25263 Demonstrate knowledge of human digestive
advertisement

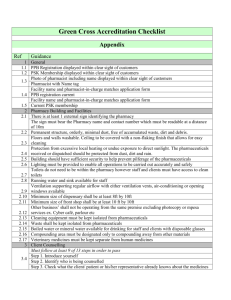
NZQA Expiring unit standard 25263 version 2 Page 1 of 6 Title Demonstrate knowledge of human digestive, reproductive, and urinary systems and medicines used in their treatment Level 5 Purpose Credits 9 This unit standard is intended for pharmacy technicians or people working towards a pharmacy technician qualification. People credited with this unit standard are able to demonstrate knowledge of: the structure and function of the digestive system; the structure and function of the reproductive system; the structure and function of the urinary system; digestive system disorders and medicines used in their treatment; medicines used in the treatment of reproductive system disorders and contraceptive medicines used for manipulation of reproductive function; and urinary system disorders and medicines used in their treatment. Classification Pharmacy > Pharmacy Services Available grade Achieved Entry information Critical health and safety prerequisites This unit standard is Explanatory notes expiring Pharmacy technicians must act within the legislative limits of their role as specified by the Open. Medicines Regulations 1984 and must always act under the supervision of a pharmacist and under the direct personal supervision of a pharmacist when dispensing or compounding. 2 Legislation, codes, and standards relevant to this unit standard include: Consumer Guarantees Act 1993; Fair Trading Act 1986; Hazardous Substances and New Organisms Act 1996; Health and Disability Commissioner (Code of Health and Disability Services Consumers’ Rights) Regulations 1996; Code of Health and Disability Services Consumers’ Rights, available from the Health and Disability Commissioner’s office; Health Information Privacy Code 1994, available from the Privacy Commissioner’s office; Health (Retention of Health Information) Regulations 1996; Health (Needles and Syringes) Regulations 1998; Pharmacy Industry Training Organisation SSB Code 102150 New Zealand Qualifications Authority 2016 NZQA Expiring unit standard 25263 version 2 Page 2 of 6 Health and Safety in Employment Act 1992; Health and Safety in Employment Regulations 1995; Medicines Act 1981; Medicines Regulations 1984; Misuse of Drugs Act 1975; Privacy Act 1993; Misuse of Drugs Regulations 1977; and any subsequent amendments. New Zealand Code of Good Manufacturing Practice for Manufacture and Distribution of Therapeutic Goods, Part 3 Compounding and Dispensing (1993), available from Medsafe; The New Zealand Pharmaceutical Schedule, available from http://www.pharmac.govt.nz; Pharmacy Council of New Zealand Code of Ethics 2004, available at http://www.pharmacycouncil.org.nz. 3 Access to appropriate reference resources in accordance with Medsafe audit requirements is required for completion of assessment against this unit standard. Evidence presented by the candidate must be in accordance with reference resources. 4 This unit standard is designed to cover knowledge sufficient to provide, under direct personal supervision of a pharmacist, client counselling when dispensed medicine is handed out. The Pharmacy Council advises the public that before leaving the pharmacy they will receive counselling that confirms and explains: who the medicine is for what the medicine is for the directions for proper use and what special precautions to take foods or medicines to avoid common side effects or interactions, what to expect and how to treat, if needed how soon to expect the medicine to work what to do if you miss a dose how to store your medicine and dispose of unused medicines safely if there are repeats or a balance owing and how and when to collect them. This unit standard is This information is available on the Pharmacy Council website: www.pharmacycouncil.org.nz.expiring Digestive disorders include but are not limited to – constipation, diarrhoea, gastric ulcer, gastro-oesophageal reflux, haemorrhoids, indigestion, inflammatory bowel disease, mouth and throat disorders, spasm, vomiting. Reproductive disorders include but are not limited to – endometriosis, erectile dysfunction, infection, infertility, menstrual disorders, morning sickness, polycystic ovary syndrome. Urinary disorders include but are not limited to – calculi, cystitis, enuresis, frequency, incontinence, infection, retention. 5 6 Manipulation of the reproductive function includes but is not limited to – contraception, fertility treatment, labour. Outcomes and evidence requirements Outcome 1 Pharmacy Industry Training Organisation SSB Code 102150 New Zealand Qualifications Authority 2016 NZQA Expiring unit standard 25263 version 2 Page 3 of 6 Demonstrate knowledge of the structure and function of the digestive system. Evidence requirements 1.1 The location and structure of the parts of the digestive system and accessory glands of digestion are identified and explained in terms of their function. 1.2 Digestion is explained in terms of the mechanical movements and chemical processes of the gastrointestinal tract. Outcome 2 Demonstrate knowledge of the structure and function of the reproductive system. Evidence requirements 2.1 The location and structure of parts of the female and male reproductive systems are identified and explained in terms of their function. 2.2 The menstrual cycle is explained in terms of uterine, ovarian, hormonal, and physiological changes. 2.3 Spermatogenesis and ejaculation are explained in terms of physiological processes and the role of hormonal and neural activity. Outcome 3 Demonstrate knowledge of the structure and function of the urinary system. Evidence requirements 3.1 3.2 This unit standard is The functions of the urinary system are explained in terms of physiological expiring processes. The location and structure of parts of the male and female urinary systems are identified and explained in terms of their function. Range 3.3 includes but is not limited to – water balance, electrolyte balance, removal of waste products. The urination process is explained in terms of mechanisms and control of the processes. Range includes but is not limited to – renin-angiotensin system. Outcome 4 Demonstrate knowledge of digestive system disorders and medicines used in their treatment. Pharmacy Industry Training Organisation SSB Code 102150 New Zealand Qualifications Authority 2016 NZQA Expiring unit standard Range 25263 version 2 Page 4 of 6 medicines include but are not limited to – alginates, aminosalicylates, antacids, antidiarrhoeals, anti-nausea, antispasmodics, combination therapy for Helicobactor Pylori eradication, haemorrhoidal preparations, H2-receptor antagonists, laxatives, motility stimulants, oropharyngeal preparations, proton pump inhibitors. Evidence requirements 4.1 Digestive disorders and their signs and symtoms are identified and explained in terms of the effects on body functions. 4.2 Medicines are identified according to their use for treatment of digestive disorders. 4.3 Actions and limitations of the medicines are identified and explained in terms of their effect on body functions. Range 4.4 may include but is not limited to – cautions, contra-indications, clinically significant adverse effects, drug interactions. Type of advice required by the Pharmacy Council for pharmacy clients is identified Range evidence is required for a minimum of one treatment for a digestive disorder. Outcome 5 Demonstrate knowledge of medicines used in the treatment of reproductive system disorders and contraceptive medicines used for manipulation of reproductive function. Range medicines include but are not limited to – contraceptives, fertility treatment, hormonal preparations, hormone antagonists, hormone replacement therapy, erectile dysfunction therapy, prostaglandins. 5.1 Reproductive disorders and their signs and symptoms are identified and explained in terms of the effects on body functions. 5.2 Medicines and contraceptive methods are identified according to their use for treatment of reproductive disorders and for manipulation of reproductive function. 5.3 Actions and limitations of the medicines and contraceptive methods are identified and explained in terms of the effects on body functions. This unit standard is Evidence requirements expiring Range may include but is not limited to – cautions, contra-indications, clinically significant adverse effects, drug interactions, contraceptive reliability and reversibility. Pharmacy Industry Training Organisation SSB Code 102150 New Zealand Qualifications Authority 2016 NZQA Expiring unit standard 5.4 25263 version 2 Page 5 of 6 Type of advice required by the Pharmacy Council for pharmacy clients is identified Range evidence is required for a minimum of one treatment for a reproductive disorder and one treatment for the manipulation of reproductive function. Outcome 6 Demonstrate knowledge of urinary system disorders and medicines used in their treatment. Range medicines include but are not limited to – alpha-blockers, diuretics (thiazide, carbonic anhydrase inhibitor, loop, potassium-sparing, spironolactone), parasympathomimetics, antimuscarinics, tricyclics, urinary alkinisers. Evidence requirements 6.1 Urinary disorders and their signs and symptoms are identified and explained in terms of the effects on body functions. 6.2 Medicines are identified according to their use for treatment of urinary disorders. 6.3 Actions and limitations of the medicines are identified and explained in terms of their effects on body functions. may include but is not limited to – cautions, contra-indications, clinically significant adverse effects, drug interactions. Range 6.4 Type of advice required by the Pharmacy Council for pharmacy clients is identified evidence is required for a minimum of one treatment for a urinary This unit standard is disorder. expiring Range Replacement information Dummie Replacement Information This unit standard is expiring. Assessment against the standard must take place by the last date for assessment set out below. Status information and last date for assessment for superseded versions Process Version Date Last Date for Assessment Registration 1 21 November 2008 31 December 2017 Review 2 16 April 2015 31 December 2017 Pharmacy Industry Training Organisation SSB Code 102150 New Zealand Qualifications Authority 2016 NZQA Expiring unit standard 25263 version 2 Page 6 of 6 Consent and Moderation Requirements (CMR) reference 0128 This CMR can be accessed at http://www.nzqa.govt.nz/framework/search/index.do. Please note Providers must be granted consent to assess against standards (accredited) by NZQA, before they can report credits from assessment against unit standards or deliver courses of study leading to that assessment. Industry Training Organisations must be granted consent to assess against standards by NZQA before they can register credits from assessment against unit standards. Providers and Industry Training Organisations, which have been granted consent and which are assessing against unit standards must engage with the moderation system that applies to those standards. Requirements for consent to assess and an outline of the moderation system that applies to this standard are outlined in the Consent and Moderation Requirements (CMR). The CMR also includes useful information about special requirements for organisations wishing to develop education and training programmes, such as minimum qualifications for tutors and assessors, and special resource requirements. This unit standard is expiring Pharmacy Industry Training Organisation SSB Code 102150 New Zealand Qualifications Authority 2016