Preparing for Lab (Osmosis in a Potato)
advertisement

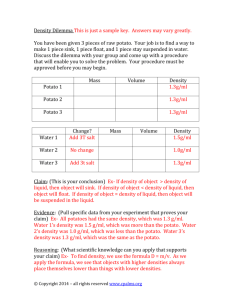
Preparing for Lab (Osmosis in a Potato) Today you will be setting up an experiment that will be completed tomorrow. When you have finished the procedure below, turn in your materials to your teacher for refrigeration overnight. You will be testing some potato tissue (made of living potato cells) to see what happens to it when it is covered with a solution that is less concentrated or more concentrated than the material inside the cells themselves. Use the information you have learned so far to predict what will happen to the pieces of potato you will test. One piece of potato will be covered with pure distilled water. The water has nothing dissolved in it, so it is far less concentrated that the fluid inside the potato. The other piece of potato will be covered with some very salty water. The salt water has a much higher concentration of salt than the fluid inside the potato. Safety: Remember: never taste experimental materials unless expressly told to do so by your instructor. Wash your hands with soap after each lab activity. Leave your lab area in the same condition as you found it. Materials: 1 small potato or ½ medium potato 1 ruler 1 plastic knife 2 zipper seal sandwich bags 100 mL distilled water 100 mL saltwater 1 graduated cylinder 1 balance 1 pen for marking bags Procedure: 1. Mark one bag, “BAG 1 DISTILLED WATER.” Write your initials on the bag. 2. Mark the other bag, “BAG 2 SALTWATER.” Write your initials on the bag. 3. Using a ruler and a knife cut two 1-cm cubes of potato. The pieces should have all the potato skin removed. 4. Weigh one piece of potato and record the mass in your lab log. 5. Place the piece of potato in bag 1. 6. Cover it with 100 mL distilled water. Seal the bag. 7. Weigh the second piece of potato and record its mass in your lab log. 8. Place the second piece of potato in bag 2. 9. Cover it with 100 mL saltwater. 10. In your experimental log, write your hypothesis for bag 1. What do you think will happen to the potato in distilled water? When you weigh it tomorrow, do you think it will weigh less, the same, or more than it does today? In other words, do you think extra water will leave the potato, go into the potato, or go in and out evenly? Write why you chose as you did. 11. Now write your hypothesis for bag 2. What do you think will happen to the potato covered with saltwater? When you weigh it tomorrow, do you think it will weigh less, the same, or more than it does today? Write why you chose as you did. That is all you can do for today. Tomorrow you will weigh the two pieces of potato to see if any changes occurred overnight. Preparing for Lab (Osmosis in an Egg Part 2) Your egg has been soaking in vinegar. It is time to take a look at it and see if there have been any changes. Safety: Raw eggs can be sources of Salmonella bacteria. Wear goggles and gloves when doing this experiment. Wash your hands with soap after each lab activity. Leave your lab area in the same condition as you found it. Procedure: 1. Remove the closed container of egg and vinegar from the refrigerator. 2. Without opening the container, describe, in your experimental log, any changes you may see. 3. Return the unopened container to the refrigerator. Questions: 1. What changes did you observe to the egg and to the vinegar? 2. What evidence did you find that a chemical reaction had occurred? 3. Predict: do you think there will be further changes next time you check the container? If so, what do you think they will be? If not, why do you think things will be the same? That is all you can do for now. Next time, you will use the egg to do an experiment.