Density Lab (Teacher Copy) Florida Sunshine State Standards
advertisement

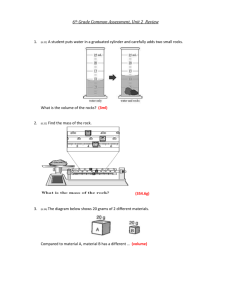
Density Lab (Teacher Copy) Florida Sunshine State Standards Benchmark: SC.A.1.3.1 AA The student identifies various ways in which substance differ (e.g., mass, volume, shape, density, texture, and reaction to temperature and light). (Also assesses A.1.3.2 and A.1.3.6) MC, GR Objective: SC.A.1.3.1 I can calculate the density of an irregularly shaped object using a triple beam balance, water displacement and the density equation: mass/volume (D=m/v). Background Knowledge: All matter has mass. Mass is a physical property that is a measure of the amount of matter in an object. Matter also takes up space. The measure of the amount of space that matter occupies is a physical property called volume. In science, milliliters (mL) and cubic centimeters (cm³) are often used to express volume. One milliliter is equal to one cubic centimeter (1 mL = 1 cm³). Density is the mass of an object per unit of volume. Density equals mass divided by volume. Determining the volume of an object depends on its shape. A rectangular prism will use the formula: volume = length x width x height. The volume of a cube will use the formula: volume = side x side x side or s³. The water displacement method is used to measure the volume of an irregularly shaped object. For example, to find the volume of a rock, simply fill a graduated cylinder with water to a specific level such as 50 mL. Adding the rock to the graduated cylinder causes the water level to rise to 65 mL. So, the rock displaces 15 mL of water. Since 1 mL = 1 cm³, the volume of the rock is 15 cm³. Graduated cylinders are used to measure the liquid volume when accuracy is important. The surface of a liquid in a container may be curved. The curve at the surface of the liquid is called the meniscus. To measure the volume of liquids you must look at the bottom of the meniscus. Explain to the students that a triple beam balance is an expensive piece of equipment, and they should use it properly. Make sure all the balances are calibrated to zero and adjust as needed. There is a PowerPoint presentation called Mass Presentation (PPT) on the Science Spot website http://sciencespot.net/Pages/classmetric.html. You may choose to use this presentation to review the basic units of mass and how to measure mass using a triplebeam balance. Make sure to click on the on-line activity at the bottom of the PowerPoint and try it out. Engage: Give the students the following question as a challenge. They will determine if the two substances are gold by using the values given in the problem. As a visual aid, you can label two substances that look like gold. Show the students substance (A) and then show them substance (B). (Question is on the next page.) Cristian is trekking through a desert; he stumbles across a substance (A) that resembles gold. Later in the day he stumbles across another substance that looks similar to the first, substance (B). Since he wants to cash in on his discoveries, how can he ensure that what he has is gold? Gold’s density: 19.32 g/cm³ Substance A: m=120g v= 6.21cm³ Substance B: m=120g v= 5.1cm³ Problem Statement: How can you calculate the density of an irregularly shaped object? Hypothesis: Have the students write a hypothesis. For example: If I use the water displacement method and a triple beam balance, then I can determine the density of the unknown substance. If (independent variable), then (dependent variable) Materials: Each group will need: triple beam balance graduated cylinder or beaker rock samples labeled A, B, C and D (Use a variety of rocks, for example: granite, rose quartz, limestone, etc.) Density Lab Sheet for each student Explore: Procedures: 1. Hand out the Density Lab Sheet to each student. 2. Give each group one rock sample labeled A, B, C or D. 3. Have them identify on their lab sheet which rock sample they received. For example: Object A, B, C, or D. 4. Draw the rock in the space provided on the lab observation sheet. Tracing is acceptable for accuracy of shape. 5. Instruct students to measure the mass of the rock using the triple beam balance. Find the measurement of the object three times for accuracy. 6. Have the students then record the mass in grams (g) to the nearest tenths place on data table 1. 7. Next the students will measure the volume of the rock using the water displacement method. Repeat the water displacement method three times for accuracy. 8. Instruct students to record the volume in mL on the data table 2. 9. The students will calculate the density for each trial and record on data table 3. 2 10. 11. 12. 13. The students will average the three densities and record on data table 3. As a class complete data table 4. Complete Density of Different Rock Samples graph. Complete all the questions on the lab sheet. Explain: Students will answer the questions and complete the conclusion using the Density Lab Sheet. Rock Sample: ___________ Rock Drawing: ________ Mass of Rock: Data Table 1 Rock Sample Mass Trial 1 (g) Volume of Rock: Data Table 2 Mass Trial 2 (g) Mass Trial 3 (g) *mL is used for liquids, cm³ is used for solids Trial 1 (mL) Trial 2 (mL) Trial 3 (mL) Original amount of water in cylinder: Water level in cylinder + rock: Volume of rock: Density of Rock: Data Table 3 Density of Density of Rock Sample Trial 1 Trial 2 (g/mL) (g/mL) Density of Trial 3 (g/mL) Average Calculated Density (g/mL) Density Calculations (Show all work and include units) 3 Class Data: Data Table 4 Average Calculated Density (g/mL) Rock Sample Rock Sample A Rock Sample B Rock Sample C Rock Sample D Density of Different Rock Samples: Density of Rock Sample Rock Samples 4 Elaborate: Option 1: Students may explore the Gizmo Determining Density Via Water Displacement. Option 2: Using density cubes, have the students write a hypothesis for the following problem statement: The density cubes have the same volume, but different masses. Therefore, will the density be the same or different for each cube? Explain. Evaluate: The teacher may use the student’s data sheet, questions, and conclusion as an evaluation. FCAT Style Question Melanie measured the mass of 5 cm³ of each of the substances and recorded the data as follows: Aluminum 13.5 grams Diamond 17.5 grams Water 5.0 grams Wax 4.5 grams Which substance has the highest density? a. Aluminum b. Diamond c. Water d. Wax Answer: b. Diamond 5 Scientist: ____________________________ Date: ____________ Period: _____ Density Lab (Student Copy) Objective: SC.A.1.3.1 I can calculate the density of an irregularly shaped object using a triple beam balance, water displacement, and the density equation: mass/volume (D=m/v). Background Information: All matter has mass. Mass is a physical property that is a measure of the amount of matter in an object. Matter also takes up space. The measure of the amount of space that matter occupies is a physical property called volume. In science, milliliters (mL) and cubic centimeters (cm³) are often used to express volume. One millimeter is equal to one cubic centimeter (1 mL = 1 cm³). Density is the mass of an object per unit of volume. Density equals mass divided by volume. The water displacement method is used to measure the volume of an irregularly shaped object. For example, to find the volume of a rock, simply fill a graduated cylinder with water to a specific level such as 50 mL. Adding the rock to the graduated cylinder causes the water level to rise to 65 mL. So, the rock displaces 15 mL of water. Since 1 mL = 1 cm³, the volume of the rock is 15 cm³. Graduated cylinders are used to measure the liquid volume when accuracy is important. The surface of a liquid in a container may be curved. The curve at the surface of the liquid is called the meniscus. To measure the volume of liquids you must look at the bottom of the meniscus. Problem Statement: How can you calculate the density of an irregularly shaped object? Hypothesis: If (independent variable), then (dependent variable) 6 Materials: triple beam balance graduated cylinder or beaker a rock sample labeled A, B, C and D Density Lab Sheet for each student Procedures: 1. Identify on your lab sheet which rock sample you have. For example: Object A, B, C, or D. 2. Draw the rock in the space provided on the lab observation sheet. Tracing is acceptable for accuracy of shape. 3. Measure the mass of the rock using the triple beam balance. Find the measurement of the object three times for accuracy. 4. Record the mass in grams (g) to the nearest tenths place on data table 1. 5. Measure the volume of the rock using the water displacement method. Repeat the water displacement method three times for accuracy. 6. Record the volume in mL on the data table 2. 7. Calculate the density for each trial and record on data table 3. 8. Average the three densities and record on data table 3. 9. As a class complete data table 4. 10. Complete Density of Different Rock Samples graph. 11. Complete all the questions on the lab sheet. 7 Rock Sample: ___________ Rock Drawing: ________ Mass of Rock: Data Table 1 Rock Sample Mass Trial 1 (g) Volume of Rock: Data Table 2 Mass Trial 2 (g) Mass Trial 3 (g) *mL is used for liquids, cm³ is used for solids Trial 1 (mL) Trial 2 (mL) Trial 3 (mL) Original amount of water in cylinder: Water level in cylinder + rock: Volume of rock: Density of Rock: Data Table 3 Rock Sample Density of Trial 1 (g/mL) Density of Trial 2 (g/mL) Density of Trial 3 (g/mL) Average Calculated Density (g/mL) Density Calculations (Show all work and include units) 8 Class Data: Data Table 4 Average Calculated Density (g/mL) Rock Sample Rock Sample A Rock Sample B Rock Sample C Rock Sample D Density of Different Rock Samples: Density of Rock Sample Rock Samples 9 Results and Conclusions: 1. What is the type of tool used to measure the volume of liquids or irregular solids? 2. What is the meniscus? 3. Explain the water displacement method. 4. Outline how to determine the density of an irregularly shaped object. 5. Why is density an important physical property of matter? 6. Based on the results of this lab, develop a hypothesis about how unknown substances can be distinguished from one another by using their densities. 10