Supplementary Information (doc 41K)
advertisement

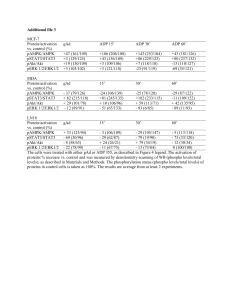
Supplementary methods. Organotypic Invasion Assay and Immunohistochemistry The organotypic invasion assay is described eleswhere (Edward et al., 2005). Briefly, ~7.5x104/ml primary human fibroblasts were embedded in a threedimensional matrix of rat-tail collagen І detached from the culture dish. The collagen matrix was allowed to contract to ~1.5 cm diameter for approximately 6 days in complete medium (10% FCS/DMEM). Subsequently, 4x104 Scc9 cells were plated on top of the matrix in complete medium and allowed to grow for 5 days. The matrix was then mounted on a metal grid and raised to the air/liquid interface resulting in the matrix being fed from below with complete medium that was changed every 2 days. After 8-12 days, the cultures were fixed in 4% formaldehyde. For immunohistochemistry, paraffin embedded tissue was sectioned and stained with hematoxylin and eosin (H&E) using standard methods. Sections were labeled with Ki67 using the Dako EnVision rabbit kit (K4011) and counterstained with hematoxylin. Immunoprecipitation and western blotting. For EGFR immunoprecipitations, cells were grown to confluence and starved overnight with 0.1% normal culture media with or without Ca2+. Cells were stimulated with 25 ng/ml EGF for 10 minutes and lysed on ice with TNE buffer (50mM Tris pH 7.5, 140mM NaCl, 5mM EDTA) containing protease and phosphatase inhibitors. 1 g of EGFR antibody was used to IP from 500 g of protein for 2 hours at 4 0C followed by 20 l protein G beads for 1 hour. Beads were washed 3x in lysis buffer and boiled in 2x SDS-PAGE buffer (Invitrogen). For Western blotting, stimulated cells were lysed and equal amounts of protein were resolved on 4-12% SDS-PAGE gels (Invitrogen) and blotted to PVDF membrane using standard methods. Membranes were blocked and probed over night with the appropriate antibody. Blots were washed and incubated with appropriate secondary antibody for an hour. Blots were then washed extensively and developed using SuperSignal West Pico ECL from (Pierce) using a Syngene Chemigenius gel imaging system. MTT proliferation assay. Cells were seeded at 1 x 104 cells per well of a 12-well plate in (4 replicate samples) and cultured overnight. The following day, cells were switched to 0.1% normal medium containing 10 ng/ml EGF. For each time point, cells were incubated with 1 mg/ml methylthiazolyldiphenyl-tetrazolium bromide (MTT) for 4 hours and the blue formazan product dissolved in DMSO. The absorbance of samples was measured on a spectrophotometer plate reader using 560 nm filter and 690 nm filter for a reference wavelength. For Gefitinib and Erlotinib treatments, 2000 cells were seeded per well of a 96 well plate. The following day drugs were added at increasing concentrations and cells incubated for 5 d. Plates were developed as described above. Data was analyzed in GraphPad Prism and curves were fitted using a sigmoidal doseresponse (variable slope) equation. Significance was calculated using non-linear regression. Antibodies Anti-EGFR for western blotting (1005, Santa Cruz Biotech.), total Akt (cat no. 9272), phosphorylated Akt (cat no. 9271S), total Erk1/2 (cat no. 9102), phosphorylated Erk1/2 (cat no. 9101S) from Cell Signaling Technology. EGFR for immunoprecitation (SC-03, Cat no. 555996, BD Pharminogen), anti-Tubulin was from Sigma-Aldrich (DM1A, cat no. T6199), anti-E-cadherin (cat no. 610182; BD Biosciences, 1:1000) and anti-Ki67 (cat. no. RM-9106-S, Lab Vision, 1/200). Mtss1 antibody is described in (Bompard et al., 2005). Supplementary Figure 1. Mtss1 over-expression does not alter the surface expression of E-cadherin. Scc9 cells ± Mtss1-GFP were starved overnight in 0.1% growth medium, were stimulated with 25 ng/ml EGF for 0, 10 and 30 minutes and their surface proteins biotinylated. (A) Lyastes were probed for Ecadherin expression. (B) Quantification of E-cadherin expression relative to total E-cadherin in Scc9 cells ± Mtss1-GFP showing mean ± S.E.M. from 3 independent experiments. Supplementary Figure 2. Transient over-expression of myc-Mtss1 affects EGF-receptor signaling. HeLa cells were transfected with myc-Mtss1, serum starved overnight and stimulated with 25 ng/ml EGF. Lysates were probed for EGFR (EGF-R), total Akt (t-Akt) and Mtss1 (A) and the expression levels are quantified in (B) by densitometry. Mean ± S.E.M. from 3 experiments is shown relative to Scc9 cells at t = 0 minutes. (C) Lystes were probed for total (tErk1/2) and phosphorylated Erk1/2 (pErk1/2). (D) Graphs show quantification of Erk1/2 phosphorylation by densitometry analysis of western blots. Mean density of bands relative to Scc9 controls at t = 30 minutes ± S.E.M from 3 experiments is shown. Significance in both (B) and (D) was calculated using an unpaired T-test, ** p < 0.01, * p < 0.05. Supplementary Figure 3. Increasing cell density reduces Mtss1-enhanced EGF signaling. (A) Scc9 cells ± Mtss1-GFP were grown to confluence and left for 1-3 days. Lysates were analyzed for basal Erk1/2 and Akt phosphorylation. (B) Quantification of phospho-Erk1/2 and Akt in Scc9 cells ± Mtss1-GFP. Mean intensity is shown normalized to tubulin ± S.E.M. from 3 independent experiments. Significance in (B) was calculated using an unpaired T-test, ** p < 0.01, * p < 0.05. Scc9 cells and Mtss1-GFP expressing cells were grown to confluence, starved overnight and were then stimulated with 25 ng/ml EGF for 30 minutes and lysates prepared and analyzed for Erk1/2 and Akt phosphorylation. (C) Mtss1-expressing Scc9 cells were pre-treated with two rounds of non-targeting (NT) or E-cadherin siRNA and grown to confluence, starved overnight and stimulated with EGF. Lysates were western blotted for phospho- and total-Erk1/2 (pErk1/2 and tErk1/2) and phospho- and total-Akt (pAkt and tAkt). Typical western blots are shown from 3 experiments.