Introduction Part A
advertisement

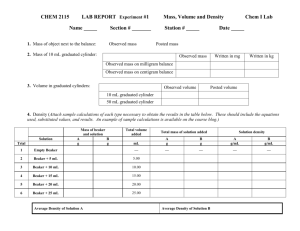
Reaction Rates Purpose: To investigate the effect of concentration, surface area, temperature and the nature of the reactants on the rate of a chemical reaction. Science 10 Materials: marble chips (calcium carbonate chips same size and shape) Powered calcium carbonate beakers HCl solutions (hydrochloric acid) (different concentrations) Thermometer Ethanoic acid Magnesium ribbon graduated cylinder Introduction Part A. Do temperature changes affect the rate of reaction? In this experiment you will investigate the role of temperature in a chemical reaction. This can be done by using different temperatures and recording the relative rates for the compound to react at these different temperatures. Procedure: 1. In beaker, #1 add 25.0 mL of 1M HCl at room temperature (20-25ºC). Place the thermometer in the beaker; stir until the temperature is constant and record temperature. Add one of the marble chips and record the relative rate of reaction. Compare this reaction rate to the rate of the reaction below in number 2. (The rate of reaction is seen by the rate of bubbling.) 2. In beaker #2 add 25 mL of 1M HCl from the hot water bath (at a temperature of 5055ºC). Record temperature of the water bath, and add one of the marble chips to this warm acid. Again, record the relative rate of reaction. Questions: 1. What evidence is there that this is a chemical reaction? 2. What happens to the reaction rate as the temperature increases? 3. Why do you think this happens? Conclusion: Should answer your purpose Introduction Part B Do changes in surface area affect the rate of reaction? In this experiment you will investigate the role of surface area in a chemical reaction. This can be done by using different surface areas of one of the reactants and measuring the time required for the the reactant compound to disappear. Purpose: To investigate the effect of surface area on the reaction rate. Procedure: 1. Add 25.0 mL of 1M HCl to beaker#1 and beaker#2. 2. Obtain a calcium carbonate chip and equal volume of powered calcium carbonate. 3. Add the chip to beaker#1 and the power to beaker#2. Compare the rates of reaction. Questions: 1. Which sample had the greatest exposed surface area? Why? 2. What happened to the reaction rate as the surface area increased? 3. Why do you think this happened? Conclusion: Should answer purpose. 1 Reaction Rates Science 10 Introduction Part C In order to investigate the effect of changing the concentration of one of the reactants on the reaction time you will prepare dilutions of the hydrochloric acid solution. Purpose: To investigate the effect of concentration on the reaction rate. Procedure: 1. Obtain three solutions of hydrochloric acid as follows. Beaker #1 50 mL of 0.5 M HCL acid Beaker #2 50 mL of 1.0 M HCL acid. Beaker #3 50 mL of 2.0 M HCL acid 2. Obtain three pieces of magnesium ribbon, each one centimetre long. 3. Add a piece of magnesium to each beaker and compare the rates of reaction for each solution. Questions: 1. Write a balanced chemical equation for this reaction. 2. How does the reaction rate change as the concentration increases? 3. Explain why you think this happens. Conclusion: Should answer purpose. Introduction Part D Does the reactant used change the rate of reaction? In this experiment you will investigate the role of the reactan in a chemical reaction. This can be done by using different reactantss and recording the relative rates for each of the different reactants. We will use different acids and react them with the same compound, calcium carbonate. Procedure: 1. Add 25.0 mL of 2M HCl to beaker#1 and 25.0 mL of 2M CH3COOH to beaker beaker#2. 2. Obtain two calcium carbonate chips equal in size and shape. 3. At the same time add the chip to beaker#1 and to beaker#2. Compare the rates of reaction. Questions: 1. Write a balanced chemical equation for these reactions. 2. How does the reactant affect the reaction rate? 3. Explain why you think this happens. Conclusion: Should answer purpose. 2