained by
Uchameyshvili et al. (1966). Results obtained by
Striibel (1967) and Templeton (1960) are generally
of barite solubilityin HrO solutions
Table2. Exoerimentaldeterminations
Experimental
condition
Sanple
Nunber
Tenptgrature
in "C
Pressure
in bars
Approach
Direction
Equilib.
I rme uays.
BaSOoin
nilliroles
kg HrO
36
47
46
u2
DJ
96
97
99
u12
u37
0 . 0 1 . 74
0 . 0 17 6
0 . 0 17 t
B4
B5
95
496
460
U5
u11
0.0258
0.0242
B6
B7
97
96.5
882
862
U3
U6
0.0575
0.0342
2R
B8
97
466
U8
0.0253
4
B9
810
189
189
402
404
S4
S9
0 . 0 15 0
0 . 0 1 46
B11
872
813
189
189
188
100
100
106
S4
U3
U5
0 . 0 10 0
0 .0 l 0 4
0.0105
o
874
190
189
904
902
U6
u11
0.0237
0 .0 2 3 9
7
B16
248
s22
s2
0.0083
/K
B17
249
484
U5
0 . 0 07 9
8
818
B19
247249
956
1010
U4
U8
0.0140
0.0147
9
B20
247
249
92
104
S3
JO
0.0048
0.0048
bz5
279
279
994
996
U5
u7
0.0120
0.0125
824
ZLJ
492
468
DJ
B1
B2
bzL
10
11
822
ZI6.
)
s10
0.0124
0.0118
1lR
826
827
277
277
472
456
U8
0.0115
0.0117
I2
828
829
277
224
106
176
S3
s14
0.007s
0.0066
15
YI
\2
24
24
I002
1 00 8
U2
U8
0. 0289
0.028d
S5
S6
s10
0.0182
0.0r77
0.0r87
U7
u10
ull
0. 0546
0.05s2
0.0s18
S3
0.0179
0.0156
0.0160
T4
t5
16
I5
23
Y4
Y5
22
500
518
508
Y6
Y7
Y8
r57
156
158
r406
L392
1 41 0
Y9
Yl0
Yll
t55
157
102
702
110
UJ
50
S9
947
BLOUNT: BARITE SOLUBILITIES
B o r i t e S o l u b i l i ty
i n Wo te r
Table 4
Experimental determinations of barite solubility in
NaCl-H"O solutions
NaCl in
noles
xcH-F
Baso, in
n
illinoles
--Tc
H-I-
0,20
0.077
0 .069
Tenperature
in oc
Pressure
in bars
Equilib,
Tine
in days
z7
z2
224
t34
726
3
5
z3
z4
253
253
L24
728
3
9
0.058
0.057
zs
z6
177
767
t2
8
3
10
0.077
0.076
z7
118
5
2
0. 084
ol
z8
z9
270
98
9 8. 5
100
478
492
492
73
20
49
0.106
0.108
0.113
c.oo7
zt7
zt2
t49
150
500
500
t2
14
0.115
o.!2t
274
245
245
502
482
9
19
0.088
0.092
246
106
103
2
3
0.058
0.062
Y
Y
Y
96.5
94.5
520
480
500
3
7
t4
Y
Y
Y
94
96
95
106
106
92
y7
Y8
Y9
Y10
Y11
YL2
153
153
152.5
752.5
560
Y13
Y14
202
202
203
203
202.5
200
200
Smple
Nuber
a-\
c\l
c|.)
)<
.o2
a
q)
-
A\
L
^$
r-) .oo5
a
o
m
.oo3
o02
o Present Study
+ Melcher(l9lO)
^ N e u m o n( 1 9 3 3 )
r Templeton(1960)
^ srrilbet(1967)
50
roo
t50
200
250
300
350
Te mp e ro tu re
"C
Fig. 2. Solubilityof baritein HrO asa functionof temperature
at
severalpressures.Results by previous workers are shown for
comparison.
The letters"U" and "S" indicatewhetherthe data
point was approachedfrom undersaturatedor supersaturated
solutions
r l5
higherthan thoseobtainedin the presentstudy.Bar- Y 1 6
ite solubility determinations
of this study and by Y77
Y18
Uchameyshvili
et al. ( 1966)giveinternally-consistentY 1 9
data over the rangesof temperature,pressure,and
Table3 Baritesolubilities
in millimoles,/kg
HrO in HrO solution
at varioustemperatures
and pressures
100
bare
Pressure
500 barg
1400
bare
loc
Low*
Preaaure
z)
60
80
100
0.0106**
0.01.52**
0.0160**
0.0158
0.0L17
0.0163
0.0L75
0.0180
0.0184
0.0236
0,0251
0,0260
0,0290
0.0350
0.0380
0.0390
0.0480
0 .0 5 1 0
0.0535
L25
150
175
200
0.0166
0.0146
0.0113
0.0084
0.0180
0.0r.58
0.oL24
0.00920
0.0261
0.0232
0,0187
0.0141
0.0405
0,0375
0.0307
0.0240
0.0s60
0,0540
0.0465
0.0360
225
250
300f
0.00620
0.00445
0.00248
0.00575
0.00470
0.00250
0.0107
0,00790
0,0044
0.0190
0.0147
0.0089
0.0301
0.0243
1000
bars
+Vel.ue6 obtalned by short
by Melcher
qtrapolatton.
6
33
39
42
456
500
507
498
499
470
492
5
L4
77
2L
73
0.51
0.50
0.53
0.52
0.49
0,69
0.73
0.76
0.69
0.74
0.73
o,64
NaCl concentration
conditionscovered.Table5 presentsa summaryof baritesolubilitiesat evenvalues
pressure,
and NaCl concentration.
of temperature,
Geologicalapplications
*Pregsure equals I bar up to l-00oc, the vapor preseure above
1000c.
**Solubllitles
based on studleo
Tenpleton (1950).
0.324
0 .3 1 1
0.307
0.260
0,265
0.255
492
500
500
ls2
4.00
(1910) and
The behaviorof baritesolubilitybelowthe Earth's
tempersurfaceis a functionof manyfactorsbesides
ature and pressure.Such factorscould includethe
presenceofl other dissolvedsubstances
in solution,
ion exchangewith clay minerals and other substances,and the possibleinstabilityof barite under
sulfate-reducing
conditionsor underconditionsthat
favorthe formationof witherite,Ba COa.The experigive the chemicaltendencies
mental measurements
which may not be apfor idealizedcircumstances,
proximatedin nature.Striibel(1968)suggested
that
from naturalsalinewatersby
baritecouldprecipitate
BLOUNT. BARITE SOLUBILITIES
lower pressures.For the models used here, barite
solubility under the low-pressureconditions passesa
maximum at about 4 km. Under the high-pressure
conditions, solubilitiesremain nearly constant below
4 km. Therefore, barite-saturated ground waters
moving in the Earth's crust could either dissolveor
precipitate barite according to the direction of flow
and the nature of pressuregradient. An important
possibility is that barite could be precipitated from
barite-saturatedground waters migrating toward hot
regions in the crust.
The importance of NaCl solutionsin barite deposi-
20
to
- TN-
nq
cr)
)l
@
(D
o2
^,
v.r
o
Eos
c
$"
a-\
ol
(/) oo5
co
oo2
o
o
r
a
o
o
PresentStudy
Templeton(1960)
S t r t i b e (l 1 9 6 7 )
P u c h e l(t1 9 6 7 )
U c h o m e y s h vei fl io l . ( 1 9 6 6 )
50
too
t50
200
250
TemoerotureoC
300
Fig. 3. Solubility of barite in HrO and 0.2 and 4 molal
NaCl-HrO solutions as a function of temperatureat the vapor
p r e s s u r ea n d 5 0 0 b a r s . P r e v i o u sr e s u l t sa r e s h o w n f o r c o m p a r i s o n
cooling, or by reaction of Ba-containing solutions
with sulfate produced by oxidation of sulfide ion;
conclusionsof this study are in agreement.
The solubility data can be used to construct diagrams depicting saturation conditions in the Earth's
crust. Such diagrams are generallyfor simplified and
essentiallyartificial conditions. Curves in Figure 5a
depict constant-composition sections through the
barite saturation surfaceprojected onto the temperature-pressureplane. The saturation surfaceis terminated at low pressureby the curve representingthe
vapor pressureof saturated solutions (which is indistinguishablefrom the vapor pressurecurve ofpure
water at the scale of the figure). The dashed lines
represent two possible temperature-pressureconditions for an assumed geothermal gradient of
30'C/km. The first is for a pressuregradient of 100
bars/km, as a simple approximation to that of a
column of water (hydrostatic). The second is for a
pressuregradient of 250 bars/km, as a simple approximation to that produced by a column of rock
(lithostatic). Solubilities under these two conditions
are illustrated in Figure 5b. At a given depth, barite
solubility is invariably greater at the higher-pressure
conditions. Barite crystals would therefore tend to
dissolveat points of pressureand precipitatebetween
detrital grains where the pressure is lower. Barite
could also precipitateat constant temperatureby migration of solutions along pressuregradients as, for
example,from placesat high pressureto fissureswith
oN
-
c'l
)<
a
(D
o
E
=
C.
Og
U)
r
o
a
o
o
m
NoCl
Templeton(1960)
Uchomeyshvili
et ol. (1966)
srriiber(1967)
PresentStudy
/ Kg,H2O
.JVloles
o.r.2.3.4.5.6.7
m
-2( N o C t )/, ,l ?
m
-t ( N o C t )
Fig.4. Baritesolubilityin NaCl-H,O solutionsas a functionof
the NaCl concentration.Previous results are shown for
comparison.The curvesrepresentsolubilitiescalculatedfrom
equilibrium constantsand an extendedDebye-Hiickelequation
(Equationl2 in text)
BLOUNT: BARITE SOLUBILITIES
pressure,
and NaCl concentration
in NaCl-HrO solutionsat evenvaluesof temperature,
Table5. Baritesolubilities
Temp.
NaCl in
Press.
'c
bars
moles/kg
o,2
0.0
HrO
1*
0.5*
(BaSOO in
nillimoles/kg
HrO
100
1
500
0.0168
0.0260
0.058
0.084
0.079
0.1r2
0.123
0.L47
o.L72
0,233
o.240
0.330
0.290
0.38
0.333
0.415
0.38
0.48
150
4-5r
500
0,0r45
o,0232
0.056
0 . 07 8
0.080
0.118
0.143
0.188
0.220
0.297
0.337
0.44
0.425
0. 5 2
0.50
0.63
0.61
0.74
13-151
500
0.0084
0.014r
0.0415
0.070
0.057
0. 108
0.130
0. 208
o.232
0.340
0.43
0.562
u.)))
0.68
0.90
0.90
r. 15
36- 39r
500
0 .0044
0.0079
0 .0305
0 .0505
0.053
0.087
0.L25
0.195
o,247
0.350
0.495
0.72
0.725
1.03
0.94
r.32
1.30
L.77
*interpolated
**extrapolated
frange
of
solublLity
solubility
vapor pressures
tion is attestedto by the occurrenceof concentrated
salt solutions in barite fluid inclusions (Roedder,
1972).ln solutionswith NaCl concentrationsabove I
mole/kg HrO, barite solubility shows no maximum
but increasescontinuously with increasing temperature. Barite solubility in NaCl solutions would increasewith depth becauseof increasedtemperature
and pressure.Therefore, constant-NaCl barite-saturated solutions that migrate upward from depth toward the surfacecould depositsome barite. The dilution of barite-saturatedNaCl solutions with HrO at
constant temperature and pressure would produce
undersaturated solutions. Barite solubility in HrO
and in NaCl-HrO solutions is quite low. The presenceof MgCl, and CaCl, (Uchameyshviliet al.,1966)
increasesbarite solubilities,but only to a minor extent. Oxidation and reduction of sulfur speciesin
solution probably have a more important role in the
precipitation or dissolution of barite in nature than
solubility. Barite solubilitiesare so low that the transp o r t o f l a r g e a m o u n t s o f B a S O ni n s i m p l e H r O o r
NaCl-HrO solutions seems highly improbable'
Therefore, the deposition of barite in nature most
likely requires the mixing of two different ground-
HzO
Millimoles/Kg
BoSOo
Pressurein bors
?o
roo
500
rooo
1400
0
0r
50
.o2
03
04
%"
r\6
2
Y4;
1O
o)
(J
o
o
40)
F
,""
Y
L 2nn
o-
7 -C.
(D
q(D
@
"o
/"e
o-:
9
350
(5a) Constant-conposition
F i g . 5 . B a r i t e s o l u b i l i t y i n H z O s o l u t i o n s a t d e p t h u n d e r s p e c i f i e dg e o t h e r m a lp r e s s u r e - g r a d i e ncto n d i t i o n s
obtained
c u r v e s ( m i l l i m o l e s / k g H z O ) o n t h e b a r i t e s a t u r a t i o n s u r f a c ep r o j e c t e do n t h e T , P p l a n e ( 5 b ) B a r i t e s o l u b i l i t y a t d e p t h i n k m ,
p
r
e
s
s
u
r
e
s
l
i
t
h
o
s
t
a
t
i
c
a
n
d
h
y
d
r
o
s
t
a
t
i
c
a
p
p
r
o
x
i
m
a
t
i
n
g
a
t
c
o
n
d
i
t
i
o
n
s
g
r
a
d
i
e
n
t
o
f
3
0
"
C
/
k
m
f r o m 5 a a s s u m i n ga g e o t h e r m a l
950
BLOUNT.' BARITE SOLUBILITIES
Table 6. Evaluationof log K for the solution of barite in water
1 bar
-Iog K
I00 bars
-Iog K
25
60
80
100
9.98
9.67
9 .6 3
9.s9
9.88
9.61
L25
150
I75
200
9.62
9.13
9.94
LO.22
9.65
9. 86
10.11
225
250
300
IC.53
10, 80
LL.44
10.40
t0 .69
11.31
I"C
9.53
500 bars
-1og X
1000 bars 1400 bars
-Iog K
-Iog K
9 .5 1
9.30
9.24
9.22
9.L2
8.96
8. 89
8.87
9.22
8.57
8. 61
9.50
9.75
8.85
8 .9 1
9.09
9. 30
9.99
r0.24
10.73
9.50
9.72
1 0, 1 3
9.14
o
lt
8.69
8.65
8.61
o. t 5
8.95
clude: logarithms of thermodynamic equilibrium
constants, log K; standard free energies AG; enthalpies, A11; entropies, AS; volume changes,A Z;
and heat capacitiesat constant pressure,ACo; and
heat capacitiesat constant temperature, AC7.1 The
reaction consideredfor thesecalculationswas.
(l)
B a S O o ( c ): B a ' * ( a q ) + S O ! - ( a q ) ,
in which the reactant is barite (c) and the products
are the ions in solution (aq) at the hypotheticalstand a r d s t a t eo f u n i t m o l a l i t y ( m : l ) .
Eualuation log K and LG
The first stepin the treatment of the solubility data
water solutions. For example, a Ba2+solution could
was
to calculatevalues for log K using the relatione n c o u n t e ra S O ! s o l u t i o n , o r a s o l u t i o n c o n t a i n i n g
s
h
i
p
,
both Ba2+ and some sulfide ion could encounter
oxygen-bearingground waters that can oxidize sul- ( 2 )
log K : 2log m 'r 2log 7^
fide to sulfate.
in which m representsthe molality of saturatedsolutions and 7- is the mean activity coefficienton the
Thermodynamic quantities
molality scale. Activity coefficientswere calculated
Inlroduction
using an extended form of the Debye-Hiickel equaThis section presents thermodynamic quantities
calculated from barite solubility data and compares
' T h e s u p e r s c r i p t sa n d s u b s c r i p t sc o m m o n l y u s e d i n
thermodythem with literature values.The calculatedquantities n a m i c e x p r e s s i o n sa, s i n t h e t a b l e s a n d f i g u r e s ,h a v e b e e n o m i t t e d
for the reaction of barite with aqueous solutions in- f r o m t h e t e x t t o r e d u c e t h e t i m e a n d c o s t o f t y p e s e t t i n g .
Table 7. Values for -log K for barite solubility at I bar
oo
Fta
P
tr>
.
od
E
o(J
.iv
i@
OP
'ru)
ov
9. 98
35
50
60
P^
9. 96
9.70
6.i
F
9. 96
9.83
9.71
dd
=
Oa
40
O\O
boo
oe
9.72
9.70
9.49
9. 4 2
s
op
Prd
.do
.io
9.97
9.97
9.97
v. 50
9.60
9.66
9.53
9.59
9.s2
9.60
9.75
o o/
9.59
9.65
9.78
9.96
10.20
TI.47
ON
pv
</)
dv
9.67
9.66
o)
I .44
80
95
9.63
L62
9.59
r00
v.
L Z>
9.60
v. J)
150
775
J.t5
9.94
10.22
10.53
10.80
LI,12
L t, 4 4
200
225
2s0
275
300
Jv
*Calculated
9 .s 8
9.22
9.50
9.49
9.70
9.90
9.34
L40
9.50
1 0 .r 5
10.45
10.90
11.45
9.67
9.47
r0.00
1 0 .3 4
9.60
r 0. 4 7
r0.18
r0.46
10.75
11.05
9.83
1I.10
I1.40
9.45
TZ.LJ
using Thermodynamic data in the literature.
Refer to text.
r0.47
r0.77
951
BLOUNT. BARITE SOLUBILITIES
tion
(3)
log^y^: GIZ*Z-l A* {l^)
/(l * B^a,l
I^).
in which lZ*Z-l is the absolutevalue of the product
of ionic charges(4 for BaSOn),A^ and B^ are temperature and solution density dependentparameters
that include the dielectricand severalother physical
constants,-f- is the ionic strengthof solutionson the
molality scale, and c1 is an ion-sizeparameter. The
required physical constants were obtained from the
ReuisedList of Physical Constants (National Bureau
o f S t a n d a r d s ,l 9 7 l ) . T h e d i e l e c t r i cc o n s t a n tw a s c a l culated from equations 1, 2, and 3 given by Quist and
M a r s h a l l ( 1 9 6 5 ) ,w h i c h d e p e n d o n l y u p o n t e m p e r a t u r e a n d s o l u t i o n d e n s i t y .S o l u t i o n d e n s i t i e sa t t h e
low concentrations considered were assumedto be
the same as those for water at the same temperature
and pressure. Water densities were obtained from
Kennedy et al. (1958) and Holser and Kennedy
(1958). An averagevalue for the ion-size parameter
was obtained from Kielland (1937). Log ^y^ values
ranged from 0.013 to 0.036 for the experimentalconditions covered in this study. As previously stated,
uncertainties in the solubility data were about t5
percent or t0.02 in log m. Since uncertaintiesin log
^yn are much less than the calculatedvalues, the uncertainty in log K is essentially2 times the uncertainty
in log m or t0.04. The 300'C valuesof log K have an
uncertainty of *0.10 introduced by the short extrapolation neededto obtain log rn values.
Calculated valuesfor log K are presentedin Tables
6 andT and plotted againstl/Iin Figure 6. There is a
maximum in log K near l00oC at all pressures.Increasingpressurecauseslog K values to increasealmost linearly. The I bar log K values above l00oC
were calculatedfrom solubilitiesestimatedby extrapolating downward from solubilitiesat the vapor pressures.Uncertaintiesintroduced by the short extrapolation are small and therefore do not significantly
affect the uncertainty in log K values.Many different
valuesfor log K, most of which were at or near 25oC,
but some at elevatedconditions, have been cited by
S i l l e n a n d M a r t e l l ( 1 9 6 4 , l 9 7 l ) . V a l u e sa t 2 5 o C a n d
a b o v e ,o b t a i n e df r o m t h e s t u d i e sb y M e l c h e r ( 1 9 1 0 ) ,
Templeton (1960), Stri.ibel (1967), Malinin et al.
( 1 9 6 9 ) ,a n d H e l g e s o n( 1 9 6 9 )a r e g i v e ni n T a b l e 7 . T h e
solubility data of Melcher and Templeton were in
agreementwith the present study, and their log K
valuesnaturally agree.Values reported by Malinin el
al. are higher (lessnegative) below 150'C and lower
above this temperature.Helgeson'slog K values are
"€ooooo
o
s':*':::--.u
i
. f-u;:,$tti"*t'-3:=;"" u---oir'::'{
ir'$':',
.uo9.-i .6
g-bu'u--
ot
I M o l i n i ne l o l . ( 1 9 6 9 )
(A ) t
r H e l g e s o(n1 9 6 9 )
. Bosedon Crisss Cobble
( 1 9 6 4o , b )
oC
-i
roo
r50
60
Y'
I
300
16
25
20
l/T
3.O
34
K x lO3
F i g . 6 . V a l u e s f o r l o g K a s a f u n c t i o n o f t h e r e c i p r o c a lo f t h e
a b s o l u t et e m p e r a t u r ea t I , 5 0 0 , 1 0 0 0 ,a n d 1 4 0 0b a r s . L o g K v a l u e s
obtained in previous determinations or calculated from
thermodynamic data are shown for comparison Dotted curves
r e p r e s e n tv a l u e s c a l c u l a t e df r o m e q u a t i o n 5 i n T a b l e l 0 D a s h e d
c u r v e s r e p r e s e n t sv a l u e s c a l c u l a t e df r o m e q u a t i o n 3 i n T a b l e l 0
Size of open circle representsthe uncertainty.
higher at all temperatures.Log K values calculated
using thermal data for crystalline barite (Kelley,
1 9 6 0 ) ,e n t r o p i e sf o r i o n s ( L a t i m e r , 1 9 5 2 ) , a n d e n tropy and heat capacity data for the ions in aqueous
solution at higher temperatures(Criss and Cobble,
1964a,b) differ from valuesobtained in other studies
except at25"C. The differencescan be accountedfor
by changing L,H by 300 caloriesor AS by I entropy
unit.
AG valuescalculatedfrom the equation,
(4)
LG : -2.3025 RT log K,
are listed in Table 9. Uncertaintiesin AG range from
60 to 250 calories,depending upon temperatureand
uncertainty in log K. AG values are positive for all
conditions, and steadily increase in value with increasingtemperature. The value at 25"C and I bar
agrees with Parker et al. (1971) but disagreeswith
Latimer et ql. (1933) and Latimer (1952) (seeTable
8).
Eualuationof L,V, AH, and AS
The calculationof other thermodynamic quantities
requiresevaluation of the following relationships,
BLOUNT: BARITE SOLUBILITIES
Table 8. Comparison of thermochemical data for dissolution of
barite at 298.15'K and I bar
Source
Aao^
ACo-
of
kcal/rcle
+13.6
I 0.2
Parker et af.
(1971)*
aso-
EH
kcal/m1e
data
caUrcle
+5.46
+6.3
to.25
Latiner et a1.
(1933)
K
-zc.)
i 0.45
-27.7
Latl.mer et a1.
( 1 9s 2 )
+ 1 2, 0 6
+4.63
- 2 4. 5
Present study
(Delta Process)
+I3.6
! 0.2
+6. 35
Equatlon 3
Table L0
+13.61
+ 5 . 03
-24.3
r 3.5
-28.8
*Uncertainty
fron
Parker
=\. z
as a personal
comunlcation
(6)
-R[a ln K/a(l/T)lp : LH, and
-QLG/aT)p : R@ tn K/07)p
*RlnK:A,S.
(7)
AV = Vsa++ *
(8)
QLG/aP)r : -RT(a ln K/7P)r : AV
tla(LG/Dl/la(t/rt]tP :
(5)
with almost an equal degree of fitness. Calculated
vafues for LV are illustrated in Figure 7. Values are
markedly negativeunder all conditions. A maximum
occurs near 75oC. Quadratic equation values are
more negativethan linear equation values.Also, values obtained from quadratic equations all showed
that A Z increasedwith pressure,as illustrated by the
1000 bar curve in Figure 7. Uncertaintiesin the AZ
values are estimatedto be about + l0 percent of the
value.
For comparison, A Z values were also calculated
from the molar volumes of barite (Robie et al.,1967),
corrected for thermal expansion(Skinner, 1966) and
the partial molal volumes for the ions (Ellis, 1967,
1 9 6 8 ;a n d M i l l e r o , 1 9 7 2 )u s i n g t h e e q u a t i o n ,
The data for log K and AG can be modeledby many
differentempiricalequations(e.g.equations3,4, and
5 in Table l0). Valuesfor AV, AH andAS arerepresentedby the appropriatefirst derivativesof these
equations.The valuesobtained,however,depend
upon the type of model equationused.At present
a particular
thereis no theoreticalbasisfor selecting
modelequationoverothers.
Volumechanges.A Z valueswere obtainedby differentiating
both linearand quadraticequationsthat
modelledisothermalvariationsof AG with pressure
Vss^ -
Vbarrte
rc)
Resultsof thesecalculationsare shown by solid dots
in Figure 7. Below 150'C, values obtained from
linear equationsare closerto those basedon data for
ions, whereasvalues obtained from quadratic equations are closer at higher temperatures.The solid
curve in Figure 7 representsAZ values calculated
from an empirical equation (equation la in Table l0)
basedupon data for ions up to 200'C and the values
obtained from quadratic equations above this temperature.The valuesobtained from this equation are
within uncertaintyestimatesof Ellis (1967, 1968).AV
values at 1000bars were obtained by taking the differencebetweenthe l- and 1000-barvaluesobtained
from the quadratic equations,and adding this differenceto the l-bar valuescalculatedfrom the empirical
e q u a t i o n .S i n c e m o s t o f t h e 1 0 0 0 - b a rv a l u e s o f L V
fall 15 percent above the l-bar values, they differ
T a b l e 9 T h e r m o d v n a m i c o u a n t i t i e sf o r t h e s o l u t i o n o f b a r i t e a t I b a r
Temperat ure
AGO-
K
kcal
mle
298,L5
333.15
3 5 3 .L 5
3 73 . L 5
13.61
t4.74
1 5 .5 6
16.37
3 9 8 .t 5
448.15
4 7 3. 1 5
t7 .49
18.84
20.39
22.L2
498.ls
23.99
JZ5.
t q
425. L)
LJ
573.15
*Calculated
**Calculaled
AHo
R
n
{e
30.00
kcal
mole
2.97
1.04
- 0.89
- 3.30
-
).lZ
- 8.13
-10.55
-L2.96
-r5 .37
-20.20
^^o
6
n
cal
nole
mole
-24.3
-35.3
- 4 I . I
-46,2
-52,2
-58.0
-bJ.
b
-69.0
-L.24
-1 no
-1, 11
-1. 18
-I-. 35
-r. 58
-1.87
-2. ZO
-74.2
-2,58
-2.98
-6t.5
- ?
using data for ions given by Criss and Cobble
by dlfferentiation
of equation 3 ln Table 10.
R7
(1964a,b).
^,?o-*
'E
nHo **
kcal'
mole
nole
6.3
J . J
0.0
E
).u
2.7
1.0
- 0.5
l,qo *
fr
ca1
rc1e
- 2 4. 5
-34.0
-43,)
- 2.6
- 4.0
-
- 8.9
- 7.7
-10.5
-L4.4
-20.4
-L6 .7
-23.8
qn
- 6 4. 6
953
BLOUNT: BARITE SOLUBILITIES
significantlyfrom A V at 1 bar . One calculationof A I/
at i000 bars and 25'C, using partial molal ionic
c o m p r e s s i b i l i t i easn d e q u a t i o n so f O w e n a n d B r i n k l e y ( 1 9 4 1 ) ,i s 2 4 p e r c e n ta b o v e t h e I b a r v a l u e .
Heats of reaction. Differentation of empirical equations such as 3 and 4 in Table l0 which model log K
or AG variation with temperature(equations6 and 7
in this section),producesA/1 and AS values at25"C
that do not agree with values calculated from standard-state data of Parker et al. (1971). AH values
were therefore calculatedusing a delta processbased
upon text equation 6. Differencesbetweenvaluesfor
LG/7, (-RlnK), for successivedata points listed in
Table 6 were divided by the differencein l/T values.
The Atl values at mean temperaturesshown on Figure 8 do not fall along simple smooth curves.Uncertainties are large, and linear equationscalculatedfor
l, 100,500, and 1000 bars fit the data within the
uncertaintylimits shown. The terms of the four equations varied almost linearly with pressure,and the
four equationswere combined to produce equation 2
(Table 10), from which Atl valuesmay be calculated
for desired temperatures and pressures.The solid
lines of Figure 8 are basedon valuescalculatedfrom
this equation. A closer reproduction of the data
points in Figure 8 by higher-order equations is not
T a b l e 1 0 . E q u a t i o n sf o r t h e r m o d y n a m i cq u a n t i t i e sa n d p a r a m e t e r s
f o r D e b y e - H i i c k e le q u a t i o n
L. ,V, o- " = - 1 . 3 7 2 2 x r, 0^ -5?- - 1 - 1 . 2 0 3 9 L T + 7 6 7 . 5 8
^?
av,
(rb)
cm
-t.rt,
ufo =
,,p
av
an
R
/moae
- 2 . 8 7 8 8x t o - 2 ? + 1 8 . 3 5
" 103?-1
an caf/Doles
Dar
(2)
ttto, = 2.9614 * ro-2w - 96,594r - Lo.76g6p + 35L54
(3)
aco" = l.ttoou
A1o"
ln
cal/oole
,
Lo-4r2 - 4.02952 ' 1o-2? + 15.386
adoo in kcal/no1e
(4)
\
ro
-,.r.t
\
E
-l?O
fr
o Millero (1972)
o Lineor Equolions (aG'* vs P)
a Q u o d r o f i c E q u o t i o n s( a C ' , v s P )
A avh
Voluesol IOOO Bors
X From lonic Compressibilities
-r80
300
400
500
600
Temperoture
in K
Fig. 7. Change in volume for reaction of barite with HrO at
i n f i n i t e d i l u t i o n , L V a s a f u n c t i o n o f t e m p e r a t u r ea t I a n d 1 0 0 0
b a r s A Z v a l u e c a l c u l a t e df r o m p a r t i a l m o l a r v o l u m e so f B a S O , i n
s o l u t i o n a n d m o l a r v a l u e so f b a r i t e a r e s h o w n f o r c o m p a r i s o n( s e e
text)
justified, because of the large uncertainties in the
data. The averagedifference between the Ail values
calculated from the equation and the data points is
*l.2 kcal/mole. The Af1 values at 25oC and I bar
calculated in this study agree with At1 values obtained using standard data cited by Parker et al.
( 1 9 7 1 ) a n d P a r k e r ( p e r s o n a lc o m m u n i c a t i o n ,1 9 7 4 ) ,
as shown in Table 8. Valuesobtained from Latimer et
al. (1933) and Latimer (1952) do not agreewith those
from Parker or based on the present study.
Entropies and heat capacitiesof reaction. Values for
AS were calculatedfrom Gibbs-Helmholtz equation.
(9)
AS : AH/T + 2.3025R log K,
Log Ko =-1.45823 * 103?-11ogr + 3,5547LogI - 9.036696
-?
x l0 -?los?
Log .(o = (L.4g325 x Lo-2P-4a.67) loe|
+ (2.3s365p (6)
a) -80
o
toaz.tor-L
- 4.398 x to-2p *
a. = 3.839 + 5.155 x to'3r - t,z6s,
L
t3o.ot9
1 0 - 5 ? 2+ 1 . 4 0 1
x10-P-1.O2xlO-P?
4i
1n AngstroEs;
paraEeter
ln
eq 12
c = -0.484 + 2.376 \ to-3r - 3,33g * to-6r2 - t.tl
using log K data from Table 6 and AH values calculated from equation 2 (Table l0). Values for AS at I
bar are given in Table 9. The value at 25oC agrees
with that calculated from standard-statedata (see
Table 8).
As a further check on the calculation procedures,
equations I and 2 in Table 10 were compared, using
the following thermodynamic equations of state,
t lO 'P + 1.00 x 10 'Pr
(10)@AHlaP)r:
paraoeter ln eq 12
(ll)
ACr:
(aLV/oT)P:
L,V- T(aAVl7T)pand
-@AS/aP)r.
954
BLOUNT: BARITE SOLUBILITIES
o Present study
o Templeton1960
o Bosed on ion dolo
CrissB Cobble(19640.b)
o)
)<
o
The linear equationusedto representLH datavariation with temperatureassumesthat A.Co does not
It is knownthat A'Covalues
changewith temperature.
(Crissand Cobble,1964b;and
vary with temperature
Criss, personalcommunication,1974).The values
obtainedfrom equation 2 at eachpressurethenrepresentan averagevalueover the temperaturerange25
valueat I bar is fairly closeto
to 300'C. The average
the averageobtainedfrom valuesgivenby Crissand
ACo valincreases
risesignificantly
Cobble.Pressure
in
ues.Most of this changeis dueto changes theionic
and
Detailsof both the temperature
heat capacities.
pressuredependences
of ACo cannot be evaluated
from the solubilitybaseddata.
-
Calculationof log K from AH data
Integrationof equation2 in Table 10 with respect
to 1/T yieldsvaluesfor log K and AG. Integration
constantswere calculatedfrom the log K valuesof
Table 6 at 298'K. Sincethe integrationconstants
a singleequation
variedalmostlinearlywith pressure,
of log K was obtainedand is given in Table l0 as
equation5. Log K valuesobtainedfrom thisequation
are givenin Table7 and illustratedby dottedcurves
Tin K
in Figure 6. There is good agreementwith log K
Fig.8. Evaluation
of theheatof reactionat infinitedilution,AH,
valuesin Table 6 exceptat 60 to 100'C.The differas a function of temperatureat constantpressures
of l, 100,500,
intervalaregreaterthan the
encesin thistemperature
and 1000 bars. The open circlesrepresentvaluesobtained for
in log K values.The model,equation5
uncertainties
successive
temperatureintervalsof the function,
of the fit
in Table 10,is too simplified.The closeness
lLG,r",/T - AG,r,,/71/( / T, - | /T),
showsthat log K calculations
at most temperatures
plottedat the averagetemperature.
The lengthof verticalbar gives arenot verysensitive
to the valuesof ACp.Valuesfor
thelncertainty in the calculation.Referto text for descrintionof
successive
differentiationof equaACp obtainedby
which
4
in
Table
10,
modelthe log K and
tions 3 or
AG data very well, differ markedlyfrom thoseobValuesfor ACs calculatedfrom equationsI and 2 in
tainedfrom Crissand Cobble(1964b)and Criss(perTable 10 are in good agreement,
considering
the unsonal communication,1974). However, valuesof
certainties
in the data.The crossderivatives
in equaare generally
AC, obtainedby such differentiations
tion I I werealsofound to bewithin acceptableagreelittle betterthan guesses.
ment.
Valuesfor All and AS from 25 to 300oCwerealso
calculated,using data for ionic entropiesand heat Actiuity cofficientsfor BaSOn(aq)
givenby Crissand Cobble(1964a,1964b), Barite solubilitiesin salt solutionsand the precapacities
data for baritegivenby Kelley(1960),standarddata viously-determined
equilibriumconstantswereused
for ions givenby Latimer(1952),and standard-state to calculatelogarithmsof the mean activity codatagivenby Parkeret al. (1971).The resultsofthese efficient,7-, for BaSOoin solution, using text
calculations
are givenin Table9. AH valuesat I bar equation2. Baritesolubilitiesin 0.2, l, and 4 molal
are also shownin Figure8. The agreement
between NaCl solutionsfrom Table 5, the valuesof log K at
the setsof AH and A^Sdatain Table9 is good.Most thetemperature
and pressure
specified,
and thecalcuvaluesare within estimateduncertaintyranges.
lated log 7. valuesare given in Table 11. Barite
Values for ACo obtained by differentiationof solubilitiesat 25oC were obtained from Puchelt
equation2 in Table l0 are markedlynegative(-96 (1967,Fig. 2). Log 7- valuesbecomemore negative
cal/moleoK at I bar). Uncertainties
are very large. with increasing
NaCl conor increasing
temperature
955
BLOUNT.' BARITE SOLUBILITIES
Table I l. Measuredand calculatedactivity coefficients
for BaSOoin NaCl-HrO solutions
Toc
25
p t
gn/Cnr
0.2
0. 0 3 7 *
1. 0 1 0
9. 9 8
-u.50
,r
0.079
0.LLz
0.082
0.r18
0.068
0.108
0.055
0.087
0.966
0.988
0.924
0.951
0.875
0.905
0.823
0.858
9 .5 9
9.22
9.73
9.32
10.2L
9.75
r0.76
r0.24
-0.69
-0.66
-0.78
-0.73
-0.94
-0.91
- 1 .r 0
-r .06
1.0
0.077*
1.036
9.98
-0.91
0.772
0.233
0.220
0.297
0.232
0.340
0.240
0.350
0.997
t .016
0.957
0.981
0.909
0.956
0.849
0.887
9.59
9.22
9.73
9.32
IO.zL
9.75
10.76
10.24
NaCL
Molal
L
r00rfi
,'
500
150s"
,'
500
200
15
500
"
tqn
BaSO4
MilliMoIal
P
bars
?a
"500[
25
r00
'r
150
"
200
"
250
,'
I
I
500
5
500
15
500
58
s00
-1og Ko
Eq (2)
log yn
Es, (2)
1og yn
Eq (12)
-I.UJ
-0 .98
-I.2L
-r.15
-1.41
-7.76
-t.66
p
i
x10"Cn
4.25
-0.073
-0.68
-0.6s
-0.79
-0.75
-0.94
-0.89
-1.13
-1.06
4.00
4.r4
3.76
3.82
3.45
3.44
3.07
2,99
-0.062
-0.052
-0.076
-0.064
- 0 .r 0 7
-0.092
- 0 .1 5 3
-0.737
0.88
4.25
-0.073
-r .04
-0.98
-I.20
-1.14
-1.45
-1.58
-1.80
-L.70
4.00
4.74
3.76
3.82
3.45
3.44
3. 0 7
2,99
-0.062
-0.052
- 0 .0 7 6
-0.064
-0.107
-0.092
- 0 .1 5 3
-0-137
centration.Increasingpressurecausesvaluesto bebaritesolubilityvariationwith
Curvesrepresenting
comelessnegative.
NaCl concentration
in Figures4 and9 are basedon
The solubility-based
log 1- valueswerecompared solubilityvaluescalculatedusingequation 2, log 7*
with valuescalculatedfrom text equation3, an ex- from equation12 andlog K from Table6. A marked
tendedform of the Debye-Hiickelequation.Values discrepancy
existsbetweensolubilitymeasurements
obtainedfrom equation3 disagreedsystematically at 25oC of Puchelt(1967)and those by Neuman
with those obtainedfrom equation2, Equation 3 (1933,in KCI solutions),Templeton(1960),and
assumesthat the mean ion-sizeparameterdoesnot Striibel(1967).Puchelt'sresultsagreewith thosecalchangewith temperatureand pressure.Malinin et al. culatedfrom equation12.To fit the solubilitydataof
(1969)variedthec1valuesin their calculations
of log
K at high temperature.
However,the useof their a,
valuesresultedin an overcorrection.
Also, variation
of the ai parameteralonedid not resultin a satisfac- N
r
tory correction.In a studyof the thermodynamics
of
ct
rbo{ ^
CaSOn.2H2Oin NaCl solutions,Marshall and Y t o
l^c'
o
8
--/
Slusher(1966)varied the product B^a1 and added o
?9'" .
c)
termsto the Debye-Htickel
equationthat werepro- o . o 6
portional to the ionic strengthand the squareof
Q4
the ionic strength.It was found necessary
o
to add a
o3
^,/
a N e u m o n ( 1 9 3 31 i n
term linearwith respectto ionic strengthto equation c o 2
o
r T e m p l e t o n( 1 9 6 0 )
produce
3 to
the equation,
^$
(1967)
I
I
E
(t 2 )
a Strribel
o Puchelt
a
l o s ^ y ^ :? l Z *Z -l A ^ fi )/
(l*B-a1{7*1
I
+CI*
in which ai and C are temperature- and pressuredependentparameters.Log 7* values calculatedusing equation 12 and the appropriate as and C- parameter valuesare listed in Table I l. Equations for calculating at and C are given as equations 6 and 7 in
Table 10.
(n or
00
(1967)
t\/l
'''(NoCl)
.?
|
or?3.4567
tl
-(ttoCt)
-
h
/
t t
,
l+
lTl-1106;1
Fig,9 Baritesolubilityin NaCl-HzOsolutionsat25oC,I bar,as
solubilities
The curverepresents
a functionofNaCl concentration.
Debye-Hiickel
equation
usingan extended
baseduponcalculations
(referto text).
BLOUNT: BARITE SOLUBILITIES
other workers, 41 in equation 12 would have to be
near 2 X l0-8 cm, which does not agree with Kielland's value (1937). Furthermore, an abrupt change
in the temperature-dependence
of the ion-sizeparameter ai would be required.Therefore,\thesolubility
measurementsby Neuman, Templeton, and Striibel
in salt solutions are probably too large. Templeton's
(1960) data were used to calculate log K and AG
values at 25"C (Parker, personal communication),
and this value is based upon extrapolation of data
that may not represent the solubility of coarselycrystalline barite. The AG value may still be correct
within the uncertainty limit given by Parker (see
Table 8).
Conclusions
Barite solubilities of this study have been compared with previous determinations and have also
been used to calculatethermodynamic quantities for
the dissolution of barite in aqueous solutions.There
is considerabledisagreementbetween the results of
this study and most previous studies.Barite solubilities of this study were made using a pure, coarselycrystalline barite under reproducible and reversible
conditions. Presentresultsshow smoother variations
with temperature,pressure,and NaCl concentration
than do most previous results.An additional important observation that seems to confirm the greater
accuracyof the presentsolubility measurementsis the
close agreementbetween thermodynamic quantities
calculatedfrom thesesolubilitiesand those calculated
from thermal and solution density data. Barite solubilities of this study are compatible with the standard-state quantities reported for barite and BaSOn
in solution (Parker et al., l97l). Although uncertainties are rather large, thermodynamic quantities
calculated from the solubility data at temperatures
from 25 to 300'C agreed with those obtained from
the literature for partial molal volumes, enthalpies,
entropies, and heat capacities.Activity coefficients
obtained from barite solubilities in NaCl solutions
and solubility-based log K values were accurately
modeled by an extended form of the Debye-Hiickel
equation up to high NaCl concentrations.Most of
the previous solubility data produce calculatedthermodynamic quantities that differ significantly from
those basedon thermal measurements.The resultsof
those thermodynamic calculationssupport the accuracy of the solubilitiesof this study and those of the
studies by Puchelt (1967) and Uchameyshvilli et al.
( 19 6 6 ) .
The solubility of barite is very low under all experi-
mentalconditionsexaminedin the presentstudy.No
can be predictedfrom solubilitydata or
mechanism
the thermodynamiccalculationsthat would significantlyalterthe solubilityrelationssothat appreciable
BaSOrcanbe carriedin hydrothermalsolutions.The
most probablecauseof BaSOrdepositionis the oxito sulfatein solutions
dationof reducedsulfurspecies
carryingbarium.
Acknowledgments
This study was supported by NSF grants GA-1667 and GA3 5 6 2 3 .T h e a u t h o r g r a t e f u l l y a c k n o w l e d g e sa s s i s t a n c ei n t h e s o l u b i l i t y m e a s u r e m e n t sb y M r . H a r o l d M o r i t z a n d M r s . M e l i n d a
G u i l l . C o m m e n t s a n d d i s c u s s i o n sw i t h P r o f e s s o r G e o r g e B o y d
were especially helpful in working on the thermodynamic section
of the paper. Assistance with programming the Hewlett-Packard
M o d e l 9 8 2 0 4 w a s p r o v i d e d b y M r . J a m e s S p a u l d i n g .T h e a u t h o r
also thanks Dr David Wenner and Dr. Frank Dickson for their
c o m m e n t s a n d s u g g e s t i o n sr e g a r d i n gt h e m a n u s c r i p t .
References
|. Z. anorg.allg.
Balarew,D. (1925)L6slichkeitund Korngrdsse.
Chem..145. 122-126.
to
Blount,C. W. (1971)Baritesolubilityin waterat temperatures
250"C and pressuresto 1000bars. Geol.Soc. Am Abstr. Programs.,3,508
(1974)Synthesis
anglesite,
witherite,and
of barite,celestite,
lrz. Mineral, 59,1209-1219.
in aqueous
solutions.
strontianite
-,
R. E. Channelland D. E. Leyden(1971)An improved
techniquefor the preparationof pelletsfor X-ray spectrographic
Anal.Chim.Acta,56,456-458.
resins.
analysis
on ion-exchange
(SrSOl)
n1d F. W. Dickson( 1967)The solubilityof celestite
in H,O from 50' to 250"Cand 100to 1500bars.Geol.Soc.Am.
Abstr for 1966,19-20.
(1969)The solubilityof anhydrite(CaSOr)in
s1d NaCl-H,O from 100to 450oCand 1 to 1000bars. Geochim.
Acta, 33, 22'7-245.
Cosmochim.
-,
D. E. Leyden,T. L. Thomasand S. M. Guill (1973)
Applicationof chelatingion exchangeresinsfor traceelement
Anal.
analysisof geologicalsamplesusingX-ray fluorescence,
C h e m. 4 5 . 1 0 4 5 - 1 0 5 0 .
reserves,
andfuture
Brobst,D. A. (1970)Barite:world production,
prospects.U. S. Geol.Suru.Bull. 1321
propCriss,C. M. andJ. W. Cobble(1964a)The thermodynamic
aqueoussolutions.IV. Entropiesof
ertiesof high temperature
principle.J. Am.
the ions up to 200oand the correspondence
Chem.Soc..86. 5385-5390.
(1964b)The thermodynamic
properties
of high
and V. The calculation
of ionicheat
aqueous
solutions.
temperature
above200o
capacitiesup to 200o.Entropiesand heatcapacities
J . A m C h e m] o c . . 8 6 . 5 3 9 0 - 5 3 9 3 .
D i c k s o n ,F W , C . W . B l o u n t a n d G . T u n e l l( 1 9 6 3 ) ,U s e o f
the solubilityof
solutionequipment
to determine
hydrothermal
anhydritein waterfrom 100"CIo 275"Cand from 1 bar to 1000
Am. J Sci.,261,6l-78.
barspressure.
Ellis,A. J (1967)Partialmolal volumesof MgCl,, CaCl,,SrCl,,
and BaCl, in aqueoussolution to 200o.J. Chem Soc. (A),
660-664.
(1968) Partial molal volumesin high-temperature
water.
BLOUNT: BARITE SOLUBILITIES
Part III. Halide and oxyanion salts. "/. Chem. Soc. (A),
I 1 3 8 - l1 4 3 .
Fraps, G S. (1902) The solubility of barium sulphate in ferric
chloride, aluminum chloride, and magnesium chloride. Am.
Chem J ,27,288-291.
F r e s e n i u s ,R . a n d E . H i n t z ( 1 8 9 6 ) U e b e r e i g e n t h i . i m l i c h e
LdslichkeitsverhZiltnissedes schwefelsaurenBaryts. Z. Anal. Chem., 35,
170-183
Gallo, G. (1935) Equilibrio fra solfato distronzio ed acqua alle
varie temperature Ann Chim. Appl., 25, 628-631.
Gundlach H., D. Stoppel and G. Striibel (1972) Zur hydrothermalen Ldslichkeit von Baryt. NeuesJahrb. Miner. Abh., 116,
3 2I - 3 3 8 .
H a a s , J . L . ( 1 9 7 0 )A n e q u a t i o n f o r t h e d e n s i t yo f v a p o r - s a t u r a t e d
NaCl-HrO solutions from 75o to 325'C. Am. J. Sci., 269,
489-493.
(1971) The effect of salinity on the maximum thermal
gradient of a hydrothermal system at hydrostatic pressure.Ecor.
Geol..66.940-946.
Heinrich, E. Wm (1966) The Geology of Carbonatites. Rand
McNally, New York.
H e l g e s o n ,H . C . ( 1 9 6 9 )T h e r m o d y n a m i c so f h y d r o t h e r m a l s y s t e m s
at elevated temperatures and pressures. Am. J. Sci., 267,
729-804.
H e r o v s k y ,J a n d S . B e r e z i c k y( 1 9 2 9 ) T h e d e p o s i t i o no f r a d i u m a n d
other alkaline earth metals at the dropping mercury kathode.
Coll Czeckoslouaki Chem. Comm.. l.19-46.
H o l s e r , W . T a n d G . C . K e n n e d y ( 1 9 5 8 ) P r o p e r t i e so f w a t e r p a r t
IV: Pressure-volume-temperature relations of water in the range
4 0 0 - 1 0 0 0 ' Ca n d 1 0 0 - 1 4 0 0b a r s .A m . J . 9 c i . , 2 5 7 , 7 1 - ' 7 7 .
Kelley, K. K. (1960) Contributions to the data on theoretical
m e t a l l u r g y .X I I I : H i g h - t e m p e r a t u r eh e a t - c o n t e n t h
, eat capacity,
a n d e n t r o p y d a t a f o r t h e e l e m e n t sa n d i n o r g a n i c c o m p o u n d s .
U S Bur. Mines 8u11.,584.
K e n n e d y ,G . C . , W . L . K n i g h t a n d W . T H o l s e r ( 1 9 5 8 )P r o p e r t i e s
o f w a t e r p a r t I I I : S p e c i f i cv o l u m e o f l i q u i d w a t e r t o l 0 0 o C a n d
1 4 0 0b a r s A m . J . S c i . , 2 5 6 , 5 9 0 - 5 9 5 .
K i e l l a n d f . ( 1 9 3 7 )I n d i v i d u a l a c t i v i t y c o e f f i c i e n t so f i o n s i n a q u e o u s
s o l u t i o n s J. . A m C h e m .S o c . 5 9 . 1 6 7 5 - 1 6 7 8 .
Latimer, W. M. (1952) Oxidation Potentials; the Oxidation States
of the Elements and Their Polentials in Aqueous Solutions,2nd
E d . P r e n t i c eH a l l , I n c . , N e w Y o r k .
-,
J. F. G. Hicks, Jr. and P W Schutz (1933) The
heat capacities and entropies of calcium and barium sulfates
from I 5 to 300'K The entropy and free energy of sulfate ion.
"/
Chem. Phys , 1, 620-624
L e m a r c h a n d s ,M . ( 1 9 2 8 ) S € p a r a t i o nq u a n t i t a t i v ed u b a r y u m e t d u
calcium. C. R. Acad Sci. Paris, 187, ffil-603.
L i e s e r ,K . H ( 1 9 6 5 ) R a d i o c h e m i s c h eM e s s u n gd e r L d s l i c h k e i tv o n
Erdalkalisulfaten in Wasser und in Natriumsulfatl<isungen. Z.
anorg. allg. Chem , 335,225-231.
M a g e r , H . a n d K H L i e s e r ( 1 9 7 2 ) R e k r i s t a l l i s a t i o nu n d A l t e r u n g
v o n S t r o n t i u m s u l f a t ,I I . R e k r i s t a l l i s a t i o nu n d A l t e r u n g n a c h d e r
Fiillung aus verdtinnten Lrisungen. Z. Phys. Chem. Neue Folge,
81.8l-88.
M a l i n i n , S D . , N . Y e . U c h a m e y s c h v i l ia n d N . I K h i t a r o v ( 1 9 6 9 )
Application of the theory of strong electrolytes to the solubility
of barite in aqueous metal chlorides under hydrothermal conditions. G eokhimiya, 927 -938.
M a r s h a l l , W . L a n d R . S l u s h e r( 1 9 6 6 ) T h e r m o d y n a m i c s o f c a l c i u m s u l f a t ed i h y d r a t e i n a q u e o u ss o d i u m c h l o r i d e s o l u t i o n s , 0 ll0" . J. Phys. Chem., 70, 4015-4027.
957
Melcher, A. C. (1910) The solubility of silver chloride, barium
sulphate and calcium sulphate at high temperalure.J. Am Chem.
Soc , 32,50-66
M i l l e r o , F . J ( 1 9 7 2 ) T h e p a r t i a l m o l a l v o l u m e s o f e l e c t r o l y t e si n
aqueous solutions. In R. A. Horne, Ed., LTater and Aqueous
Solutions, p 519-595. Wiley Interscience.
M o r e y , G . W . a n d J . M . H e s s e l g e s s e( 1r 9 5 1 )T h e s o l u b i l i t y o f s o m e
minerals in superheatedsteam at high pressures.Econ. Geol.,46,
8 2l - 8 3 5 .
Neuman, E. W. (1933) Solubility relations of barium sulfate in
a q u e o u ss o l u t i o n s o f s t r o n g e l e c t r o l y t e sJ. A m . C h e m . S o c . , 5 5 ,
879-884.
O w e n , B . B . a n d S . R . B r i n k l e y , J r ( 1 9 4 1 )C a l c u l a t i o n o f t h e e f f e c t
of pressureupon ionic equilibria in pure water and in salt
s o l u t i o n s .C h e m . R e u . , 2 9 , 4 6 1 - 4 7 4 .
Parker, V. B., D. D. Wagman and W. H. Evans (1971) Selected
values of chemical thermodynamic properties. Natl. Bur. Stand.
Tech. Note 270-6.
Puchelt, H. (1967) Zur Geochemie des Bariums im exogenen
Zyklus. S it zungsber. H eidelberger A kad. W issenschaft en, M alh.naturw issenschaft. Klasse
Q u i s t , A . S . a n d W . L . M a r s h a l l ( 1 9 6 5 ) E s t i m a t i o no f t h e d i e l e c t r i c
constant of water to 800o. J Phys. Chem., 69, 3165-316'7.
R a d k e , A S a n d G . E . B r o w n ( 1 9 7 4 ) F r a n k d i c k s o n i t e ,B a F r , a
new mineral from Nevada.Am. Mineral..59, 885-888.
Reuised List of Physical Constants (1971) Natl. Bur Stand Tech.
NewsBull.,7l-72
R o b i e , R . A . , P . M . B e t h k e a n d K . M . B e a r d s l e y( 1 9 6 7 ) S e l e c t e d
X - r a y c r y s t a l l o g r a p h i cd a t a , m o l a r v o l u m e s , a n d d e n s i t i e so f
minerals and related substances.U. S. Geol. Suru. Bull., 1248.
Roedder, E. (1972) Composition of fluid inclusions. In M.
Fleischer, Tech. Ed., The Data of Geochemistry. U. S. Geol.
Suro Prof. Paper 440-JJ.
Rowe, A M. and J. C. S. Chou (1970) Pressure-volumet e m p e r a t u r e - c o n C e n t r a t i o rne l a t i o n o f a q u e o u sN a C l s o l u t i o n s .
J. Chem Eng Data, i,5,6l-66.
S i l l e n , L G . a n d A . E , .M a r t e l l ( 1 9 6 4 ) S t a b i l i t y c o n s t a n t so f m e t a l ion complexes. Chem. Soc. Lond., Spec, Pub. 17.
( 1 9 7 1 ) S t a b i l i t y C o n s t a n t s , S u p p l e m e n tN o L
snfl Chem. Soc. Lond. Suppl. I to Spec Pub. 17.
Skinner, B. J. (1966) Thermal expansion. In S. P. Clarke, Ed,
Handbook of Physical Constants. Geol. Soc Am. Mem. 97,
75 - 9 6 .
S t r t i b e l ,G . ( 1 9 6 6 ) D i e H y d r o t h e r m a l e L i i s l i c h k e i t v o n C i i l e s t i n i n
System SrSO,-NaCI-H,O. Neues Jahrb. Mineral. Monatsh.,
99- I08.
( 1 9 6 7 ) Z u r K e n n t n i s u n d g e n e t i s c h e nB e d e u t u n g d e s S y s tems BaSOr-NaCl-HrO. Neues Jahrb. Mineral. Monatsh.,
223-234.
-
( 1 9 6 8 ) H y d r o t h e r m a l e L d s u n g e n , e x p e r i m e n t e l l eU n t e r suchungsergebnisseiiber hydrothermalsynthetische Ldsungen
bis 600"C und 2000 Bar. Geol. Rundsch.. 58.259-273.
Templeton, C C. (1960) Solubility of barium sulfate in sodium
chloride solutions from 25' to 95oC J. Chem Eng. Data, 5,
5l 4 - 5 I 6 .
U c h a m e y s h v i l i ,N . Y e . , S D . M a l i n i n a n d N . L K h i t a r o v ( 1 9 6 6 )
S o l u b i l i t y o f b a r i t e i n c o n c e n t r a t e dc h l o r i d e s o l u t i o n s o f s o m e
metals at elevated temperatures in relation to problems of the
genesisof barite deposits Geokhimiya, I 193-1205.
Manwcript
receiued, October 23, 1974; occepted
for publication, February 18, 1977.