Relative Risk
advertisement

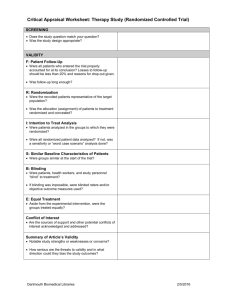
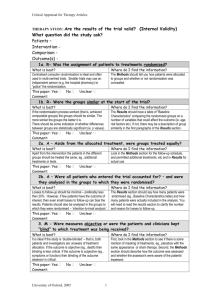
Continuing Professional Pharmacy Development (CPPD) Program Title Putting Evidence in to Practice David J Woods, Consultant Pharmacist; Managing Editor New Zealand Medicines Formulary November 28, 2012 Disclaimer: PRESENTING AUTHOR HAS NO RELATIONSHIPS TO DISCLOSE 1 Workshop Learning Outcomes By the end of this session, participants will be able to : 1. Discuss how clinical trial results are reported in scientific papers, the media, and by the pharmaceutical industry 2. Demonstrate competence in estimating the benefits and risks of treatment in numerical terms (NNT – number needed to treat; NNH – number needed to harm) 3. Assess the benefits and risks of treatment in patients at different levels of baseline risk – the concept of PEER – patient expected event rate 4. Discuss the application of clinical trial results in to practice – the concept of generalizability 2 How benefits and harms are expressed • Clinical trials • Media • Advertising 3 Relative Risk • Risk is: no. events/no. people exposed • Risk is a RATE or event rate • 5 heart attacks per 100 risk is 5/100 or 5% or 0.05 • NB event rate can have positive or negative implications 4 Relative Risk • Compares the risk (event rates) of two populations as a ratio • Event rate in control population: Control Event Rate or CER • Event rate with experimental (exposed) population: Experimental Event Rate or EER • Relative Risk (RR) = EER/CER 5 Relative Risk – example 1 • Event rate (MI) with drug A = 5/100 EER = 5% or 0.05 • Event rate in non-exposed controls is 10/100 CER = 10% or 0.1 • RR = EER/CER or 0.05/0.1 RR = 0.5 or 50% • Event rate with A is 50% of that with control 6 Relative Risk example 2 • EER with B is 0.04 • CER is 0.1 • RR = 0.04/0.1 RR = 40 % or 0.04 • Event Rate with B is 40% of that with control 7 Relative Risk Reduction (RRR) • Often more intuitive to express by how much risk is reduced compared to control or background risk • RRR = CER – EER (Risk Difference)/CER • Example 2 RRR = 0.1 – 0.04/0.1 = 0.06/0.1 RRR = 0.6 or 60 % 8 Relative Risk Reduction (RRR) 2 • In example 2 the risk of an event is reduced by 60 % • RRR is also 1 – RR In this case 1 – 0.4 = 0.6 or 60% • RRR is often assumed to have a linear relationship with time; E.g. RRR of 60% over three years is 20% per year 9 RR and RRR are only RELATIVE • Example 1 RR is 0.5 and RRR is 50% 5/100 divided by 10/100 • But; 5/1000 divided by 10/1000 is also 0.5 or 50% • RR and RRR give no indication of the effect in absolute terms 10 Absolute Risk Reduction (ARR) • Risk difference, i.e. difference between CER and EER • Example 2 ARR is CER – EER ; 0.1 – 0.04 ARR is 0.06 or 6% • In absolute terms risk is reduced from 10% to 6% 11 ARR examples • First case CER = 10%, EER is 6% RR 0.6, RRR is 0.4 Can say RR is 60% and risk reduced by 40 % • ARR (CER – EER) is 4% (large impact on population ?) 12 ARR examples • Second case CER = 1%, EER is 0.6% RR 0.6, RRR is 0.4 Can also say RR is 60% and risk reduced by 40% • ARR is 1% - 0.6% = 0.4% (less impact at population level than first case, but relative figures the same) 13 Number Needed to Treat • Number needed to treat to gain one benefit • E.g. if ARR is 1% need to treat 100 patients to gain one benefit • If ARR is 0.1% need to treat 1000 patients to gain one benefit • NNT is 100/ARR e.g. 100/1% or 1/0.01 Care with above ! 14 Real Example • Fracture intervention trial Black et al.Lancet 1996;348:1535-41 • 2027 post-menopausal women treated with alendronate or placebo • Reduction in hip fracture was 49% In advertising this would look impressive ! 15 Fracture Intervention trial • CER is 2.19; EER is 1.08 • RR = 49 % Can say fractures reduced by 50% Impressive ! • Absolute Risk Reduction ARR = 1.11% NNT is 100/1.11 or about 90 More realistic 16 More on NNTs • Not the perfect solution, e.g. difficult to compare NNTs from different trials • Time factor • Numbers Needed to Harm (NNH) exactly the same concept • NNT/NNH ratio should give a benefit to harm estimate; see www.thennt.com • 17 18 19 20 Varenicline and CV problems 21 Varenicline and CV problems • Taking Chantix was associated with an increased risk of serious heart problems. These events were reported in 52 people out of 4,908 (1.06%) who took Chantix, compared with 27 of the 3,308 participants (0.82%) who took the placebo. • ARI = 1.06 – 0.82 = 0.24 NNH = 100/0.24 = approx. 400 22 Other considerations • Study population – Already at CV risk • Benefits of varenicline – NNT of about 10 cf NNH of 400 23 Benefits and harms in advertising • RR or RRR never ARR or NNT • Benefits usually exaggerated • Harms usually minimised • Critically appraise – Do your own calcualtions 25 “Proven to significantly reduce Vertebral fractures in just one year” Reduces new vertebral fractures by up to 65% 52% risk reduction 65% risk reduction Benefits and Harms in Clinical Trials • Usually RR, RRR or Odds ratios • ARR should be easily accessible • Some now report NNTs • Subject to bias • You often have to do some work from the data to get a clearer picture 28 Cardiovascular Harm and NSAIDs DB/FM 27 Mar 2012 Drug Summary Relative Risk for Cardiovascular Event (95% CI) Rofecoxib ≤ 25 mg 1.33 (1.0 – 1.79) Rofecoxib > 25 mg 2.19 (1.64 – 2.91) Celecoxib 1.06 (0.91 – 1.23) Diclofenac 1.40 (1.16 – 1.70) Naproxen 0.97 (0.87 – 1.07) Piroxicam 1.06 (0.7 – 1.59) Ibuprofen 1.07 (0.97 - 1.18) Meloxicam 1.25 (1.00 – 1.55) Indomethacin 1.30 (1.07 – 1.60) 29 Problems 1 and 4 • Group discussions • Report back answers and discuss issues arising 30 Problem 1 • A RR = 1.9/3.9 = 0.48 RRR = 0.52 ARR 3.9 – 1.9 = 2%; NNT = 100/2 = 50 • Second case RR = 0.19/0.39 = 0.48 RRR = 0.52 ARR 0.39 – 0.19 = 0.2%; NNT = 100/0.2 = 500 31 Problem 1 B • ARR = CER – EER = 9.6 – 2.8 ARR = 6.8 % NNT = 100/6.8 = 15 over 5 years RRR = ARR/CER RRR = 6.8/9.6 = 0.7 or 70% 32 Problem 4 a quick appraisal 1 • ARR warfarin cf aspirin (BENEFITS) 20 – 16.7 = 3.3 % NNT 100/3.3 = 30 For every 30 patients treated with warfarin instead of aspirin over 4 years one patient will benefit 33 Problem 4 (Harms) • Warfarin cf Aspirin (Harms) • Bleeds; Warfarin 0.62% per year vs 0.17 per year with aspirin • Over 4 years 2.48% vs 0.68 ARI 1.8 % over 4 years; NNH 100/1.8 = 55 34 Problem 4 (benefits:harms) • NNT = 30 • NNH = 55 • Unacceptable benefit:harm ratio • Discussion: Statins ? Aspirin for primary CV prevention ? 35 Consideration of baseline risk – the PEER • For statins RRR for CV events is about 30 % across all populations (NB a generalisation for demonstration) • If CV risk is 20% over 10 years then this is reduced to 14 % with statin • If CV risk is 5% over 10 years then this is reduced to 3.5 % with statin 36 Compare absolute benefits • CV risk 20 % ARR = 20 – 14 = 6%; NNT is 100/6 = 17 • CV risk 5 % ARR = 5 – 3.5 = 1.5%; NNT is 100/1.5 = 67 • Benefits much greater in patients with higher baseline risk 37 Aspirin vs Warfarin in non-valvular AF • Baseline risk can influence drug choice • Risk of AF and stroke increase with age • Other factors can affect stroke risk • RRR for stroke prevention; warfarin vs aspirin about 40 % 38 39 Baseline stroke risk and treatment choice See http://www.bpac.org.nz/magazine/2009/february/warfarin.asp 40 What is the PEER • Patient Expected Event rate • Baseline risk or CER • By applying RRR to a patient with known PEER can assess benefit of treatment in an individual • Algorithms available to estimate NNTs from PEER and RRR 41 Problems 2 and 3 • Influence of baseline risk • More on benefits vs harms • Discuss and report back 42 Problem 2 – patient S • S has PEER of 1% per year or 3 % over three years • X has RRR of 33% over three years • ARR = 3% - 2% = 1% ; NNT = 100 43 Problem 2 – patient A • A has a PEER of 10% per year or 30 % over 3 years • X has RRR of 33% over three years • ARR is 30 – 20% = 10%; NNT = 10 44 Drug X harms • Severe GI bleed 0.1% per year or 0.3% over three years • Drug X increases risk of bleed by three times to 0.9% over three years • ARI = 0.9 – 0.3 = 0.6 NNH 100/0.6 = 166 45 Benefit vs Harms • Patient S NNT = 100; NNH = 166 • Patient A NNT = 10 ; NNH =166 Benefits vs Harms more favourable with Patient A than in patient S 46 Problem 3 • PEER is 10 %; RRR is 50% • We know that RRR = ARR/CER (PEER) • Also NNT is 100/ARR or ARR = 100/NNT • RRR = 100/NNT x PEER Less confusing to use fractions instead of % RRR = 1/NNT x PEER 47 Problem 3 • NNT = 1/RRR x PEER (RRR is constant) • PEER of 10%; NNT = 1/0.5 x 0.1 = 20 • PEER of 5% ; NNT = 1/0.5 x 0.05 = 40 • PEER of 1 %; NNT = 1/0.5 x 0.01 = 200 • Lower the PEER the higher the NNT 48 NNT/PEER nomograms 49 Generalisability/External validity • How the trial results can be applied in practice • Consider – Trial setting – Selection and exclusion of patients – Patient characteristics – The intervention and trial management – Outcome measures/follow-up – Adverse effects 50 Problem 5 • Consider aspects of clinical trial design to assess the effectiveness of a new antidepressant, i.e. what factors would increase its external validity ? • In trials of nicotine dependence what factors might influence the generalizability of the results ? Hint; consider inclusion and exclusion criteria 51 Summary • Basic understanding of RR, ARR, NNT, NNH and PEER is vital in order to put evidence in to practice • Associated calculations are not difficult • Baseline risk is important when assessing treatment benefits and harms • The generalizability (or lack of) a clinical trial needs to be recognized 52 References • Scott IA, Greenberg PB. Cautionary tales and the interpretation of clinical results. Int Med J 2006;35:611-21 • Barratt A et al. Relative risk reduction, absolute risk reduction, NNT. CMAJ 2004; 171(4):353-58 • The NNT www.thennt.com • Centre for Evidence Based Medicine www.cebm.net DB/FM 27 Mar 2012 53