Experiment: To Verify Boyle`s Law - Dominican

Experiment: To Verify Boyle’s Law
Materials used: A Boyle’s Law Apparatus, Air Pump, Hand Vacuum
Pump.
Method:
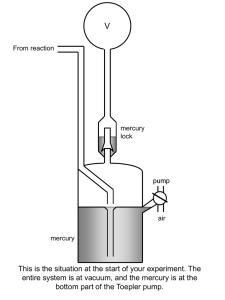
1) Set up apparatus as shown in the diagram.
2) Connect air pump to the in let of the oil reservoir.
3) Open air tap and pump in air until pressure gauge reads its max value.
4) Quickly close tap.
5) Leave for a minute or two, after changing the pressure of the trapped air wait a minute or two before reading the pressure or volume, to allow the air to reach room temperature. This is necessary because when the air is compressed or expanded there may be slight changes in temperature.
6) Then measure the gas pressure by reading it off the Bourdon gauge. Record these volumes.
7) Read the volume of gas off the scale next to the glass tube.
8) Gently open then quickly close tap to release some air.
9) Leave for a minute or two, then measure the gas pressure by reading it off the Bourdon gauge.
10) Record these values.
11) Repeat steps 7-9 at least six times until the pressure of the gas is
back to atmospheric pressure
12) Plot a graph of pressure (p) against the inverse of volume (1/V)
Results:
The graph is a straight line through the origin, verifying that the pressure
(p) is proportional to (1/V), verifying Boyles Law. Also all values of pV are the same.
P α 1/V therefore p=k(1/V) therefore pV =k
Results:
Pressure p/ atms
120
180
220
280
320
380
440
Volume
V/ cm 3
9.0
6.0
5.0
4.0
3.5
3.0
2.5
1
Volume / cm -3
0.11
0.17
0.20
0.25
0.29
0.33
0.40
(Based on 2003 figures given)
pV=k
To graph p against 1/V
500
450
400
350
300
250
200
150
100
50
0
0 0.05
0.1
0.15
0.2
0.25
0.3
0.35
0.4
0.45
Boyles Law States……. That at constant temperature the volume of a fixed mass of gas is inversely proportional to its pressure.
P α 1/V
This proves Boyles law as it is a straight line graph through the origin. That graph is P α 1/V
Precautions:
1) After changing the pressure of the trapped air wait a minute or two before reading the pressure or volume, to allow the air to reach room temperature. This is necessary because when the air is compressed or expanded there may be slight changes in temperature which will affect t the volume of gas )due to expansion or contraction)
2) When reading volume make sure your eye is level with the Mercury
Meniscus.
3) Make sure air is connected tightly to oil reservoir in let.
4) If a hand suction pump is available you will be able to reduce the pressure of the gas below atmospheric pressure. You can then take a future series of values of p and V include them in the table and graph.
Problems:
Q1.
The volume of a certain mass of gas is 600cm 3 at a pressure of
1 x 10 5 Pa. Find its volume if the pressure changes to 3.2 x 10 5 Pa, the temperature remaining constant.
pV = constant therefore p
1
V
1
= p
2
V
2
where V
2 is the required volume. Thus (1 x 10 5 )(600) = (3.2 x 10 5 ) ( V
2
) therefore
V
2
= 187.5 cm 3 .
Q2.
The height of mercury in a mercury barometer was 75cm on a certain day. Given that the density of mercury is 1.36 x 10 4 kgm -3 and g =9.8ms
-2 what was the atmospheric pressure on that day?
P = ρgh = (1.36 x 10 4) (9.8)(0.75) = 99960 Pa.
Q3. If a gas under the same atmospheric pressure as above had a volume of 15cm 3 , what volume would it have if the pressure on it increased to 1.2x 10 5 Pa and the temperature remained the same?
From Boyle’s Law, p
1
V
1
= p
2
V
2
therefore V
2
= p
1
V
1
P
2
Therefore: V
2
= 99960 x 15 = 12.5 cm 3
1.2 x 10 5